I II IV Nuclear Chemistry Radioactive Decay Unstable

I II IV Nuclear Chemistry
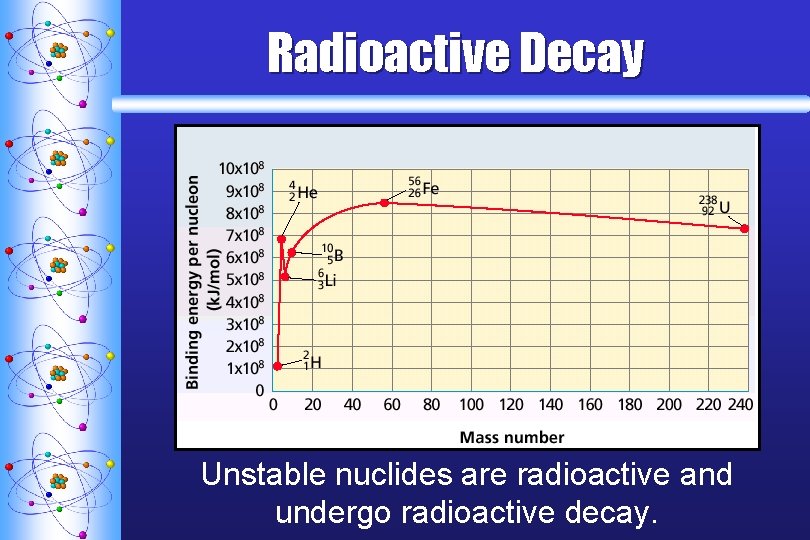
Radioactive Decay Unstable nuclides are radioactive and undergo radioactive decay.

Fission ª Splitting a nucleus into two or more smaller nuclei by bombarding it with a neutron ª 1 g of 235 U = 3 tons of coal C. Johannesson

Fission ª Bombarding a nuclide with a neutron ª chain reaction - self-propagating reaction ª Example: Nuclear ª bomb ª Creates a lot of nuclear waste C. Johannesson

Nuclear Power ª Fission Reactors https: //www. youtube. com/watch? v=D 91 T-B-PVE 0 C. Johannesson
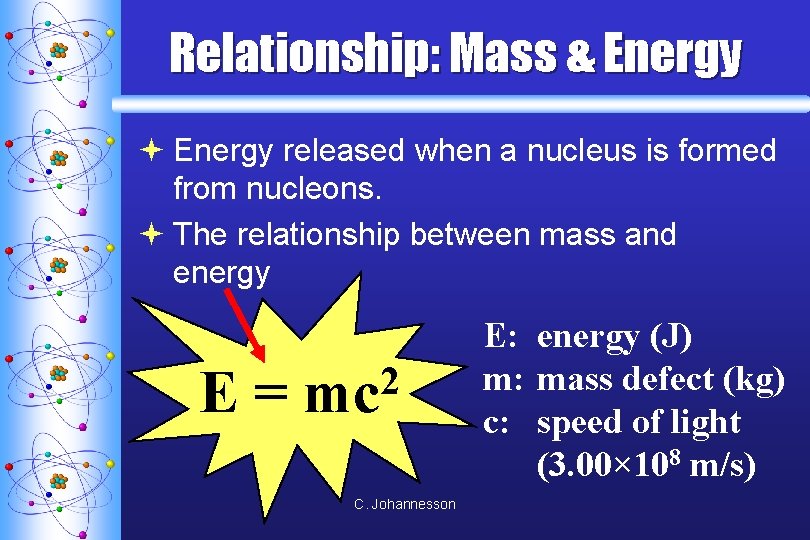
Relationship: Mass & Energy ª Energy released when a nucleus is formed from nucleons. ª The relationship between mass and energy E= 2 mc C. Johannesson E: energy (J) m: mass defect (kg) c: speed of light (3. 00× 108 m/s)
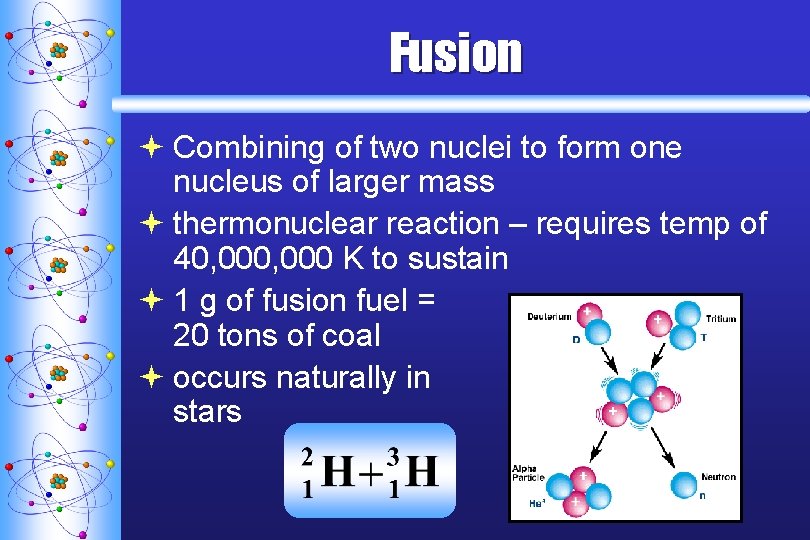
Fusion ª Combining of two nuclei to form one nucleus of larger mass ª thermonuclear reaction – requires temp of 40, 000 K to sustain ª 1 g of fusion fuel = 20 tons of coal ª occurs naturally in stars C. Johannesson

