I MEMBRANE BIOCHEMISTRY 1 2 Polysaccharides 1 2

I. MEMBRANE BIOCHEMISTRY § 1. 2 Polysaccharides § 1. 2 a Disaccharides § 1. 2 b Homoglycans § 1. 2 c Heteroglycans
§ 1. 2 a Disaccharides

Synopsis 1. 2 a - Polysaccharides (n > 1) are usually classified according to the number (n) of sugar units they harbor: Monosaccharide => n=1 Disaccharide => n=2 Oligosaccharide => 3 n < 10* Polysaccharide => n 10* *somewhat subjective but certainly no greater than 20! - Disaccharides are the simplest form of polysaccharides - Polysaccharides (also called glycans) are comprised of two or more monosaccharides covalently linked together via O-glycosidic bonds - Polysaccharides (as well as monosaccharides) often covalently associate with proteins and lipids to form highly complex macromolecules called glycoconjugates —such as glycoproteins, proteoglycans, peptidoglycans, glycolipids, and lipopolysaccharides (outer membrane of Gram –ve bacteria) - In addition to their role as common sugars, disaccharides also play important roles as medicines and food additives - Common disaccharides include lactose, sucrose, maltose, lactulose, and trehalose
![Lactose—The Milk Sugar ( 1 4) -D-Galactose -D-Glucose [β-D-galactopyranosyl-(1→ 4)- -D-glucopyranose] - Lactose is Lactose—The Milk Sugar ( 1 4) -D-Galactose -D-Glucose [β-D-galactopyranosyl-(1→ 4)- -D-glucopyranose] - Lactose is](http://slidetodoc.com/presentation_image_h2/ba472c8084348be63489b631aaf80aab/image-4.jpg)
Lactose—The Milk Sugar ( 1 4) -D-Galactose -D-Glucose [β-D-galactopyranosyl-(1→ 4)- -D-glucopyranose] - Lactose is formed via the ( 1 4) glycosidic linkage between galactose ( -D-galactopyranose) and glucose ( -D-glucopyranose) - ( 1 4) glycosidic linkage must be stated within the parentheses along with the position of linked C atoms and their anomericity (or that of constituent sugars) - Lactose is the milk sugar and can make up as much as 10%(w/w) of milk—the suffix (w/w) signifies the %age in terms of “weight per unit weight”—ie 10 g of lactose for every 100 g of dried milk—what would be implied by 10%(v/v)? 10 ml out of 100 ml! - Oxidation of lactose to lactate by lactobacillus turns the milk sour—the sour taste is due to lactate and the rather high concentration of lactobacilli is the reason why sour milk (expired) is so beneficial to health—lactobacilli are probiotics in that they promote gut health and play a role in digestion! - Lac is Latin for milk—nb leche (Spanish), latte (Italian), lait (French), milch (German), et cetera
![Sucrose—The Table Sugar ( 1 2) -D-Glucose -D-Fructose [ -D-glucopyranosyl-(1→ 2)- -D-fructofuranose] - Sucrose Sucrose—The Table Sugar ( 1 2) -D-Glucose -D-Fructose [ -D-glucopyranosyl-(1→ 2)- -D-fructofuranose] - Sucrose](http://slidetodoc.com/presentation_image_h2/ba472c8084348be63489b631aaf80aab/image-5.jpg)
