I Introduction to Bonding Chemical Bonding C Johannesson

I. Introduction to Bonding Chemical Bonding C. Johannesson IV

A. Vocabulary • Chemical Bond • attractive force between atoms or ions that binds them together as a unit • bonds form in order to… • decrease potential energy (PE) • increase stability C. Johannesson
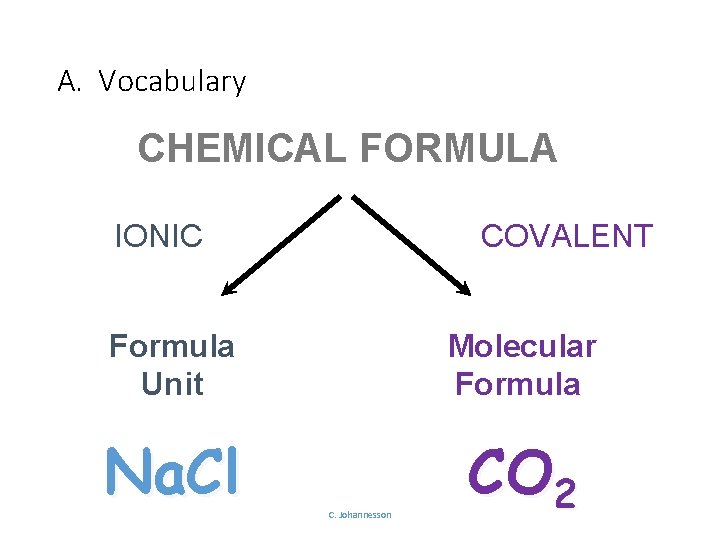
A. Vocabulary CHEMICAL FORMULA IONIC COVALENT Formula Unit Molecular Formula Na. Cl CO 2 C. Johannesson

A. Vocabulary COMPOUND more than 2 elements Binary Compound Na. Cl Ternary Compound C. Johannesson Na. NO 3
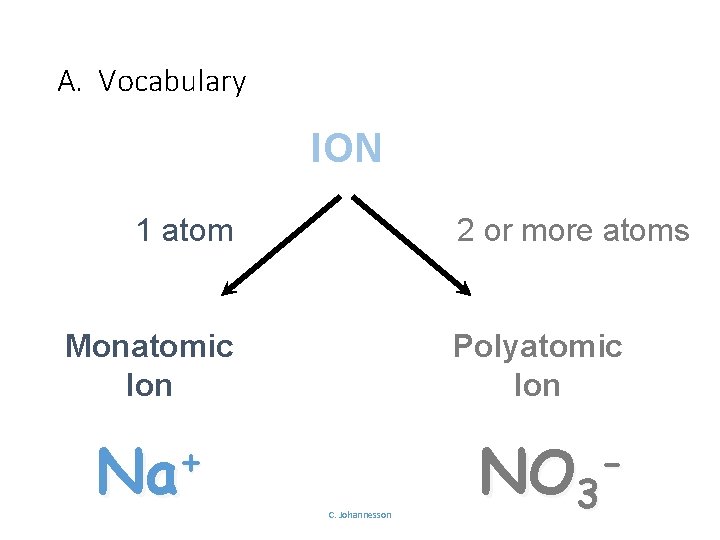
A. Vocabulary ION 2 or more atoms 1 atom Monatomic Ion + Na Polyatomic Ion C. Johannesson NO 3
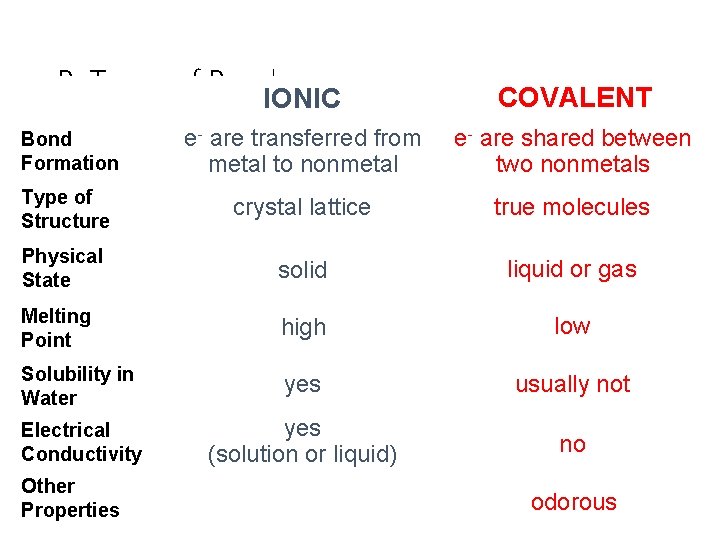
B. Types of Bonds IONIC COVALENT Bond Formation e- are transferred from metal to nonmetal e- are shared between two nonmetals Type of Structure crystal lattice true molecules Physical State solid liquid or gas Melting Point high low Solubility in Water yes usually not Electrical Conductivity yes (solution or liquid) no Other Properties C. Johannesson odorous

