I Bacterial Cell Division 5 1 Cell Growth

I. Bacterial Cell Division • • 5. 1 Cell Growth and Binary Fission 5. 2 Fts Proteins and Cell Division 5. 3 Mre. B and Determinants of Cell Morphology 5. 4 Peptidoglycan Synthesis and Cell Division © 2012 Pearson Education, Inc.

5. 1 Cell Growth and Binary Fission • Binary fission: cell division following enlargement of a cell to twice its minimum size (Figure 5. 1) • Generation time: time required for microbial cells to double in number • During cell division, each daughter cell receives a chromosome and sufficient copies of all other cell constituents to exist as an independent cell © 2012 Pearson Education, Inc.
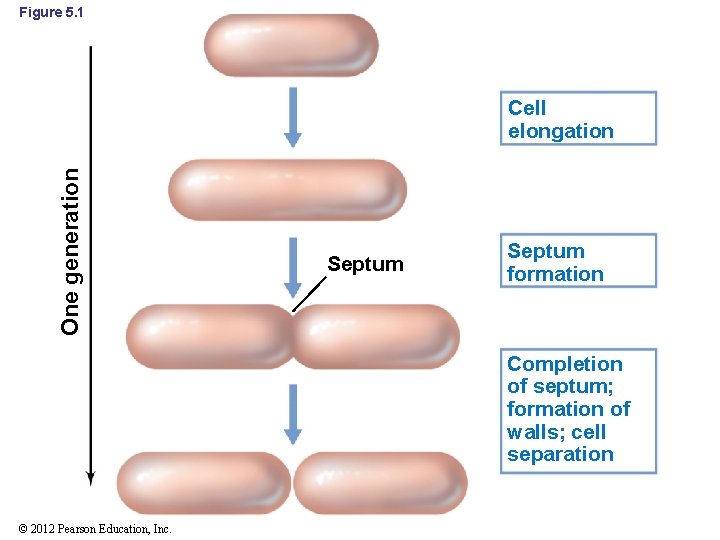
Figure 5. 1 One generation Cell elongation Septum formation Completion of septum; formation of walls; cell separation © 2012 Pearson Education, Inc.
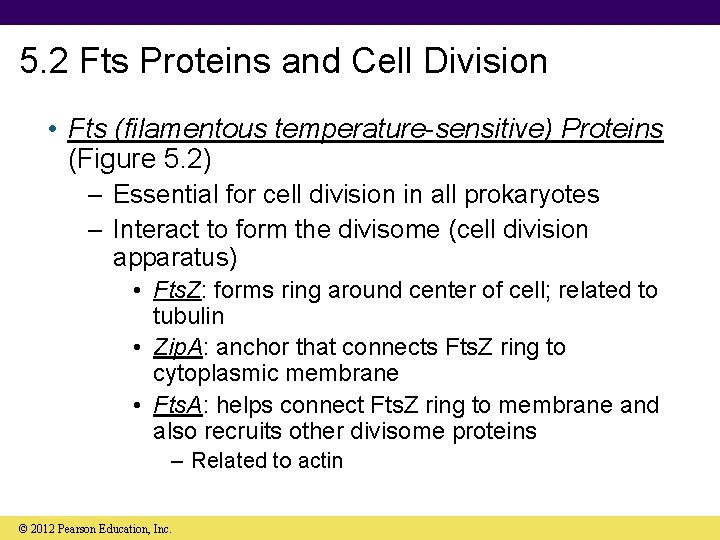
5. 2 Fts Proteins and Cell Division • Fts (filamentous temperature-sensitive) Proteins (Figure 5. 2) – Essential for cell division in all prokaryotes – Interact to form the divisome (cell division apparatus) • Fts. Z: forms ring around center of cell; related to tubulin • Zip. A: anchor that connects Fts. Z ring to cytoplasmic membrane • Fts. A: helps connect Fts. Z ring to membrane and also recruits other divisome proteins – Related to actin © 2012 Pearson Education, Inc.