Sucrose—The Table Sugar ( 1 2) -D-Glucose -D-Fructose [ -D-glucopyranosyl-(1→ 2)- -D-fructofuranose] - Sucrose is formed via the ( 1 2) glycosidic linkage between glucose ( -Dglucopyranose) and fructose ( -D-fructofuranose)—the fruit sugar - ( 1 2) glycosidic linkage must be stated within the parentheses along with the position of linked C atoms and their anomericity (or that of constituent sugars) - Sucrose is the table sugar extracted from sugarcane or sugarbeet—the name is derived from the French word for sugar (sucre)—nb zachari (Greek), sukar (Arabic), shakar (Urindi), azucar (Spanish), zucchero (Italian), zucker (German), et cetera
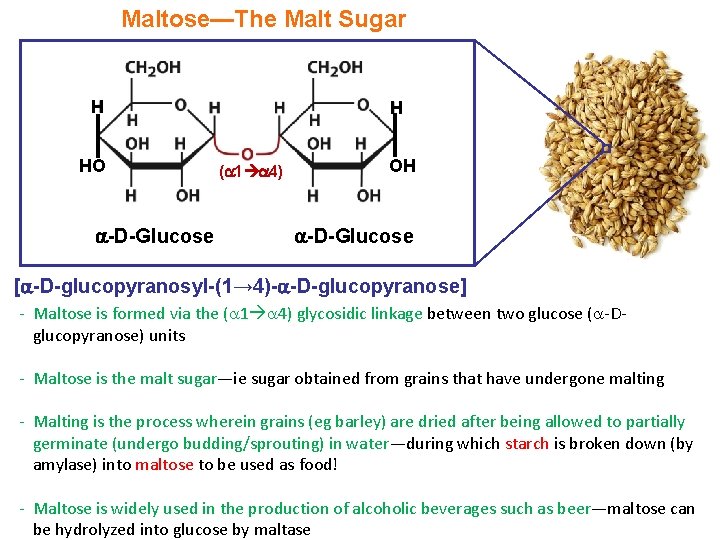
Maltose—The Malt Sugar H HO -D-Glucose H ( 1 4) OH -D-Glucose [ -D-glucopyranosyl-(1→ 4)- -D-glucopyranose] - Maltose is formed via the ( 1 4) glycosidic linkage between two glucose ( -Dglucopyranose) units - Maltose is the malt sugar—ie sugar obtained from grains that have undergone malting - Malting is the process wherein grains (eg barley) are dried after being allowed to partially germinate (undergo budding/sprouting) in water—during which starch is broken down (by amylase) into maltose to be used as food! - Maltose is widely used in the production of alcoholic beverages such as beer—maltose can be hydrolyzed into glucose by maltase
![Lactulose—A Laxative -D-Fructose ( 1 4) -D-Galactose [ -D-galactopyranosyl-(1→ 4)- -D-fructofuranose] - Lactulose is Lactulose—A Laxative -D-Fructose ( 1 4) -D-Galactose [ -D-galactopyranosyl-(1→ 4)- -D-fructofuranose] - Lactulose is](http://slidetodoc.com/presentation_image_h2/ba472c8084348be63489b631aaf80aab/image-7.jpg)
Lactulose—A Laxative -D-Fructose ( 1 4) -D-Galactose [ -D-galactopyranosyl-(1→ 4)- -D-fructofuranose] - Lactulose is formed via the ( 1 4) glycosidic linkage between galactose (β-D-galactopyranose) and fructose ( -D-fructofuranose) - Produced commercially by isomerization of lactose, lactulose is a non-absorbable sugar used as a laxative in the treatment of constipation—it is on the WHO List of Essential Medicines! - Heating milk can trigger isomerization of lactose to lactulose—the greater the heat, the greater the amount of lactulose formed (drinking warm milk is indeed beneficial!) - Lactulose is not absorbed in the small intestine nor broken down by human enzymes—thus stays in the digestive tract through most of its course, causing retention of water through osmosis leading to softer-and-easier-to-pass stool (aids bowel movement)
![Trehalose—A Life Saver ( 1 1) -D-Glucose [ -D-glucopyranosyl-(1→ 1)- -D-glucopyranose] - Trehalose is Trehalose—A Life Saver ( 1 1) -D-Glucose [ -D-glucopyranosyl-(1→ 1)- -D-glucopyranose] - Trehalose is](http://slidetodoc.com/presentation_image_h2/ba472c8084348be63489b631aaf80aab/image-8.jpg)
