I Electron Configurations A Principal energy levels B

I. Electron Configurations A. Principal energy levels B. Energy sublevels C. Orbitals
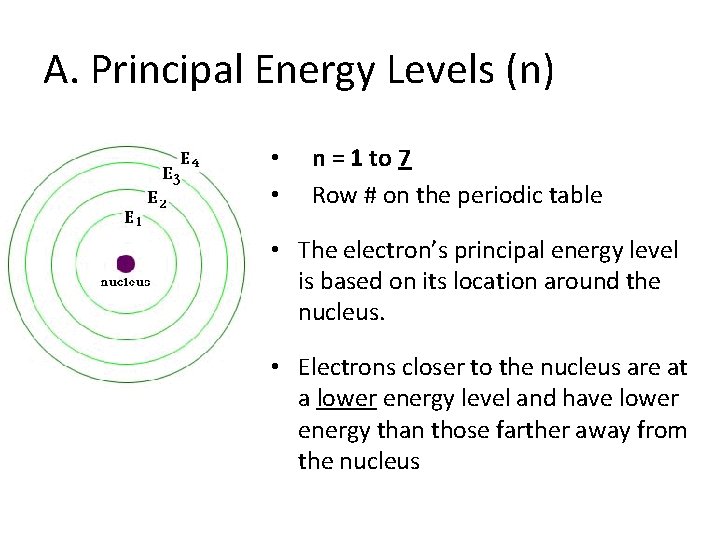
A. Principal Energy Levels (n) • • n = 1 to 7 Row # on the periodic table • The electron’s principal energy level is based on its location around the nucleus. • Electrons closer to the nucleus are at a lower energy level and have lower energy than those farther away from the nucleus

• Energy levels are like rungs of a ladder. You cannot be in between a rung • Energy levels in an atom’s electron are unequally spaced. The higher energy levels are closer together.

B. Energy Sublevels (s, p, d, f) • assigned letters s, p, d or f (smart people do fine) • Energy sublevels correspond to a shape where the electron is likely to be found • Sometimes also called “orbitals”

C. Orbitals • describes the electron’s location • orbitals are not necessarily spherical • maximum of 2 electrons per orbital – s sublevel has 1 orbital (2 electrons total) – p sublevel has 3 orbitals (6 electrons total) – d sublevel has 5 orbitals (10 electrons total ) – f sublevel has 7 orbitals (14 electrons total)
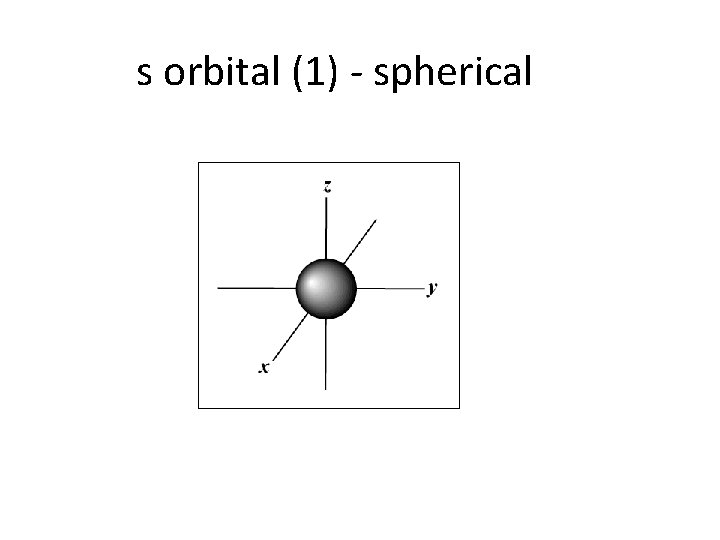
s orbital (1) - spherical
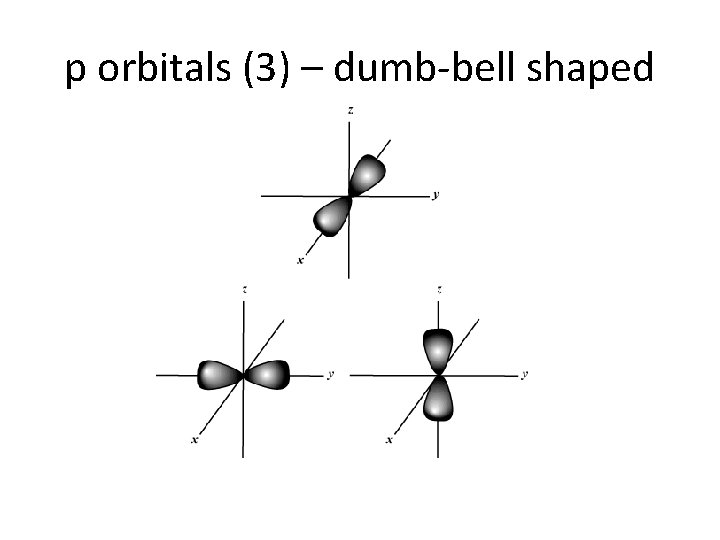
p orbitals (3) – dumb-bell shaped

d orbitals (5)

f-orbitals (7)


II. Electron Configuration Rules A. Electron configuration – the arrangement of electrons in an atom. • Example Sodium (Na) – 1 s 22 p 63 s 1 B. Three rules determine electron configurations 1. the Aufbau Principle, 2. the Pauli Exclusion Principle 3. Hund’s rule