5. 2 Fts Proteins and Cell Division • DNA replicates before the Fts. Z ring forms (Figure 5. 3) • Location of Fts. Z ring is facilitated by Min proteins – Min. C, Min. D, Min. E • Fts. K protein mediates separation of chromosomes to daughter cells © 2012 Pearson Education, Inc.
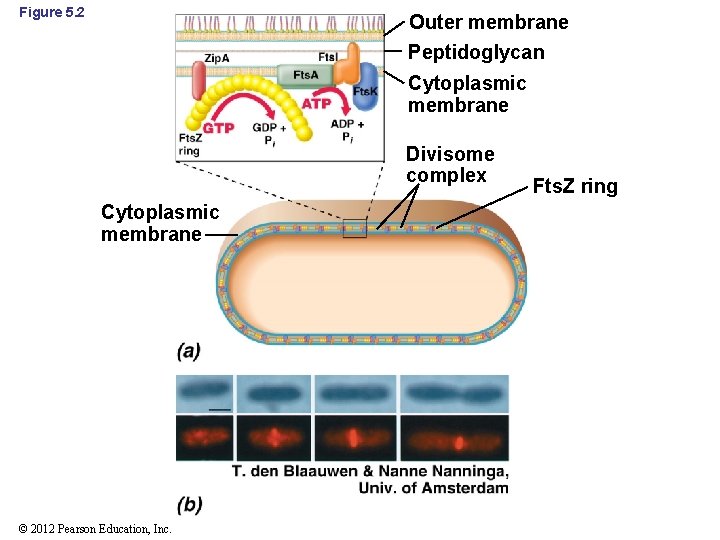
Figure 5. 2 Outer membrane Peptidoglycan Cytoplasmic membrane Divisome complex Cytoplasmic membrane © 2012 Pearson Education, Inc. Fts. Z ring

5. 3 Mre. B and Determinants of Cell Morphology • Prokaryotes contain a cell cytoskeleton that is dynamic and multifaceted • Mre. B: major shape-determining factor in prokaryotes – Forms simple cytoskeleton in Bacteria and probably Archaea – Forms spiral-shaped bands around the inside of the cell, underneath the cytoplasmic membrane (Figure 5. 4 a and b) – Not found in coccus-shaped bacteria © 2012 Pearson Education, Inc.

5. 3 Mre. B and Determinants of Cell Morphology • Mre. B (cont’d) – Localizes synthesis of new peptidoglycan and other cell wall components to specific locations along the cylinder of a rod-shaped cell during growth © 2012 Pearson Education, Inc.
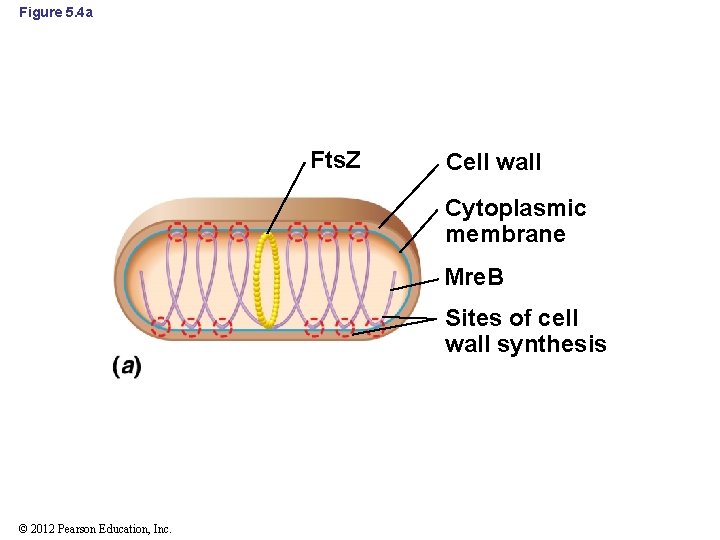
Figure 5. 4 a Fts. Z Cell wall Cytoplasmic membrane Mre. B Sites of cell wall synthesis © 2012 Pearson Education, Inc.
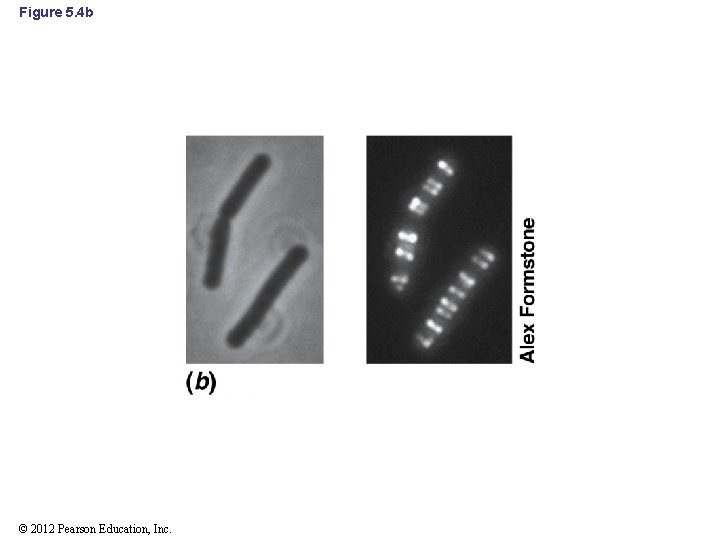
Figure 5. 4 b © 2012 Pearson Education, Inc.