B. Types of Bonds METALLIC Bond Formation e- are delocalized among metal atoms Type of Structure “electron sea” Physical State solid Melting Point very high Solubility in Water no Electrical Conductivity yes (any form) Other Properties malleable, ductile, C. Johannesson lustrous
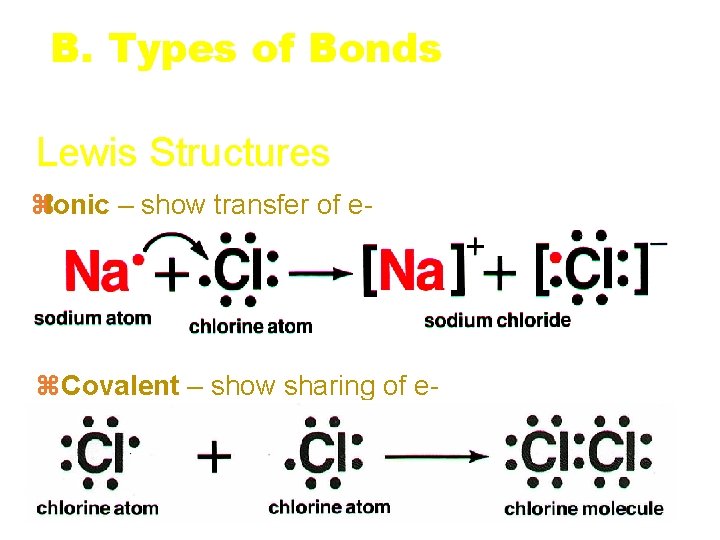
B. Types of Bonds Lewis Structures z. Ionic – show transfer of e- z. Covalent – show sharing of e- C. Johannesson RETURN
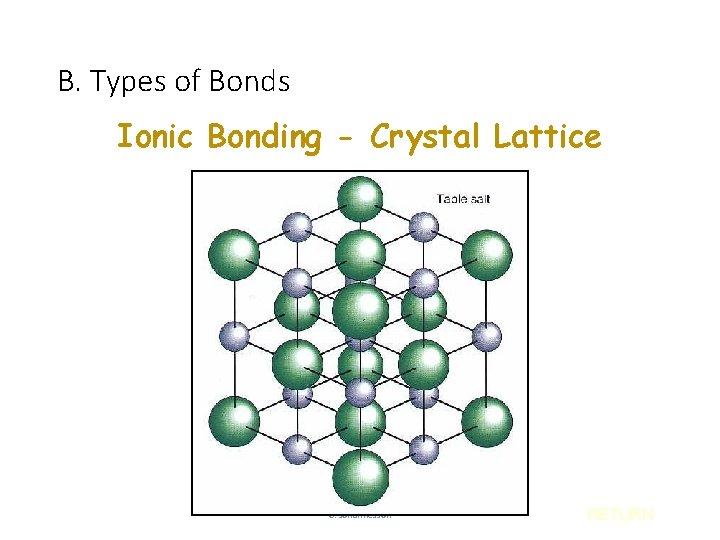
B. Types of Bonds Ionic Bonding - Crystal Lattice C. Johannesson RETURN
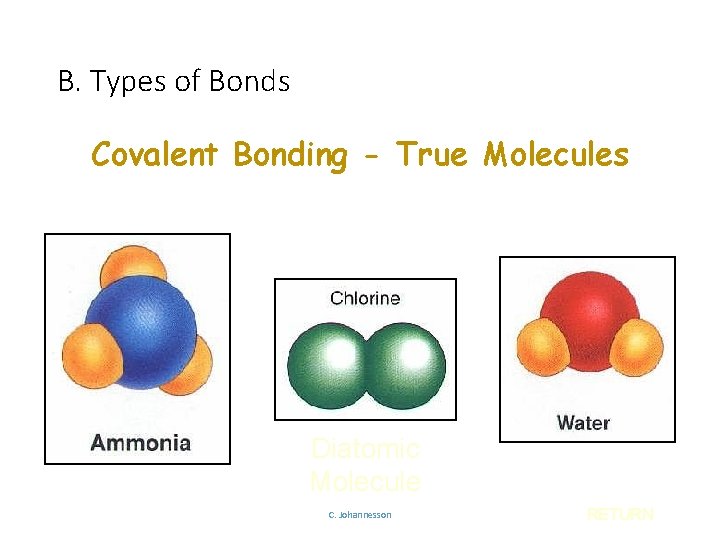
B. Types of Bonds Covalent Bonding - True Molecules Diatomic Molecule C. Johannesson RETURN
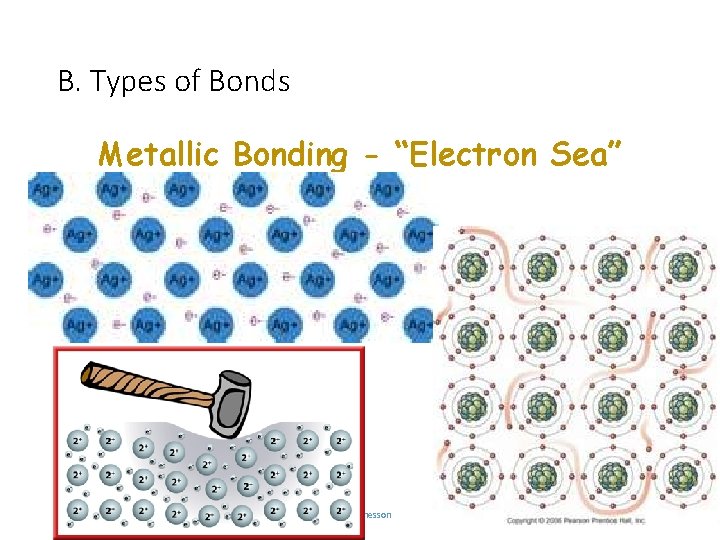
B. Types of Bonds Metallic Bonding - “Electron Sea” C. Johannesson RETURN