Trehalose—A Life Saver ( 1 1) -D-Glucose [ -D-glucopyranosyl-(1→ 1)- -D-glucopyranose] - Trehalose is formed via the ( 1 1) glycosidic linkage between two glucose ( -Dglucopyranose) units - Particularly prevalent in shrimp and insects where it is implicated in anhydrobiosis—the ability of organisms to withstand prolonged periods of desiccation/dehydration - The so-called “brine shrimps” can live for millennia —thanks to trehalose, which not only makes up over 10%(w/w) of their dry weight but also helps them survive dehydration! - Trehalose harbors high water retention capability—thus widely used in food and cosmetics
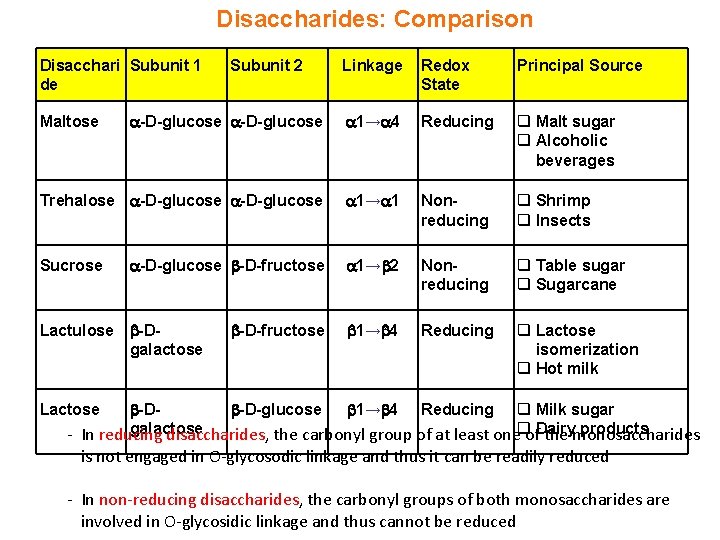
Disaccharides: Comparison Disacchari Subunit 1 de Linkage Redox State Principal Source -D-glucose 1→ 4 Reducing q Malt sugar q Alcoholic beverages Trehalose -D-glucose 1→ 1 Nonreducing q Shrimp q Insects -D-glucose -D-fructose 1→ 2 Nonreducing q Table sugar q Sugarcane 1→ 4 Reducing q Lactose isomerization q Hot milk Maltose Sucrose Lactulose -Dgalactose Subunit 2 -D-fructose -D -D-glucose 1→ 4 Reducing q Milk sugar galactose products - In reducing disaccharides, the carbonyl group of at least oneqof. Dairy the monosaccharides Lactose is not engaged in O-glycosodic linkage and thus it can be readily reduced - In non-reducing disaccharides, the carbonyl groups of both monosaccharides are involved in O-glycosidic linkage and thus cannot be reduced

Exercise 1. 2 a - Describe the monosaccharide units and their linkages in the common disaccharides - Explain why the systematic name of a disaccharide must include more than just the names of the component monosaccharides - Compare and contrast the structures and functions of lactose, sucrose, maltose, lactulose, and trehalose
§ 1. 2 b Homoglycans

Synopsis 1. 2 b - Polysaccharides are categorized according to whether they harbor identical (homoglycans) or non-identical (heteroglycans) sugar units (or residues) - Unlike their more illustrious cousins in proteins and nucleic acids, homoglycans not only form linear polymers but also branched chains—this is due to the fact that their polyhydroxy feature enables them to engage in the formation of multiple O-glycosidic bonds on the same monosaccharide - Homoglycans play key cellular roles as “fuel storage” as well as “structural components” - Common homoglycans include starch and glycogen (fuel storage) as well as cellulose and chitin (structural components)
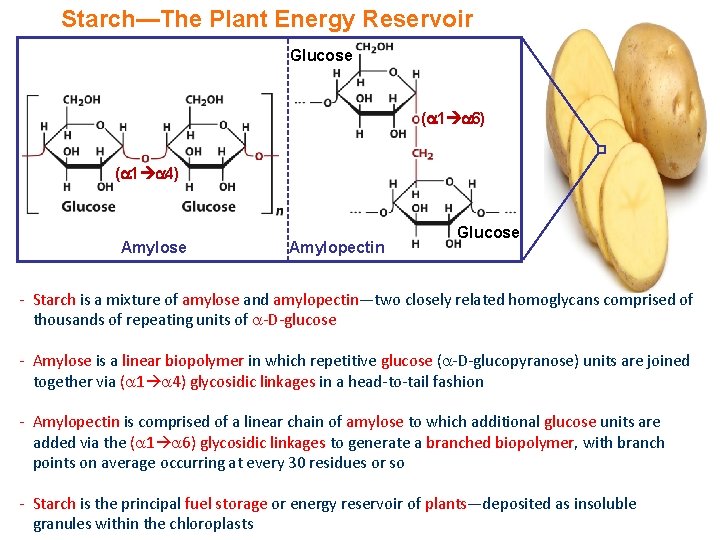
