Hypoglycaemia in Neonates Causes and Management By Nicole

Hypoglycaemia in Neonates: Causes and Management By: Nicole Stevens (RN, RM, NICU cert. )

DIABETES IN PREGNANCY • Makes a pregnancy high risk • Can have negative effects on mother & baby • Glucose is the food source which passes from the mother, across the placenta to the fetus • Complications for the fetus/neonate are most significant when the glucose control is poor • Careful glucose control decreases risk to the mother & developing fetus and improves outcomes for the neonate

Diabetes in pregnancy When a mothers blood glucose levels are too high the fetus also receives excessive amounts of glucose The fetus has the ability to increase their production of insulin substantially in order to use the large amounts of glucose This abnormal cycle of events can result in several complications The complications can be dependent on the period of time that the glucose was not well controlled.

Diabetes in pregnancy • Glucose crosses the placenta by diffusion • Glucose concentration 70 -80% maternal • Insulin does not cross the placenta • Insulin secretion is influenced by glucose and amino acid concentration, but as the fetus matures they become more glucose sensitive • ↑Glucose sensitivity ↑ β-cell mass o ↑ insulin production o Anabolism o Rapid fetal growth and laying down of fat

Diabetes in pregnancy • If a woman has pre exisiting diabetes there can be high levels of glucose at the beginning of the pregnancy (during the crucial period of organ development), as well as at any other stage through the pregnancy • In gestational diabetes the elevation may only happen later in the pregnancy • The implications for the fetus/neonate can be dependent on when there was exposure to high levels of glucose

Diabetes in pregnancy • Increase in glucose causes increases in insulin production • This causes an increase in catabolism (breakdown of complex molecules into simpler ones) which requires energy, which in turn requires oxygen • Depleted oxygen stores leads to fetal hypoxia which causes the release of a flood of adrenal catecholamines (hormones produced in the adrenal glands, eg. Adrenaline, dopamine) • This in turn causes hypertension, cardiac hypertropy, stimulation of erythropoietin (a hormone that promotes the formation of RBC’s), RBC hyperplasia and increased hematocrit levels

EMBRYOLOGY An understanding of embryology helps to explain why poor glucose control early in pregnancy can have devastating implications Organogenesis occurs between weeks 3 – 8 of pregnancy It is the series of organised integrated processes that transforms an amorphous (undifferentiated) mass of cells into a complete organ; it continues until the definitive characteristics of the organ are achieved Concurrent with this process is histogenesis: which is the formation of different tissues from undifferentiated cells

Embryology These undifferentiated cells are constituents of 3 primary germ layers (endoderm, mesoderm and ectoderm) Different parts of the body form from different layers, eg: endoderm: GI tract, respiratory tract, endocrine glands and organs; mesoderm: bones, muscles, urinary system; ectoderm: CNS, eyes, skin The end result of organogenesis and histogenesis is a structurally and functionally complete organ The accomplishment of organogenesis ends the period where the developing organism is known as an embryo and begins the period during which it is known as a fetus

Embryology The hormonal and metabolic changes that can occur with diabetes can adversely affect the developing embryo/fetus This abnormal metabolic environment is considered to be teratogenic There is a higher incidence of congenital malformations, such as cardiac, musculoskeletal, and CNS anomalies in neonates of mothers with diabetes The mechanism by which hyperglycaemia disturbs embryonic development is multifactorial and remains controversial

Embryology There are theories that altered levels of arachidonic acid and myoinositol levels or fetal hyperglycaemia promote excessive formation of oxygen radicals in cells, which damage mitochondria, inhibits prostacyclin, and leads to oxidative stress This results in an overabundance of thromboxanes and other prostaglandins which disrupts vascularisation of developing tissues Hyperglycaemia alters the expression of regulating genes, resulting in altered cellular mitosis and normal apoptosis (programmed cell death); exaggerated apoptosis results in fetal anomalies

NEONATAL COMPLICATIONS (from maternal diabetes) Macrosomia Increase in adipose tissue Increase in skin fold thickness Visceromegaly (liver) Phlethoric appearance due to polycythaemia RDS Hypoglycaemia (most common problem) Hypocalcaemia Hyperbilirubinaemia Hyperviscosity syndrome Hypertrophy cardiomyopathy Later effects: Obesity in childhood and development of type 2 diabetes.