Metal Alloys • Metal mixture • Substitutional Alloys • Atoms of original metallic solid are replaced by other metal atoms of similar size • Sterling silver, brass, pewter • Interstitial Alloys • Smalller atoms fill in the spaces (interstices) between the larger metallic atoms • Carbon steel – holes between iron atoms are filled with carbon atoms making the iron harder, stronger and less ductile C. Johannesson
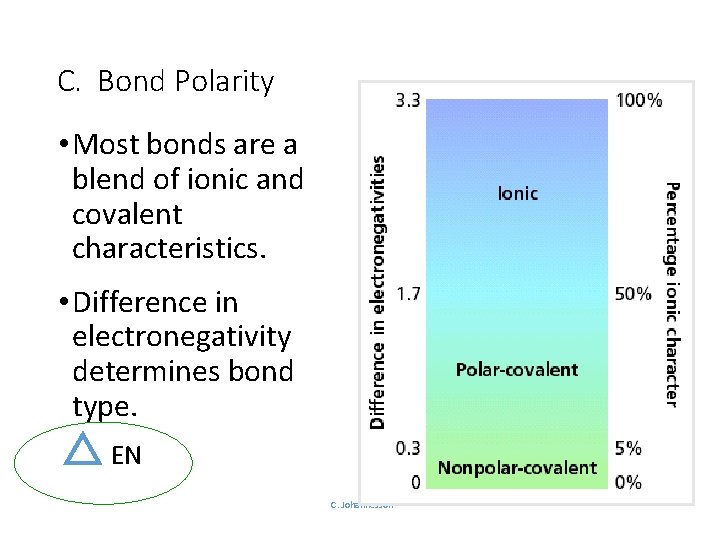
C. Bond Polarity • Most bonds are a blend of ionic and covalent characteristics. • Difference in electronegativity determines bond type. EN C. Johannesson
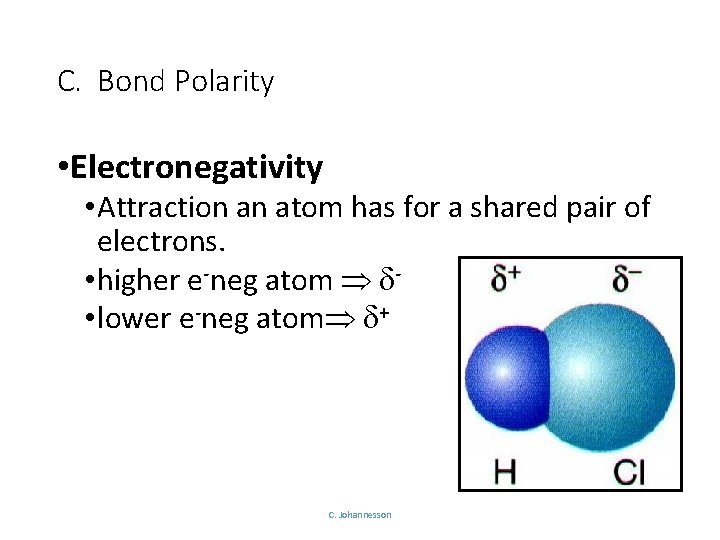
C. Bond Polarity • Electronegativity • Attraction an atom has for a shared pair of electrons. • higher e-neg atom • lower e-neg atom + C. Johannesson