Starch—The Plant Energy Reservoir Glucose ( 1 6) ( 1 4) Amylose Amylopectin Glucose - Starch is a mixture of amylose and amylopectin—two closely related homoglycans comprised of thousands of repeating units of -D-glucose - Amylose is a linear biopolymer in which repetitive glucose ( -D-glucopyranose) units are joined together via ( 1 4) glycosidic linkages in a head-to-tail fashion - Amylopectin is comprised of a linear chain of amylose to which additional glucose units are added via the ( 1 6) glycosidic linkages to generate a branched biopolymer, with branch points on average occurring at every 30 residues or so - Starch is the principal fuel storage or energy reservoir of plants—deposited as insoluble granules within the chloroplasts
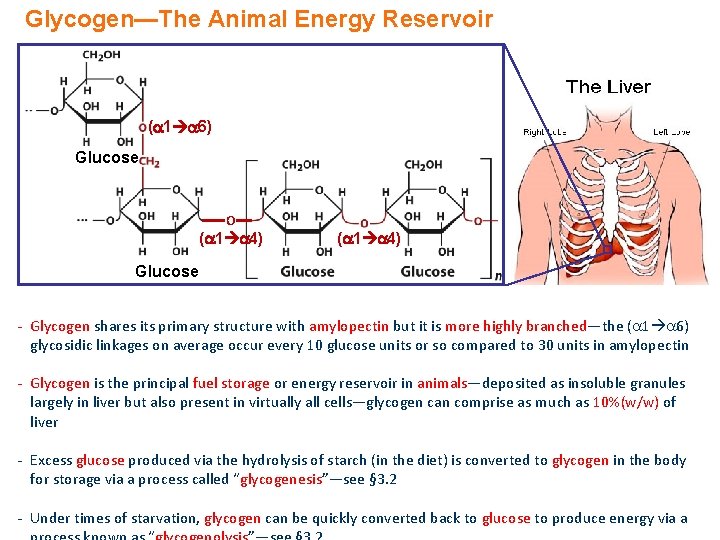
Glycogen—The Animal Energy Reservoir ( 1 6) Glucose O ( 1 4) Glucose - Glycogen shares its primary structure with amylopectin but it is more highly branched—the ( 1 6) glycosidic linkages on average occur every 10 glucose units or so compared to 30 units in amylopectin - Glycogen is the principal fuel storage or energy reservoir in animals—deposited as insoluble granules largely in liver but also present in virtually all cells—glycogen can comprise as much as 10%(w/w) of liver - Excess glucose produced via the hydrolysis of starch (in the diet) is converted to glycogen in the body for storage via a process called “glycogenesis”—see § 3. 2 - Under times of starvation, glycogen can be quickly converted back to glucose to produce energy via a
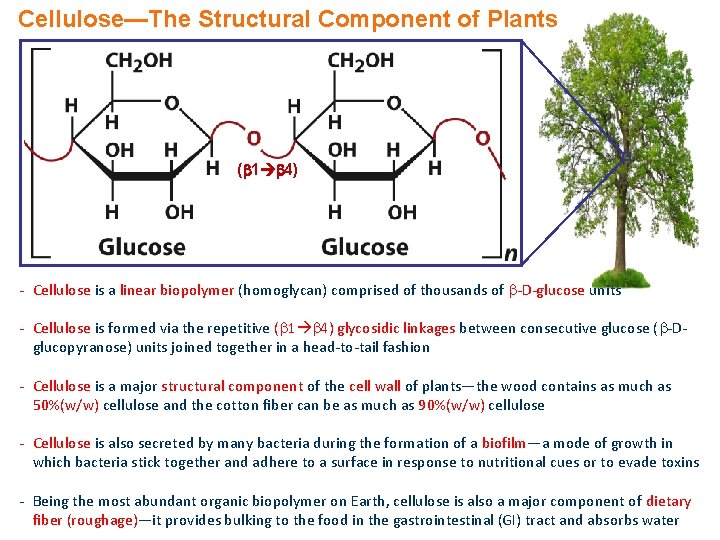
Cellulose—The Structural Component of Plants ( 1 4) - Cellulose is a linear biopolymer (homoglycan) comprised of thousands of -D-glucose units - Cellulose is formed via the repetitive ( 1 4) glycosidic linkages between consecutive glucose ( -Dglucopyranose) units joined together in a head-to-tail fashion - Cellulose is a major structural component of the cell wall of plants—the wood contains as much as 50%(w/w) cellulose and the cotton fiber can be as much as 90%(w/w) cellulose - Cellulose is also secreted by many bacteria during the formation of a biofilm—a mode of growth in which bacteria stick together and adhere to a surface in response to nutritional cues or to evade toxins - Being the most abundant organic biopolymer on Earth, cellulose is also a major component of dietary fiber (roughage)—it provides bulking to the food in the gastrointestinal (GI) tract and absorbs water
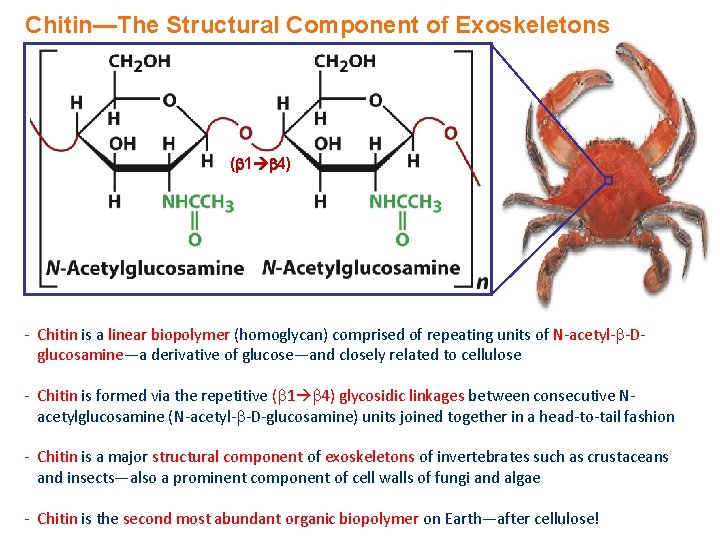
Chitin—The Structural Component of Exoskeletons ( 1 4) - Chitin is a linear biopolymer (homoglycan) comprised of repeating units of N-acetyl- -Dglucosamine—a derivative of glucose—and closely related to cellulose - Chitin is formed via the repetitive ( 1 4) glycosidic linkages between consecutive Nacetylglucosamine (N-acetyl- -D-glucosamine) units joined together in a head-to-tail fashion - Chitin is a major structural component of exoskeletons of invertebrates such as crustaceans and insects—also a prominent component of cell walls of fungi and algae - Chitin is the second most abundant organic biopolymer on Earth—after cellulose!