CARDIAC ABNORMALITIES Congenital anomalies occur more commonly in infants born to mothers with diabetes Cardiac defects are dominant The incidence of abnormalities is highest in the group where mothers were on insulin at the time of conception The most frequent cardiac anomalies are ventricular septal defect, transposition of the greater arteries and aortic stenosis Defects involving the great arteries, including truncus arteriosus and double outlet right ventricle, are also more prevalent.

Hypertrophic cardiomyopathy • The incidence of hypertrophic cardiomyopathy is increased in infants of mothers with diabetes; fetal hyperinsulinaemia, triggered by maternal hyperglycaemia during the 3 rd trimester can be the causative factor • The left ventricular mass will usually be significantly bigger and there will be hypertrophy, or thickening, of the septal wall. • The problem is usually transitory and generally resolves after a few weeks.
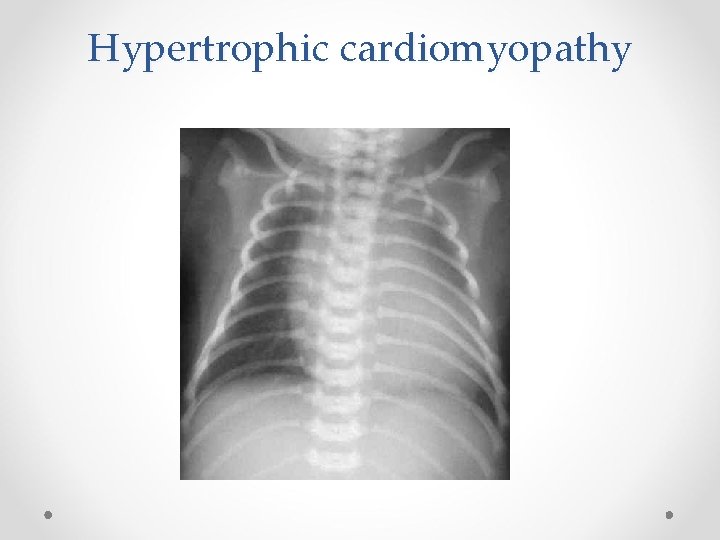
Hypertrophic cardiomyopathy

RESPIRATORY COMPLICATIONS • RDS requiring admission to SCN/NICU is 6 times more likely in the infant of a mother with diabetes • Delayed maturation of surfactant synthesis has been observed in these infants, but it is unclear if the cause is hyperglycaemia or hyperinsulinaemia inutero • Glycogen is normally depleted from the lungs as part of the maturation process and this coincides with an increase in surfactant synthesis • Insulin inhibits glycogen breakdown

HEMATOLOGY COMPLICATIONS Hyperglycaemia decreases fetal oxygen tension, this stimulates fetal erythropoietin production, which increases RBC’s Fetal hyperglycaemia & hyperinsulinaemia increase total body oxygen consumption by up to 30%, leading to accelerated erythropoiesis, polycythaemia and hyperviscosity The increase in fetal RBC’s leads to greater iron demands and depletion of iron stores in the liver, brain and heart: this can lead to myopathies and altered neurodevelopment

Hematology complications • Sludging of hyperviscous blood in the cerebral microcirculation can be responsible for symptoms of irritability, jitteriness and high-pitched cry – symptoms which are usually attributed to hypoglycaemia or hypocalcaemia • Infants of mothers with diabetes are also at risk of hyperbilirubinaemia because of the increase in RBC’s, decrease in RBC life, and immature hepatic bilirubin conjugation and excretion

NEUROLOGICAL COMPLICATIONS Brain development may be altered if the mother has poor glycaemic control In the short term these infants may present with symptoms of lethargy, poor feeding and decreased muscle tone Discharge from hospital may be delayed because of issues with establishment of suck feeds Long term studies have shown subtle developmental concerns across the domains of motor (fine & gross) & cognitive skills, coordination, language development and memory tasks

MACROSOMIA • The maternal hyperglycaemic state results in the delivery of excess fuels (carbohydrates) to the fetus • This stimulates fetal pancreatic β-cell hyperplasia and increases fetal insulin and insulin-like growth factors • These growth factors stimulate protein, lipid and glycogen synthesis, causing a high rate of growth, increased deposition of fat, and visceral enlargement