C. Bond Polarity • Electronegativity Trend (p. 151) • Increases up and to the right. C. Johannesson

C. Bond Polarity • Nonpolar Covalent Bond • e- are shared equally • symmetrical e- density • usually identical atoms C. Johannesson
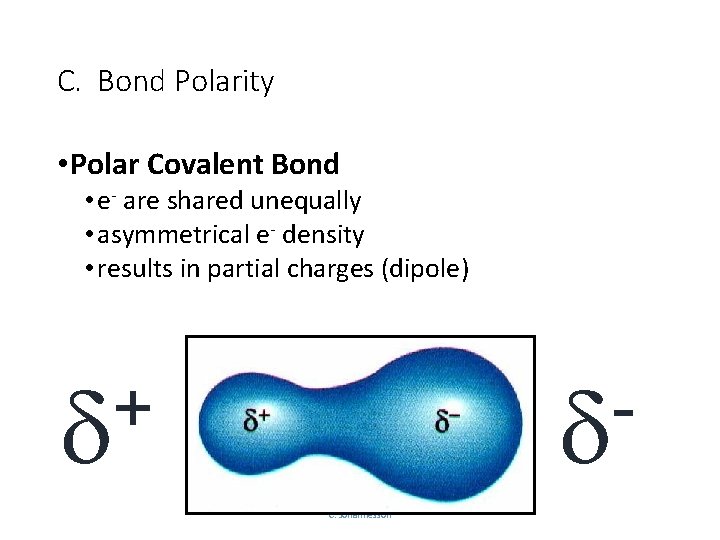
C. Bond Polarity • Polar Covalent Bond • e- are shared unequally • asymmetrical e- density • results in partial charges (dipole) + C. Johannesson
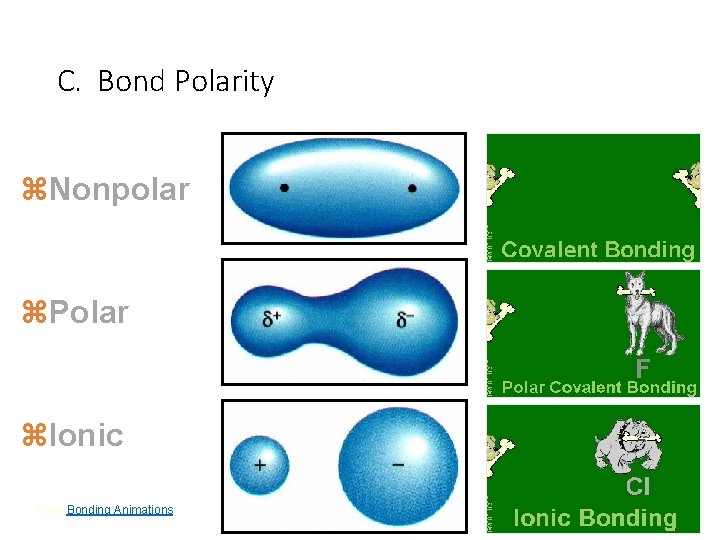
C. Bond Polarity z. Nonpolar z. Polar z. Ionic View Bonding Animations. C. Johannesson
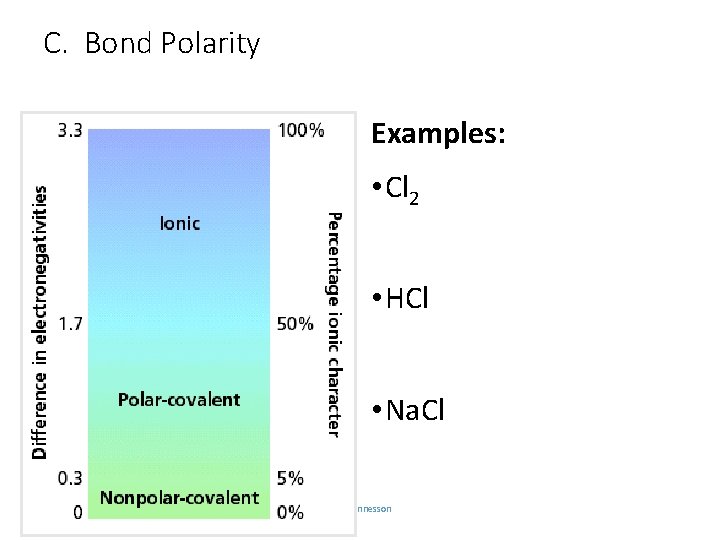
C. Bond Polarity Examples: • Cl 2 • HCl • Na. Cl C. Johannesson
- Slides: 19