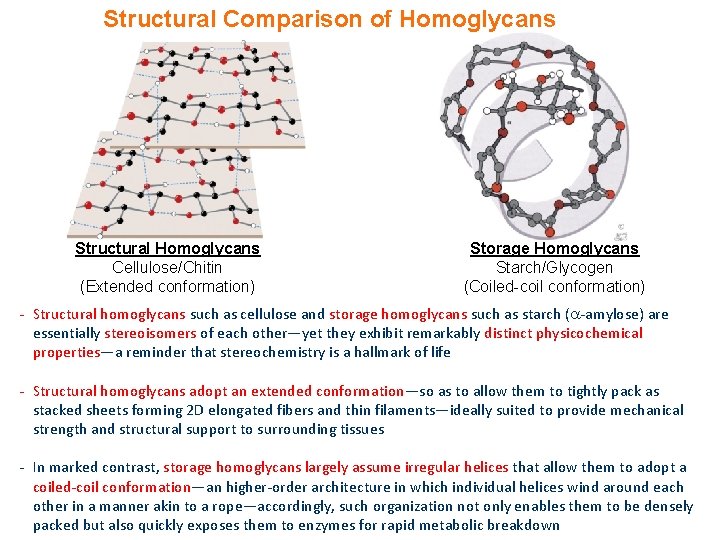
Structural Comparison of Homoglycans Structural Homoglycans Cellulose/Chitin (Extended conformation) Storage Homoglycans Starch/Glycogen (Coiled-coil conformation) - Structural homoglycans such as cellulose and storage homoglycans such as starch ( -amylose) are essentially stereoisomers of each other—yet they exhibit remarkably distinct physicochemical properties—a reminder that stereochemistry is a hallmark of life - Structural homoglycans adopt an extended conformation—so as to allow them to tightly pack as stacked sheets forming 2 D elongated fibers and thin filaments—ideally suited to provide mechanical strength and structural support to surrounding tissues - In marked contrast, storage homoglycans largely assume irregular helices that allow them to adopt a coiled-coil conformation—an higher-order architecture in which individual helices wind around each other in a manner akin to a rope—accordingly, such organization not only enables them to be densely packed but also quickly exposes them to enzymes for rapid metabolic breakdown
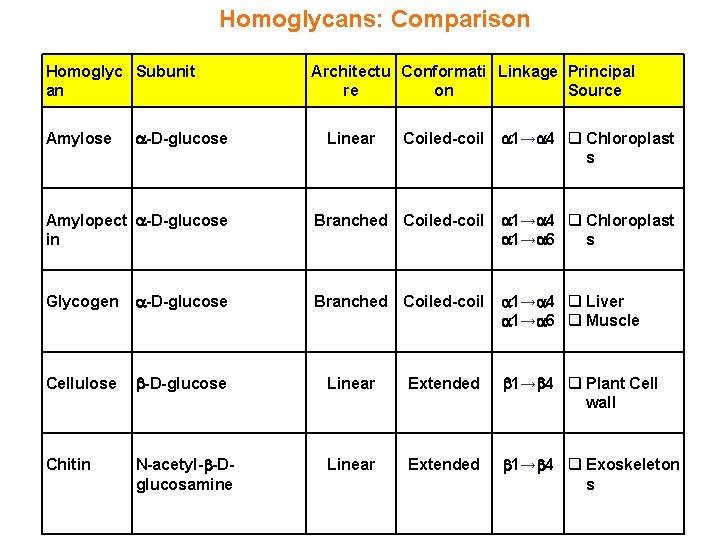
Homoglycans: Comparison Homoglyc Subunit an Coiled-coil 1→ 4 q Chloroplast s Amylopect -D-glucose in Branched Coiled-coil 1→ 4 q Chloroplast 1→ 6 s Glycogen -D-glucose Branched Coiled-coil 1→ 4 q Liver 1→ 6 q Muscle Cellulose -D-glucose Linear Extended 1→ 4 q Plant Cell wall Chitin N-acetyl- -Dglucosamine Linear Extended 1→ 4 q Exoskeleton s Amylose -D-glucose Architectu Conformati Linkage Principal re on Source Linear

Exercise 1. 2 b - Describe the monosaccharide unit components and their linkages in common polysaccharides - Compare and contrast the structures and functions of homoglycans such as cellulose, chitin, starch, and glycogen
§ 1. 2 c Heteroglycans