1. The Aufbau Principle • Each electron occupies the lowest energy orbital available • Like filling the hotel from the bottom up
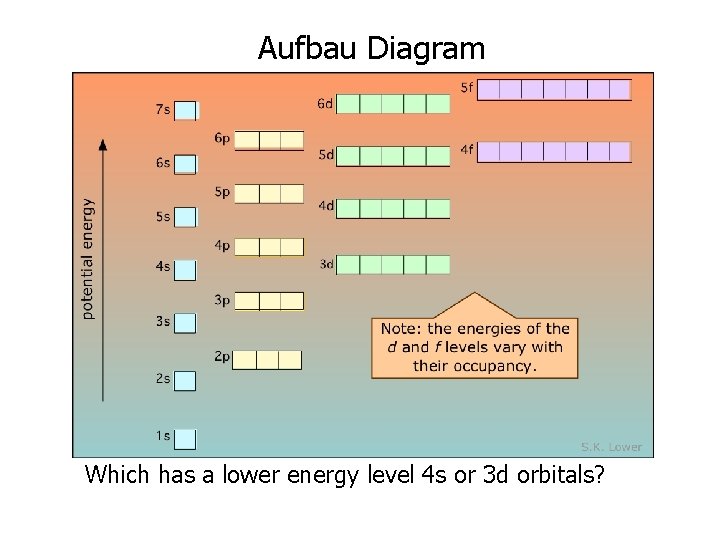
Aufbau Diagram Which has a lower energy level 4 s or 3 d orbitals?
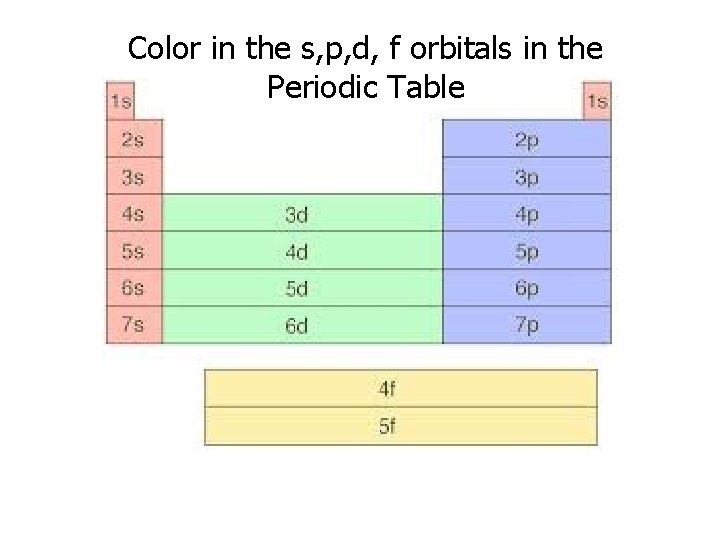
Color in the s, p, d, f orbitals in the Periodic Table

2. Pauli Exclusion Principle • A maximum of two electrons may occupy a single orbital • Like only two people sharing one bed

3. Hund’s Rule • If two or more orbitals of equal energy are available, electrons will occupy them singly with the same spin, before filling them in pairs with opposite spins • A spin is denoted with an up and down arrow to fill orbitals or ↿⇂ • This is like trying to find your own bed in the same suite before having to share a bed with someone else

III. Exceptions to the Rules • Copper (Cu) and chromium (Cr) are exceptions to the Aufbau principle • Some configurations violate the Aufbau Principle because half-filled sublevels are not as stable as filled sublevels

IV. Valence electrons A. Valence electrons are electrons in the outermost s and p orbitals • For group A elements, the group number corresponds to number of valence electrons. B. (Lewis) electron dot structures – element’s symbol surrounded by dots representing the valence electrons 8 A
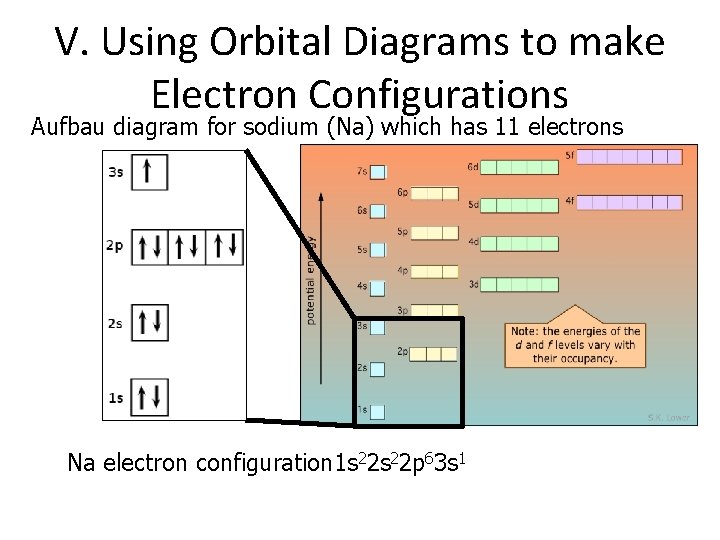
V. Using Orbital Diagrams to make Electron Configurations Aufbau diagram for sodium (Na) which has 11 electrons Na electron configuration 1 s 22 p 63 s 1

Orbital Diagrams Question Time • What two elements are exceptions to the Aufbau rule for electron configurations? • What are valence electrons?

- Slides: 21