5. 3 Mre. B and Determinants of Cell Morphology • Most archaeal genomes contain Fts. Z and Mre. B-like proteins, thus cell morphology is similar to that seen in Bacteria © 2012 Pearson Education, Inc.

5. 4 Peptidoglycan Synthesis and Cell Division • Production of new cell wall material is a major feature of cell division – In cocci, cell walls grow in opposite directions outward from the Fts. Z ring – In rod-shaped cells, growth occurs at several points along length of the cell © 2012 Pearson Education, Inc.

5. 4 Peptidoglycan Synthesis and Cell Division • Preexisting peptidoglycan needs to be severed to allow newly synthesized peptidoglycan to form – Beginning at the Fts. Z ring, small openings in the wall are created by autolysins – New cell wall material is added across the openings – Wall band: junction between new and old peptidoglycan © 2012 Pearson Education, Inc.
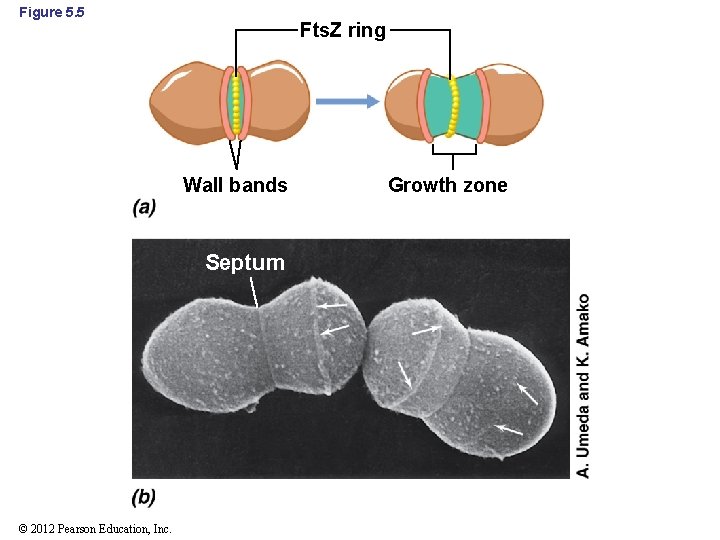
Figure 5. 5 Fts. Z ring Wall bands Septum © 2012 Pearson Education, Inc. Growth zone
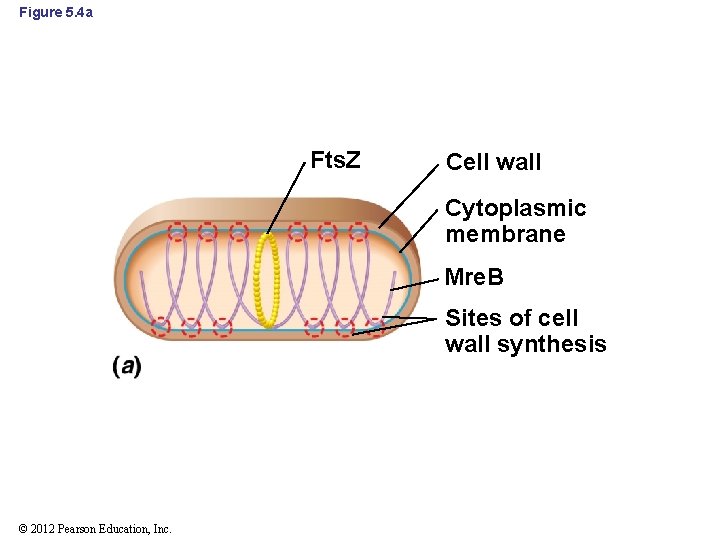
Figure 5. 4 a Fts. Z Cell wall Cytoplasmic membrane Mre. B Sites of cell wall synthesis © 2012 Pearson Education, Inc.

5. 4 Peptidoglycan Synthesis and Cell Division • Bactoprenol: carrier molecule that plays major role in insertion of peptidoglycan precursors – C 55 alcohol (Figure 5. 6) – Bonds to N-acetylglucosamine/ N-acetylmuramic acid/pentapeptide peptidoglycan precursor © 2012 Pearson Education, Inc.