Birth trauma/complications A macrosomic infant can be a causative factor of: Obstructed labour (eg. CPD), & Shoulder dystocia can result in birth trauma such as Brachial plexus injury Fractured clavicle or humerus An obstructed labour or shoulder dystocia can have life threatening implications for the neonate, such as: Perinatal hypoxia-ischaemia (reduced oxygen levels and reduced blood flow)
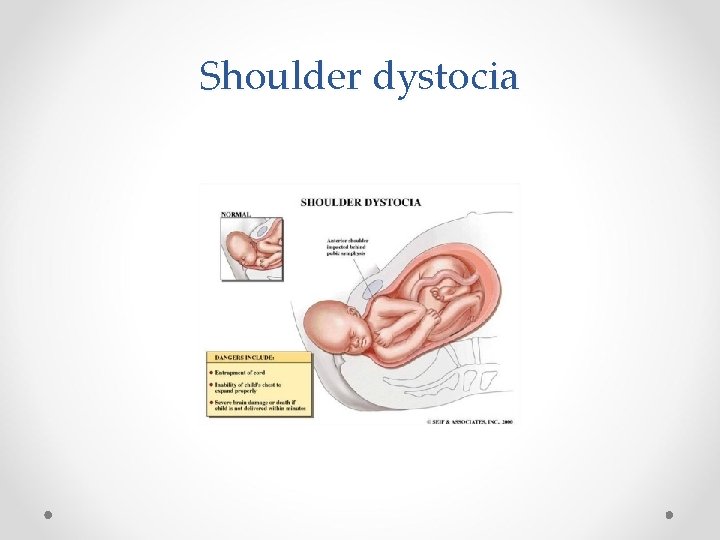
Shoulder dystocia
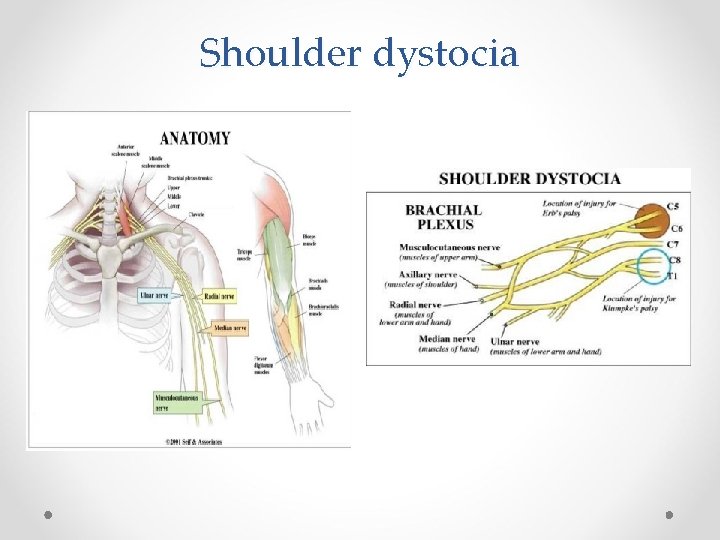
Shoulder dystocia
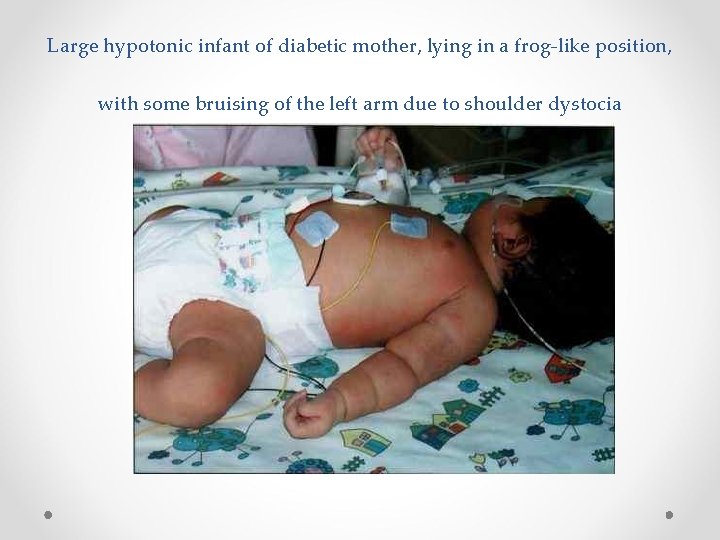
Large hypotonic infant of diabetic mother, lying in a frog-like position, with some bruising of the left arm due to shoulder dystocia