Synopsis 1. 2 c - Unlike homoglycans, heteroglycans harbor non-identical sugar units—they include complex polysaccharides such as glycosaminoglycans (GAGs) - GAGs are comprised of a disaccharide repeating unit in which one of the two monosaccharides is an amino sugar (usually either glucosamine or galactosamine) - Due to their polyanionic nature, GAGs are highly hydrated leading to their high viscosity and elasticity - GAGs are a major component of extracellular matrix (ECM)—a “filler” substance (cf bacterial biofilm) in connective tissues such as cartilage and synovial fluid at the joints between bones—wherein they help to reduce friction as well as serve as lubricants and shock absorbers due to their “spongy” nature - Covalent association of glycosaminoglycans to proteins generates the so-called “proteoglycans”—in addition to their role as structural proteins, glycoproteins also serve as enzymes, transporters, receptors, and hormones
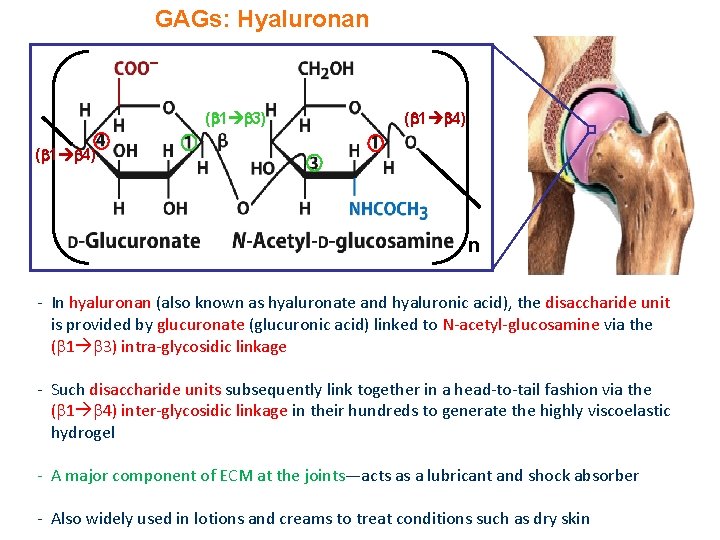
GAGs: Hyaluronan ( 1 3) ( 1 4) n - In hyaluronan (also known as hyaluronate and hyaluronic acid), the disaccharide unit is provided by glucuronate (glucuronic acid) linked to N-acetyl-glucosamine via the ( 1 3) intra-glycosidic linkage - Such disaccharide units subsequently link together in a head-to-tail fashion via the ( 1 4) inter-glycosidic linkage in their hundreds to generate the highly viscoelastic hydrogel - A major component of ECM at the joints—acts as a lubricant and shock absorber - Also widely used in lotions and creams to treat conditions such as dry skin
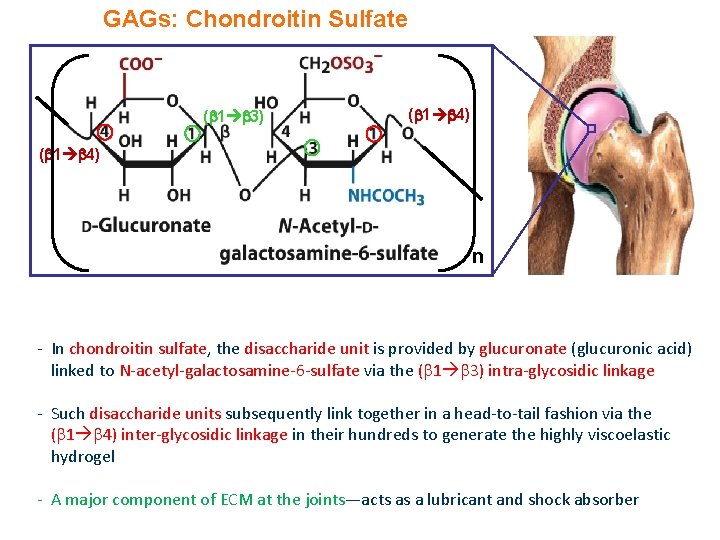
GAGs: Chondroitin Sulfate ( 1 3) ( 1 4) n - In chondroitin sulfate, the disaccharide unit is provided by glucuronate (glucuronic acid) linked to N-acetyl-galactosamine-6 -sulfate via the ( 1 3) intra-glycosidic linkage - Such disaccharide units subsequently link together in a head-to-tail fashion via the ( 1 4) inter-glycosidic linkage in their hundreds to generate the highly viscoelastic hydrogel - A major component of ECM at the joints—acts as a lubricant and shock absorber