5. 4 Peptidoglycan Synthesis and Cell Division • Glycolases: enzymes that interact with bactoprenol (Figure 5. 7 a) – Insert cell wall precursors into growing points of cell wall – Catalyze glycosidic bond formation © 2012 Pearson Education, Inc.
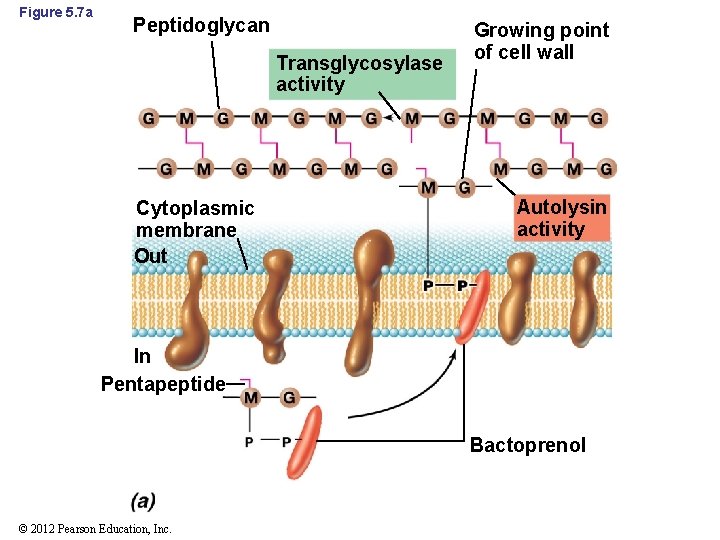
Figure 5. 7 a Peptidoglycan Transglycosylase activity Cytoplasmic membrane Out Growing point of cell wall Autolysin activity In Pentapeptide Bactoprenol © 2012 Pearson Education, Inc.

5. 4 Peptidoglycan Synthesis and Cell Division • Transpeptidation: final step in cell wall synthesis (Figure 5. 7 b) – Forms the peptide cross-links between muramic acid residues in adjacent glycan chains – Inhibited by the antibiotic penicillin © 2012 Pearson Education, Inc.
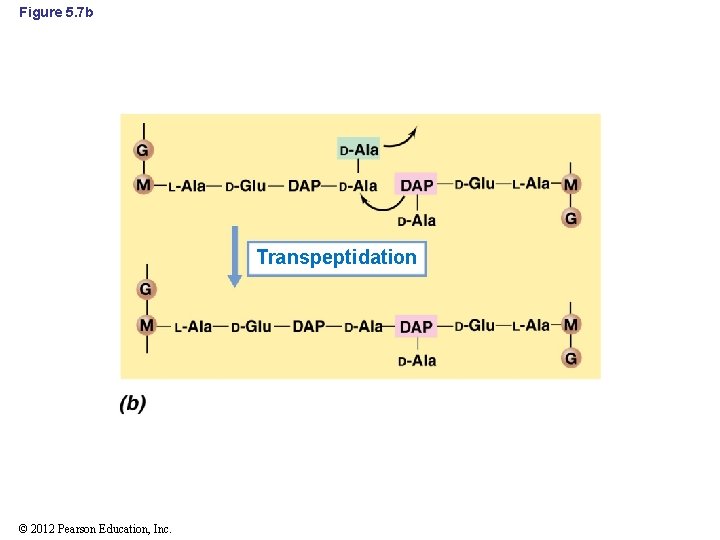
Figure 5. 7 b Transpeptidation © 2012 Pearson Education, Inc.

II. Population Growth • 5. 5 The Concept of Exponential Growth • 5. 6 The Mathematics of Exponential Growth • 5. 7 The Microbial Growth Cycle © 2012 Pearson Education, Inc.

5. 5 The Concept of Exponential Growth • Most bacteria have shorter generation times than eukaryotic microbes • Generation time is dependent on growth medium and incubation conditions © 2012 Pearson Education, Inc.

5. 5 The Concept of Exponential Growth • Exponential growth: growth of a microbial population in which cell numbers double within a specific time interval • During exponential growth, the increase in cell number is initially slow but increases at a faster rate (Figure 5. 8) © 2012 Pearson Education, Inc.