BABY A: A Case Study • 36+6/40 Em LUSCS for fetal distress • Maternal Type 1 DM – poorly controlled - Insulin (1 u/kg/d) • BW 4740 g (off the scale on the growth charts!) • 30 sec IPPV; 5 mins PEEP for grunting/hypoxia • Apgars 7, 8, 10 • SCN admission (due to high risk of hypoglycaemia, mild respiratroy issues, slight prematurity)

Baby A: A Case Study • Ongoing O 2 requirement (managed with cot oxygen, no significant distress, just saturating low. • Unrecordable BGL (<1. 1) taken before 1 hour of life. • IV access gained, baby 40 mins old by this time. • IV dextrose bolus (3 ml/kg) + 60 ml/kg/d infusion commenced • TBG 30 mins later still < 1. 1 (further 3 m. L/kg bolus, TFI increased to 90 m. L/kg) • TBG 30 mins later still < 1. 1 (further 3 ml/kg bolus, TFI increased to 120 m. L/kg) • Preparation by medical staff to insert umbilical lines • Conversation with NETS consultant regarding situation Baby noted to have continuous jittery movements

Baby A: A Case Study • TBG repeated 30 mins later now recordable number of 1. 5 (further 3 m. L/kg bolus and concentration of dextrose increased to 12. 5%) • Remained hypoglycaemia 1 st 5 hrs of life despite 5 x 3 m. L/kg 10% dex bolus • TFI inc from 60 -120 m. L/kg at 12. 5% dex • UVC inserted: increase to 15% dex at 120 m. L/kg • NETS called again, coming to retrieve baby, requested glucagon to be given • Glucagon 1 mg IM given • BGL stabilised by 7 hours of age with 15% dextrose running at 100 m. Ls/kg/d

Baby A: A Case Study • IV benpen & gentamycin for ? sepsis • Delay in NETS retrieval because of other cases being triaged as more critical • NETS T/F at 15 hrs of age to MMC Progress at MMC • Hypoglycaemia: weaned from 15% dex at 100 m. L/kg/d (10. 4 mg/kg/min) to 12. 5% with gradual intro of oral feeds and stabilisation of BGL

Baby A: A Case Study • Respiratory: Cot Oxygen initially, CPAP for transfer, 23% Fi. O 2 – weaned to SVIA at MMC • Sepsis: LP performed day 3 due to inc CRP – cefotaxime added to benpen + gent – CSF NAD; anti’s ceased day 6 • Heart murmur: ESM LSE ? flow murmur – resolved pre discharge • Thrombocytopenia: plt 63 on day 3; spont improved to 102 by day 5

Baby A: A Case Study • • Transferred to SJOG day 7 due to bed shortage at BHS TF to BHS day 9 to continue establishment of feeds DC home day 13, CA: 38. 4 wks. Weight: 4410 g For paediatric follow up, cranial ultrasound, enhanced M&CHN program This was a very severe case of neonatal hypoglycaemia due to gestational diabetes. But this isn’t the only reason babies can have experience hypoglycaemia……. .

HYPOGLYCAEMIA • There is a lack on consensus on what defines hypoglycaemia in the neonate. • A clinical trial is not an option/possible. • Clinical guidelines should be defined by operational thresholds (ie. blood glucose levels at which clinical interventions should be considered). • Profound hypoglycaemia (TBG <1. 5 mmol/L) is more likely to result in long term sequelae, with longer durations adding to the risk. • Symptomatic neonates have a worse prognosis.

Hypoglycaemia Which babies should we routinely monitor for hypoglycaemia? Maternal indications: mother who has diabetes (up to 50% of these infants may become hypoglycaemic, the incidence is also related to glucose control in pregnancy/Hb. A 1 C levels, medications and dextrose in labour) Infant indications: SGA (<10 th centile), LBW (<2500 g), macrosomic (>90 th centile), prematurity, NBM, RDS, sepsis, hypothermia, asphyxia (arterial cord p. H<7. 2) – in fact most babies admitted to a SCN/NICU will have blood glucose monitoring initially.