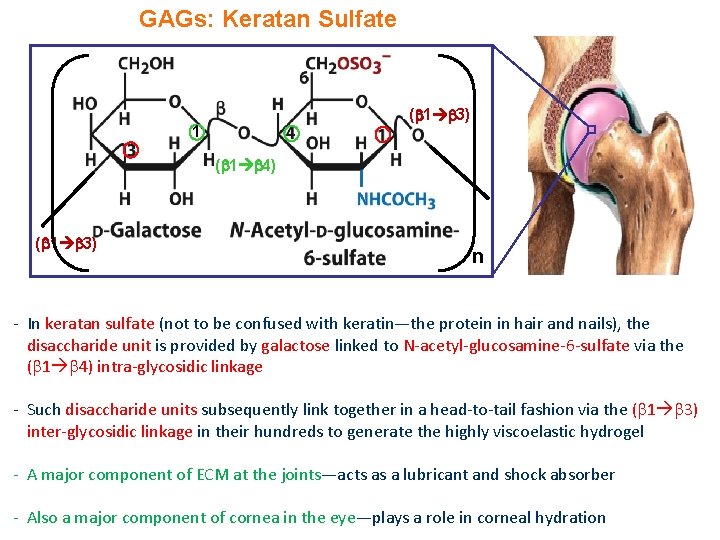
GAGs: Keratan Sulfate ( 1 3) 1 ( 1 4) ( 1 3) n - In keratan sulfate (not to be confused with keratin—the protein in hair and nails), the disaccharide unit is provided by galactose linked to N-acetyl-glucosamine-6 -sulfate via the ( 1 4) intra-glycosidic linkage - Such disaccharide units subsequently link together in a head-to-tail fashion via the ( 1 3) inter-glycosidic linkage in their hundreds to generate the highly viscoelastic hydrogel - A major component of ECM at the joints—acts as a lubricant and shock absorber - Also a major component of cornea in the eye—plays a role in corneal hydration
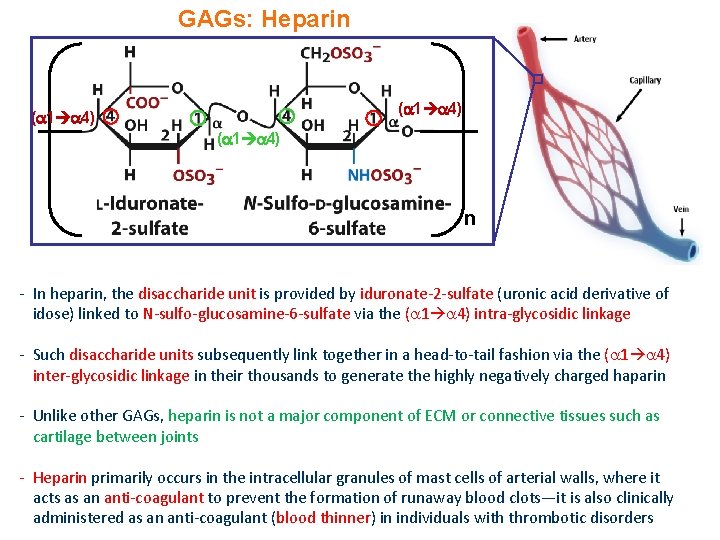
GAGs: Heparin ( 1 4) n - In heparin, the disaccharide unit is provided by iduronate-2 -sulfate (uronic acid derivative of idose) linked to N-sulfo-glucosamine-6 -sulfate via the ( 1 4) intra-glycosidic linkage - Such disaccharide units subsequently link together in a head-to-tail fashion via the ( 1 4) inter-glycosidic linkage in their thousands to generate the highly negatively charged haparin - Unlike other GAGs, heparin is not a major component of ECM or connective tissues such as cartilage between joints - Heparin primarily occurs in the intracellular granules of mast cells of arterial walls, where it acts as an anti-coagulant to prevent the formation of runaway blood clots—it is also clinically administered as an anti-coagulant (blood thinner) in individuals with thrombotic disorders
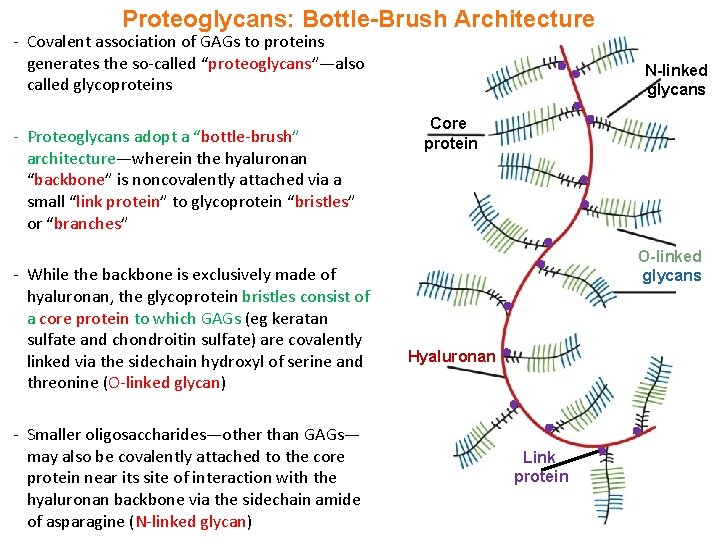