5. 7 The Microbial Growth Cycle • Batch culture: a closed-system microbial culture of fixed volume • Typical growth curve for population of cells grown in a closed system is characterized by four phases (Figure 5. 10): – – Lag phase Exponential phase Stationary phase Death phase Animation: Bacterial Growth Curve © 2012 Pearson Education, Inc.
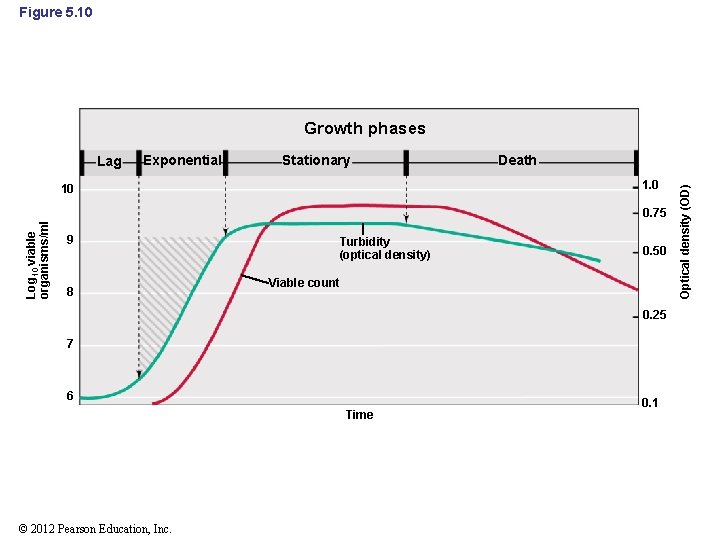
Figure 5. 10 Growth phases Exponential Stationary Death 1. 0 10 Log 10 viable organisms/ml 0. 75 9 8 Turbidity (optical density) 0. 50 Viable count 0. 25 7 6 Time © 2012 Pearson Education, Inc. 0. 1 Optical density (OD) Lag

5. 7 The Microbial Growth Cycle • Lag phase – Interval between when a culture is inoculated and when growth begins • Exponential phase – Cells in this phase are typically in the healthiest state • Stationary phase – Growth rate of population is zero – Either an essential nutrient is used up or waste product of the organism accumulates in the medium © 2012 Pearson Education, Inc.

5. 7 The Microbial Growth Cycle • Death Phase – If incubation continues after cells reach stationary phase, the cells will eventually die © 2012 Pearson Education, Inc.

IV. Temperature and Microbial Growth • 5. 12 Effect of Temperature on Growth • 5. 13 Microbial Life in the Cold • 5. 14 Microbial Life at High Temperatures © 2012 Pearson Education, Inc.
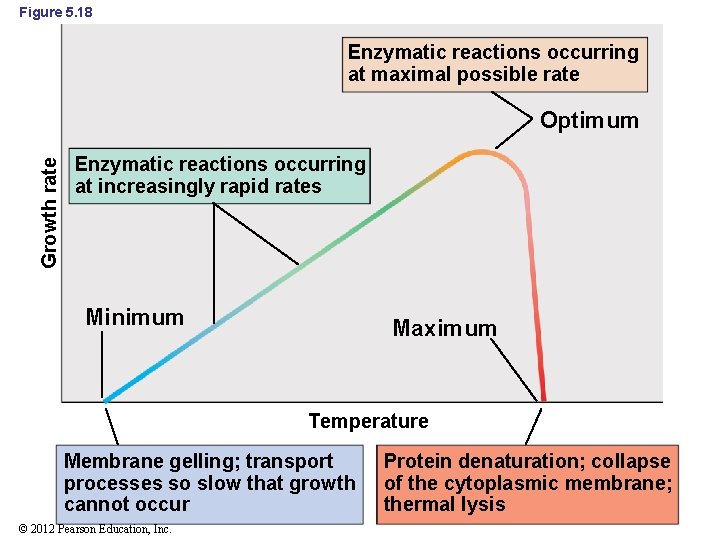
Figure 5. 18 Enzymatic reactions occurring at maximal possible rate Growth rate Optimum Enzymatic reactions occurring at increasingly rapid rates Minimum Maximum Temperature Membrane gelling; transport processes so slow that growth cannot occur © 2012 Pearson Education, Inc. Protein denaturation; collapse of the cytoplasmic membrane; thermal lysis