Hypoglycaemia • For the neonate of the mother with diabetes the biggest risk time is the first 1 – 3 hrs of life • Maternal supply of glucose abruptly ceases, but there is a lag in the reduction of circulating insulin • But problems can continue to occur over the first week of life • The macrosomic baby is at greatest risk (47% will have issues with hypoglycaemia, compared to 20% of those that are non-macrosomic)

Signs & symptoms are non-specific Abnormal muscle tone Abnormal level of consciousness, lethargy Poor oral feeding Seizures Jitteriness Tachypnoea Cyanotic episodes High pitched cry, irritability MANY BABIES WILL HAVE NO SIGNS OF HYPOGLYCAEMIA

Measuring blood glucose • Blood glucose levels (BGL’s) are measured by a blood glucose monitor and reagent strips – these are not considered to be reliable outside of the ranges of 2. 5 – 8. 0 mmol/L (particularly with neonates because of their elevated haematocrit levels) • Preferable to have low results confirmed with a TBG (from the lab, or a blood gas analyser that some units will have); the length in delay of obtaining a TBG would need to be considered with regard to prompt treatment in case the low result is correct.

Management of hypoglycaemia • Early, frequent and adequate milk feeds • IV glucose (10%): maintenance, +/- bolus • If increases in TFI are not correcting the problem the concentration of glucose can be increased to 12. 5%, 15% & 20% (anything over 12. 5% needs to be through a central line) • IV/IM glucagon • Hydrocortisone, diazoxide, octreotide

MANEGEMENT: DIAZOXIDE • Commonly used in adults as an antihypertensive agent in acute situations. • Used in neonates as an insulin suppressor. • Has become a first line drug for the management of hyperinsulinaemia (administered at 5 – 15 mg/kg/day). • Can be effective in controlling insulin secretion and promoting mobilization of glycogen stores. • Can take up to 7 days to be effective. • Doses < 20 mg/kg/day appear to be safe and can be used long term.

CALCULATING GLUCOSE LOAD Average glucose consumption should be between 46 mg/kg/min Some preterm & SGA infants may require 6 -8 mg/kg/min to maintain normal plasma glucose levels If a neonate is requiring more than 10 mg/kg/min to maintain adequate glucose levels further investigation is required Calculating glucose load (mg/kg/min): Daily volume (m. L/kg/day) x %dextrose/144=glucose load (for breast milk you can assume the % is about 7%)

CALCULATING GLUCOSE LOAD Example: Baby A Birth Weight: 4740 g 15% dextrose 100 m. L/kg/day Calculating glucose load (mg/kg/min): Daily volume (m. L/kg/day) x %dextrose/144=glucose load 100 x 15/144 = 10. 4 mg/kg/min

PERSISTENT HYPOGLYCAEMIA If hypoglycaemia continues for longer than expected and does not respond to appropriate management further studies may be required: Insulin levels, Ketones (serum and urine), Cortisol, Growth hormone, Lactate, Pyruvate & Serum electrolytes. The optimum time to perform these tests is when the infant is having a hypoglycaemic episode.

CAUSES OF HYPOGLYCAEMIA The causes of hypoglycaemia can be divided into several broad categories based on the mechanisms producing the hypoglycaemia: • Inadequate substrate supply, • Abnormal endocrine regulation of glucose utilization & • Increased rate of glucose utilization. There also several etiological conditions for which mechanisms are not well defined.

INADEQUATE SUBSTRATE SUPPLY • If supply is inadequate hepatic glucose output will not meet metabolic demands. • Usually results from subnormal fat and glycogen stores. • Infants born preterm have diminished glycogen stores. • Infant with IUGR secondary to placental insufficiency also at risk for decreased glycogen accumulation. • Glycogenolysis and gluconeogenesis can be impaired in premature infants for a prolonged period.

INADEQUATE SUBSTRATE SUPPLY • The subnormal fat and glycogen stores do not provide sufficient energy to maintain glucose homeostasis until gluconeogenesis reaches adequate levels. • Infants born preterm have diminished glycogen stores because most of the hepatic glycogen is accumulated during the 3 rd trimester. • IUGR: presumably causes problems because of diminished transfer of glycogen precursors (eg. glucose and lactate) across the placenta; the limited supply of these is then used for oxidative metabolism rather than building up the fat or glycogen stores in utero. • Up to 18% of preterm infants have problems in maintaining normoglycaemia at the time of discharge if a feed is omitted or delayed. • Inadequate cortisol secretion in very preterm infants, particularly during periods of stress, also has been cited as a cause of limited activation of gluconeogenic enzymes.