Proteoglycans: Bottle-Brush Architecture - Covalent association of GAGs to proteins generates the so-called “proteoglycans”—also called glycoproteins - Proteoglycans adopt a “bottle-brush” architecture—wherein the hyaluronan “backbone” is noncovalently attached via a small “link protein” to glycoprotein “bristles” or “branches” - While the backbone is exclusively made of hyaluronan, the glycoprotein bristles consist of a core protein to which GAGs (eg keratan sulfate and chondroitin sulfate) are covalently linked via the sidechain hydroxyl of serine and threonine (O-linked glycan) - Smaller oligosaccharides—other than GAGs— may also be covalently attached to the core protein near its site of interaction with the hyaluronan backbone via the sidechain amide of asparagine (N-linked glycan) N-linked glycans Core protein O-linked glycans Hyaluronan Link protein
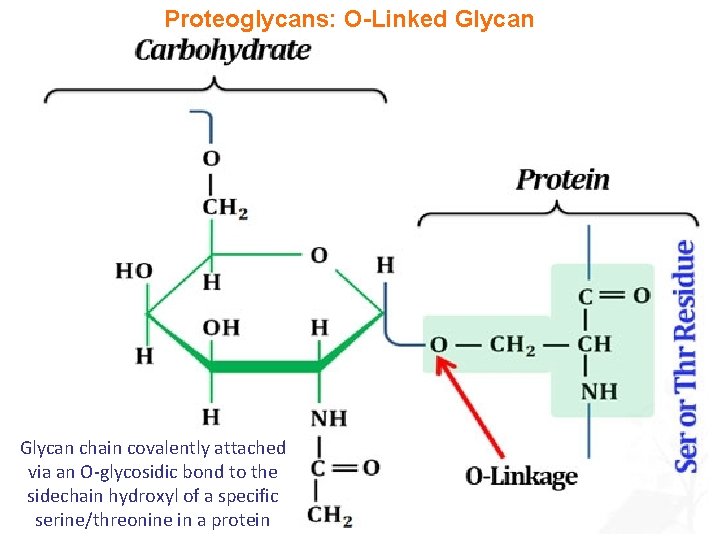
Proteoglycans: O-Linked Glycan chain covalently attached via an O-glycosidic bond to the sidechain hydroxyl of a specific serine/threonine in a protein
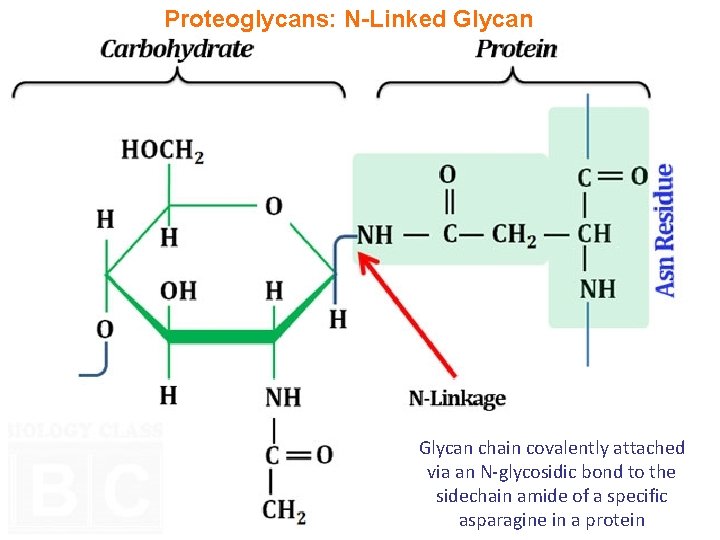
Proteoglycans: N-Linked Glycan chain covalently attached via an N-glycosidic bond to the sidechain amide of a specific asparagine in a protein
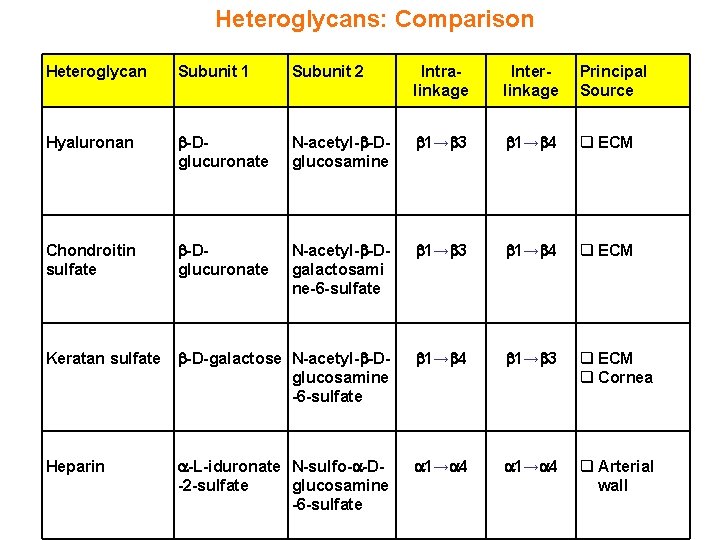
Heteroglycans: Comparison Heteroglycan Subunit 1 Subunit 2 Intralinkage Interlinkage Principal Source Hyaluronan -Dglucuronate N-acetyl- -Dglucosamine 1→ 3 1→ 4 q ECM Chondroitin sulfate -Dglucuronate N-acetyl- -Dgalactosami ne-6 -sulfate 1→ 3 1→ 4 q ECM Keratan sulfate -D-galactose N-acetyl- -Dglucosamine -6 -sulfate 1→ 4 1→ 3 q ECM q Cornea Heparin -L-iduronate N-sulfo- -D-2 -sulfate glucosamine -6 -sulfate 1→ 4 q Arterial wall

Exercise 1. 2 c - Compare and contrast the structures and functions of hyaluronan, chondroitin sulfate, keratan sulfate, and heparin - How do the physical properties of glycosaminoglycans relate to their biological roles?
- Slides: 30