5. 12 Effect of Temperature on Growth • Microorganisms can be classified into groups by their growth temperature optima (Figure 5. 19) – – Psychrophile: low temperature Mesophile: midrange temperature Thermophile: high temperature Hyperthermophile: very high temperature © 2012 Pearson Education, Inc.
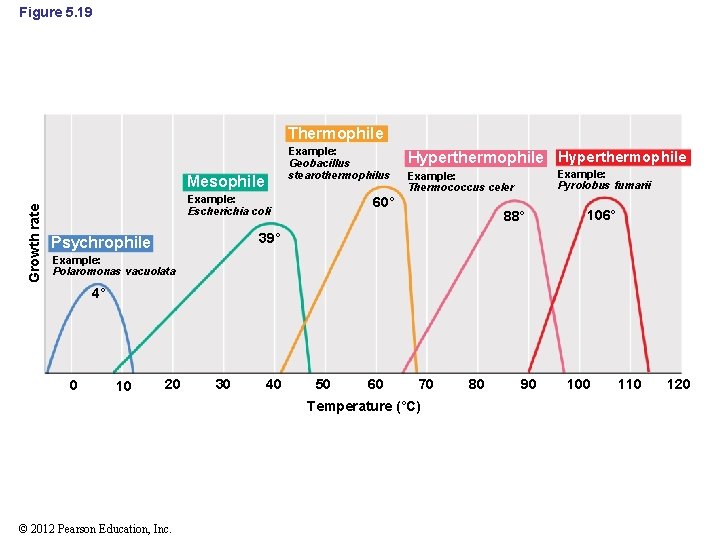
Figure 5. 19 Thermophile Example: Geobacillus stearothermophilus Growth rate Mesophile Example: Escherichia coli Hyperthermophile Example: Pyrolobus fumarii Example: Thermococcus celer 60° 88° 106° 39° Psychrophile Example: Polaromonas vacuolata 4° 0 10 20 30 40 50 60 70 Temperature (°C) © 2012 Pearson Education, Inc. 80 90 100 110 120

5. 12 Effect of Temperature on Growth • Mesophiles: organisms that have midrange temperature optima; found in – Warm-blooded animals – Terrestrial and aquatic environments – Temperate and tropical latitudes © 2012 Pearson Education, Inc.

5. 13 Microbial Life in the Cold • Extremophiles – Organisms that grow under very hot or very cold conditions • Psychrophiles – Organisms with cold temperature optima – Inhabit permanently cold environments (Figure 5. 20) • Psychrotolerant – Organisms that can grow at 0ºC but have optima of 20ºC to 40ºC – More widely distributed in nature than psychrophiles © 2012 Pearson Education, Inc.

5. 13 Microbial Life in the Cold • Molecular Adaptations to Psychrophily – Production of enzymes that function optimally in the cold; features that may provide more flexibility • • More -helices than -sheets More polar and less hydrophobic amino acids Fewer weak bonds Decreased interactions between protein domains © 2012 Pearson Education, Inc.

5. 13 Microbial Life in the Cold • Molecular Adaptations to Psychrophily (cont’d) – Transport processes function optimally at low temperatures • Modified cytoplasmic membranes – High unsaturated fatty acid content © 2012 Pearson Education, Inc.

Figure 5. 22 © 2012 Pearson Education, Inc.

Figure 5. 23 © 2012 Pearson Education, Inc.

5. 14 Microbial Life at High Temperatures • Studies of thermal habitats have revealed – Prokaryotes are able to grow at higher temperatures than eukaryotes – Organisms with the highest temperature optima are Archaea – Nonphototrophic organisms can grow at higher temperatures than phototrophic organisms © 2012 Pearson Education, Inc.

5. 14 Microbial Life at High Temperatures • Molecular Adaptations to Thermophily – Enzyme and proteins function optimally at high temperatures; features that provide thermal stability • Critical amino acid substitutions in a few locations provide more heat-tolerant folds • An increased number of ionic bonds between basic and acidic amino acids resist unfolding in the aqueous cytoplasm • Production of solutes (e. g. , di-inositol phophate, diglycerol phosphate) help stabilize proteins © 2012 Pearson Education, Inc.

5. 14 Microbial Life at High Temperatures • Molecular Adaptations to Thermophily (cont’d) – Modifications in cytoplasmic membranes to ensure heat stability • Bacteria have lipids rich in saturated fatty acids • Archaea have lipid monolayer rather than bilayer © 2012 Pearson Education, Inc.

5. 14 Microbial Life at High Temperatures • Hyperthermophiles produce enzymes widely used in industrial microbiology – Example: Taq polymerase, used to automate the repetitive steps in the polymerase chain reaction (PCR) technique © 2012 Pearson Education, Inc.