A. Nuclear Power ª Fusion Reactors (not yet sustainable) ª https: //www. youtube. com/watch? v=N 4 y. W National Spherical h. A 1 m. Vx. A Stop 2: 30 Torus Experiment Tokamak Fusion Test Reactor Princeton University

C. Fission vs. Fusion Both = change the identity and number of protons in the nucleus. F I S S I O ª 235 UN is limited ª danger of meltdown ª toxic waste ª thermal pollution F U S I O N ª fuel is abundant ª no danger of meltdown ª no toxic waste ª not yet sustainable

Synthetic Elements ª Transuranium Elements w elements with atomic #s above 92 w synthetically produced in nuclear reactors and accelerators by bombarding stable nuclei with particles. w most decay very rapidly C. Johannesson

Types of Radiation ª Alpha particle ( ) w helium nucleus ª Beta particle ( -) w electron 2+ paper 1 lead ª Positron ( +) w positron ª Gamma ( ) C. Johannesson w high-energy photon 1+ 0 concrete

Alpha ª Alpha Emission parent nuclide daughter nuclide alpha particle Numbers must balance!! C. Johannesson

Beta/Positron ª Beta Emission ª Positron Emission C. Johannesson electron positron

Electron Capture ª Electron Capture electron ª Gamma Emission w Usually follows other types of decay. ª Transmutation C. Johannesson w One element becomes another.
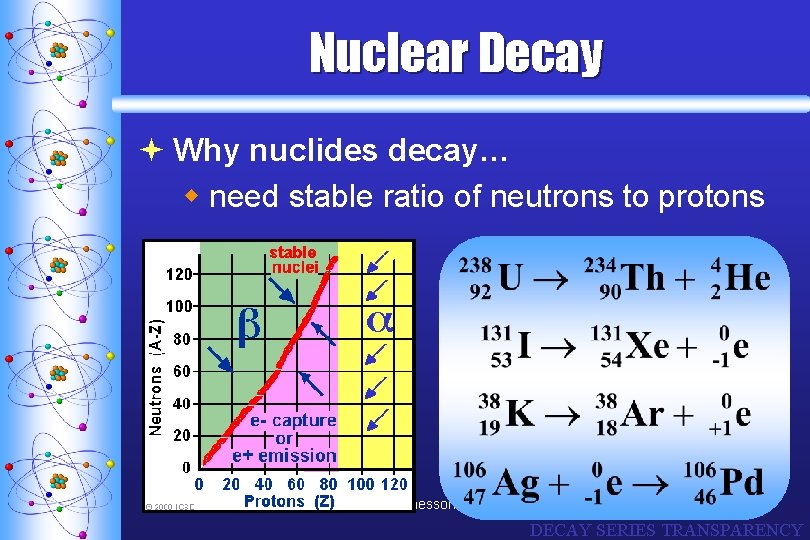
Nuclear Decay ª Why nuclides decay… w need stable ratio of neutrons to protons C. Johannesson DECAY SERIES TRANSPARENCY
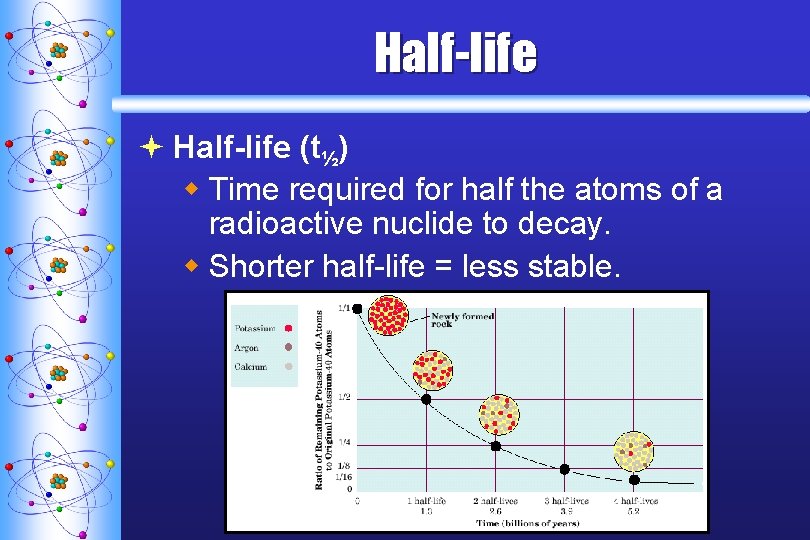
Half-life ª Half-life (t½) w Time required for half the atoms of a radioactive nuclide to decay. w Shorter half-life = less stable. C. Johannesson

Radioactive Dating ª half-life measurements of radioactive elements are used to determine the age of an object ª decay rate indicates amount of radioactive material ª EX: 14 C - up to 40, 000 years 238 U and 40 K - over 300, 000 years C. Johannesson
- Slides: 17