INCREASED GLUCOSE UTILISATION • Normal stores, intact regulating mechanisms but an increased energy demand, such as in cases: That cause a shift from aerobic to anaerobic metabolism (asphyxia, hypotension, severe lung disease, septic shock); Asphyxia results in anaerobic metabolism, this is inefficient and more glucose is used than would be under normal aerobic conditions; hypoxic damage to liver can impair normal postnatal onset of gluconeogenesis; may also be elevated insulin levels present in these situations. Hypothermia: rapid depletion of brown fat stores for nonshivering thermogenesis and secondary breakdown and exhaustion of glycogen stores.

ENZYMATIC AND GENETIC DISORDERS • Hormone deficiencies (such as growth hormone and cortisol). Growth hormone and cortisol oppose the action of insulin; they increase blood glucose concentrations by reducing glucose uptake in muscle tissue and they stimulate lipolysis and gluconeogenesis. • Enzyme deficiency conditions • Defective carbohydrate metabolism • Defects in amino acid metabolism • Defects in fatty acid metabolism

THE ENDOCRINE SYSTEM Comprises: the hypothalymus (the pituitary, pineal, thyroid, parathyroid and adrenal glands); the gonads; and the pancreatic islet cells. Some of the problems that occur in the newborn are related to the metabolism of glucose, calcium, phosphorous and magnesium that are regulated by the endocrine system.

ABNORMALITIES OF ENDOCRINE REGULATION • Hyperinsulinaemia is the most common endocrinological disturbance producing neonatal hypoglycaemia. • Excessive insulin secretion in the newborn increases glucose utilization by stimulating cellular glucose uptake in insulin-dependent tissues. • The most common clinical situation where hyperinsulinaemia occurs is in the neonate of a mother with IDDM. • Beckwith-Weidermann syndrome. • Prolonged neonatal hypoglycaemia. • Persistent hyperinsulinaemic hypoglycaemia.

PROLONGED NEONATAL HYPOGLYCAEMIA • Common, but not well recognised or understood • Usually associated with stress before, or during birth • May present as IUGR; birth asphyxia; secondary to maternal pre-eclampsia • Usually manifests in the first few days and often may be severe • May last up to 4 weeks • Does not respond well to glucocorticoids or frequent feeds • May be treated with diazoxide at doses of 5 – 10 mg/kg/day.

Avoiding harm/reducing risk • Hospitals should have written protocols for prevention and management of potential neonatal complications • If complications are expected babies should be delivered in hospitals that have the expertise to manage these complications • Aim to avoid iatrogenic harm by avoiding unnecessary preterm delivery or caesarean section and unnecessary separation of mother and baby

Iatrogenic complications • Routine admission of babies to neonatal units • Routine supplementation or replacement of breast feeds with formula • Delayed skin to skin and first feed • Poor management of temperature control • Blood glucose tested and acted upon too soon after birth

REFERENCES Hawdon J. M. NICE guidance for neonatal care after diabetes in pregnancy. Infant 2008; 4(5): 154 -58. Narchi H. & Kulaylat N. Heart disease in infants of diabetic mothers. Images in paediatric cardiology 2000; 3: 17 -23. Miller M. Q. & Morris L. A. Devlopmental considerations in working with newborn infants of mothers with diabetes. Neonatal Network 2011; 4: 37 -45. Powell L. L. Infants of diabetic mothers: The effects of hyperglycaemia on the fetus and neonate. Neonatal Network 2007; 26: 283 -290.