V. Other Environmental Factors Affecting Growth • • 5. 15 Acidity and Alkalinity 5. 16 Osmotic Effects on Microbial Growth 5. 17 Oxygen and Microorganisms 5. 18 Toxic Forms of Oxygen © 2012 Pearson Education, Inc.

5. 15 Acidity and Alkalinity • The p. H of an environment greatly affects microbial growth (Figure 5. 24) • Some organisms have evolved to grow best at low or high p. H, but most organisms grow best between p. H 6 and 8 (neutrophiles) © 2012 Pearson Education, Inc.
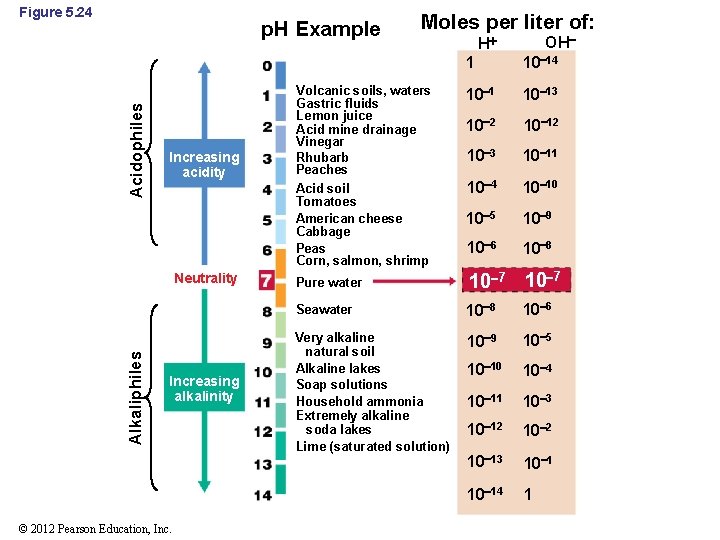
Figure 5. 24 Acidophiles p. H Example Increasing acidity Alkaliphiles Neutrality Increasing alkalinity © 2012 Pearson Education, Inc. Moles per liter of: Volcanic soils, waters Gastric fluids Lemon juice Acid mine drainage Vinegar Rhubarb Peaches Acid soil Tomatoes American cheese Cabbage Peas Corn, salmon, shrimp OH H 1 10 14 10 13 10 2 10 12 10 3 10 11 10 4 10 10 10 5 10 9 10 6 10 8 Pure water 10 7 Seawater 10 8 10 6 Very alkaline natural soil Alkaline lakes Soap solutions Household ammonia Extremely alkaline soda lakes Lime (saturated solution) 10 9 10 5 10 10 10 4 10 11 10 3 10 12 10 13 10 14 1

5. 15 Acidity and Alkalinity • Acidophiles: organisms that grow best at low p. H (<6) – Some are obligate acidophiles; membranes destroyed at neutral p. H – Stability of cytoplasmic membrane critical • Alkaliphiles: organisms that grow best at high p. H (>9) – Some have sodium motive force rather than proton motive force © 2012 Pearson Education, Inc.

5. 15 Acidity and Alkalinity • The internal p. H of a cell must stay relatively close to neutral even though the external p. H is highly acidic or basic – Internal p. H has been found to be as low as 4. 6 and as high as 9. 5 in extreme acido- and alkaliphiles, respectively © 2012 Pearson Education, Inc.

5. 15 Acidity and Alkalinity • Microbial culture media typically contain buffers to maintain constant p. H © 2012 Pearson Education, Inc.

5. 16 Osmotic Effects on Microbial Growth • Typically, the cytoplasm has a higher solute concentration than the surrounding environment, thus the tendency is for water to move into the cell (positive water balance) • When a cell is in an environment with a higher external solute concentration, water will flow out unless the cell has a mechanism to prevent this © 2012 Pearson Education, Inc.