References cont. . Cresto, J. C. , Abdenur, J. P. , Bergada, I. , & Martino, R. (1998). Long term follow up of persistent hyperinsulinaemic hypoglycaemia of infancy. Arch Dis Child. 79: 440 -444. http: //www. healthatoz. com (pancreatectomy) Gallager, M. P. , Chung, W. K. , & Oberfield, S. E. (2007). Endocrinology and metabolism. In R. A. Polin & A. R. Spitzer (Eds. ), Fetal and neonatal secrets, (2 nd ed. , pp. 130 -153). Philadelphia, Pennsylvania: Mosby, Elsevier. Glanze, W. D. (ed), (1986). Mosby’s Medical and Nursing Dictionary. St. Louis: The C. V. Mosby Company. Helwick, C. A. (2002). Pancreatectomy. Gale encyclopedia of medicine. The Gale Group: http: //www. healthatoz. com/healthatoz/Atoz/common/standard/transform. jsp? Hussain, K. (2007). Diagnosis and management of hyperinsulinaemic hypoglycaemia of infancy. Hormone Research, 69: 2 -13. Kenner, C. , & Wright Lott, J. (2004). Neonatal Nursing Handbook. St. Louis, Missouri: Saunders. Mc. Gowan, J. E. , Price-Douglas, W. & Hay, W. W. (2006). Glucose homeostasis. In G. B. Merenstein & S. L. Gardner (Eds. ), Handbook of neonatal intensive care, (6 th ed. , pp. 368 -390). St. Louis, Missouri: Mosby, Elsevier. Mims Online: http: //www. mims. com. au Normal Physiology of Insulin release – Athena Diagnostics: http: //www. athenadiagnostics. com/content/diagnostics-ed/endocrinology Stockowski, L. (2004). Endocrine disorders. In M. T. Verklan & M. Waldan (Eds. ), Core curriculum for neonatal intensive care nursing, (3 rd ed. , pp. 703 -727). St. Louis, Missouri: Elsevier, Saunders.
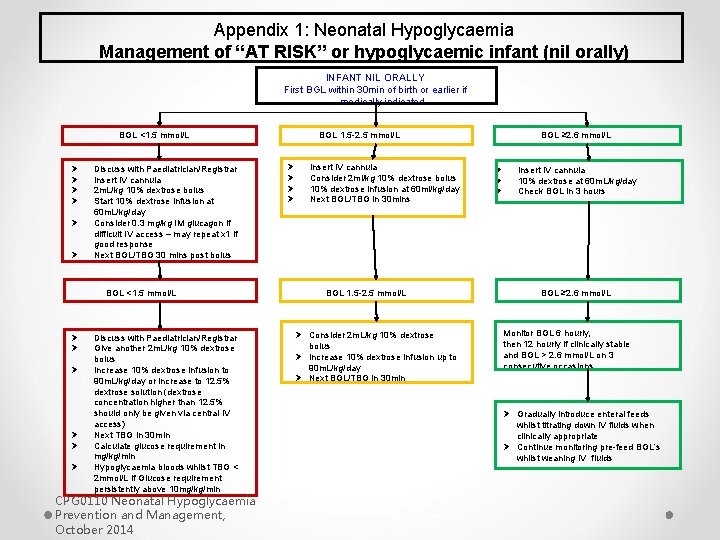
Appendix 1: Neonatal Hypoglycaemia Management of “AT RISK” or hypoglycaemic infant (nil orally) INFANT NIL ORALLY First BGL within 30 min of birth or earlier if medically indicated BGL <1. 5 mmol/L Ø Ø Ø Discuss with Paediatrician/Registrar Insert IV cannula 2 m. L/kg 10% dextrose bolus Start 10% dextrose infusion at 60 m. L/kg/day Consider 0. 3 mg/kg IM glucagon if difficult IV access – may repeat x 1 if good response Next BGL/TBG 30 mins post bolus BGL <1. 5 mmol/L Ø Ø Ø Discuss with Paediatrician/Registrar Give another 2 m. L/kg 10% dextrose bolus Increase 10% dextrose infusion to 90 m. L/kg/day or increase to 12. 5% dextrose solution (dextrose concentration higher than 12. 5% should only be given via central IV access) Next TBG in 30 min Calculate glucose requirement in mg/kg/min Hypoglycaemia bloods whilst TBG < 2 mmol/L if Glucose requirement persistently above 10 mg/kg/min CPG 0110 Neonatal Hypoglycaemia Prevention and Management, October 2014 BGL 1. 5 -2. 5 mmol/L Ø Ø Insert IV cannula Consider 2 ml/kg 10% dextrose bolus 10% dextrose infusion at 60 ml/kg/day Next BGL/TBG in 30 mins BGL 1. 5 -2. 5 mmol/L Ø Consider 2 m. L/kg 10% dextrose bolus Ø Increase 10% dextrose infusion up to 90 m. L/kg/day Ø Next BGL/TBG in 30 min BGL ≥ 2. 6 mmol/L Ø Ø Ø Insert IV cannula 10% dextrose at 60 m. L/kg/day Check BGL in 3 hours BGL ≥ 2. 6 mmol/L Monitor BGL 6 hourly, then 12 hourly if clinically stable and BGL > 2. 6 mmol/L on 3 consecutive occasions Ø Gradually introduce enteral feeds whilst titrating down IV fluids when clinically appropriate Ø Continue monitoring pre-feed BGL’s whilst weaning IV fluids
- Slides: 53