5. 16 Osmotic Effects on Microbial Growth • Halophiles: organisms that grow best at reduced water potential; have a specific requirement for Na. Cl (Figure 5. 25) • Extreme halophiles: organisms that require high levels (15– 30%) of Na. Cl for growth • Halotolerant: organisms that can tolerate some reduction in water activity of environment but generally grow best in the absence of the added solute © 2012 Pearson Education, Inc.
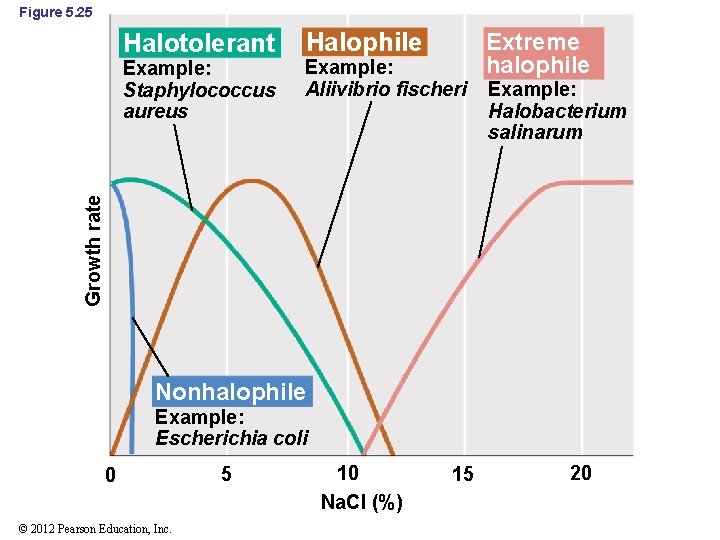
Figure 5. 25 Halotolerant Example: Aliivibrio fischeri Extreme halophile Example: Halobacterium salinarum Growth rate Example: Staphylococcus aureus Halophile Nonhalophile Example: Escherichia coli 0 © 2012 Pearson Education, Inc. 5 10 Na. Cl (%) 15 20

5. 16 Osmotic Effects on Microbial Growth • Osmophiles: organisms that live in environments high in sugar as solute • Xerophiles: organisms able to grow in very dry environments © 2012 Pearson Education, Inc.

5. 16 Osmotic Effects on Microbial Growth • Mechanisms for combating low water activity in surrounding environment involve increasing the internal solute concentration by – Pumping inorganic ions from environment into cell – Synthesis or concentration of organic solutes • compatible solutes: compounds used by cell to counteract low water activity in surrounding environment © 2012 Pearson Education, Inc.

5. 17 Oxygen and Microorganisms • Aerobes: require oxygen to live • Anaerobes: do not require oxygen and may even be killed by exposure • Facultative organisms: can live with or without oxygen • Aerotolerant anaerobes: can tolerate oxygen and grow in its presence even though they cannot use it • Microaerophiles: can use oxygen only when it is present at levels reduced from that in air © 2012 Pearson Education, Inc.

5. 17 Oxygen and Microorganisms • Thioglycolate broth (Figure 5. 26) – Complex medium that separates microbes based on oxygen requirements – Reacts with oxygen so oxygen can only penetrate the top of the tube © 2012 Pearson Education, Inc.
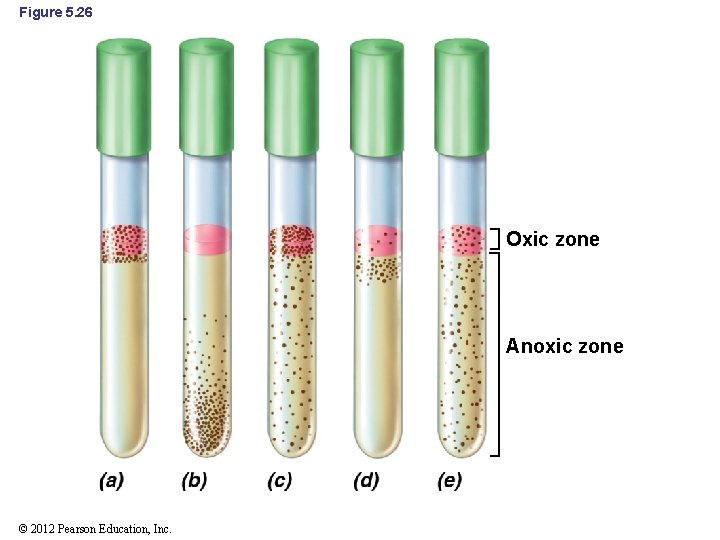
Figure 5. 26 Oxic zone Anoxic zone © 2012 Pearson Education, Inc.

5. 17 Oxygen and Microorganisms • Special techniques are needed to grow aerobic and anaerobic microorganisms (Figure 5. 27) • Reducing agents: chemicals that may be added to culture media to reduce oxygen (e. g. , thioglycolate) © 2012 Pearson Education, Inc.

Figure 5. 27 © 2012 Pearson Education, Inc.
- Slides: 57