Hydrothermal Synthesis of Nanomaterials Andrew van Bommel January

Hydrothermal Synthesis of Nanomaterials Andrew van Bommel January 18 th, 2006

Hydrothermal Synthesis • The reactants are dissolved (or placed) in water or another solvent (solvothermal) in a closed vessel • Bomb is heated above BP • Conventional or MW oven • Commercially: – Tons of zeolites daily Nazari, G. A. , Pistoria, G. Lithium Batteries; Klumer Academic Pubs. : Boston, 2004.

Conditions • Solvent above boiling point (supercritical water) • Usually basic conditions • Useful for Nano! – Oxides (Ti. O 2, VOx, Mx. Mn. O 2) – Layered oxides: nanowires/tubes – C-nanotubes can be formed this way – Some elemental nanostructures (eg: Bi)

Mechanism for HT rxn • Usually follows a liquid nucleation model • Differs from solid-state- reaction mechanism from diffusion of atoms or ions between reactants • Due to enhanced solubility: – Solubility of water increases with temperature, but alkaline solubility increases dramatically with temperature Barrer, R. M. Hydrothermal Chemistry of Zeolites; Academic Press: London, 1982
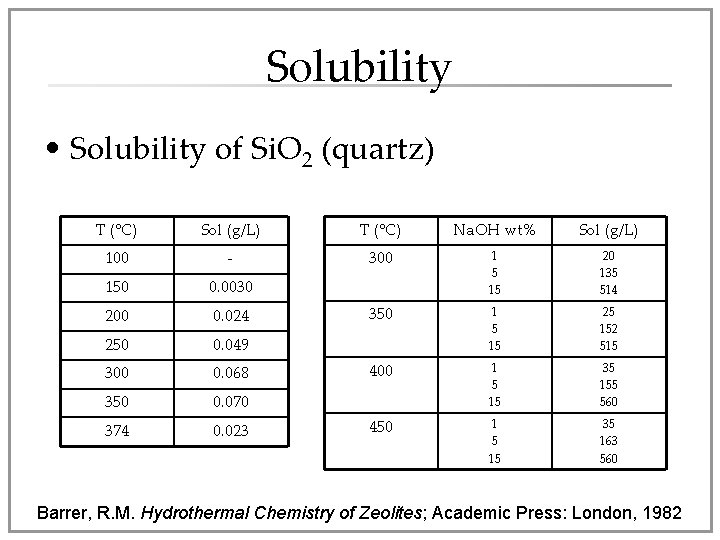
Solubility • Solubility of Si. O 2 (quartz) T (°C) Sol (g/L) T (°C) Na. OH wt% Sol (g/L) 100 - 300 150 0. 0030 1 5 15 20 135 514 200 0. 024 350 250 0. 049 1 5 15 25 152 515 300 0. 068 400 350 0. 070 1 5 15 35 155 560 374 0. 023 450 1 5 15 35 163 560 Barrer, R. M. Hydrothermal Chemistry of Zeolites; Academic Press: London, 1982

Next Slides • Examples: – Nanoparticles – Nanowires/tubes – Nanoflowers • Control of Shape

Ba. Ti. O 3 Nanoparticles • Ba(OH)2 + Ti. O 2 Ba. Ti. O 3 nanoparticles • 300 - 450°C, HT • Two proposed mechanisms: – Dissolution-recrystallization – In situ crystallization Hakuta, R. , Ura, H. Hayashi, H, and Arai, K. Ind. Eng. Chem. Res. 2005, 44, 840 -846
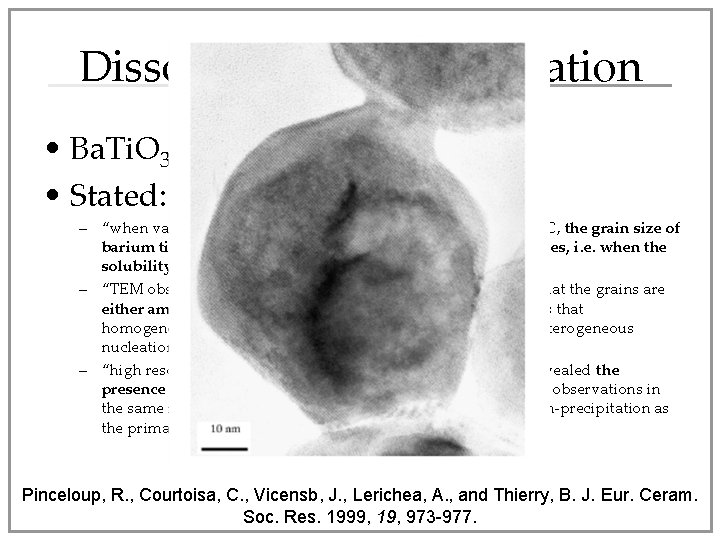
Dissolution-Recrystallization • Ba. Ti. O 3 formation at 150°C • Stated: – “when varying the water/isopropanol ratio in synthesis at 150°C, the grain size of barium titanate decreases when the amount of alcohol increases, i. e. when the solubility of the precursors decreases” – “TEM observations of incompletely reacted powders showed that the grains are either amorphous or entirely crystalline Ba. Ti. O 3, which means that homogeneous nucleation and growth is occurring instead of heterogeneous nucleation – “high resolution TEM observations of fully reacted powders revealed the presence of necks between particles. These three experimental observations in the same reaction system provide strong evidence of dissolution-precipitation as the primary reaction mechanism. ” Pinceloup, R. , Courtoisa, C. , Vicensb, J. , Lerichea, A. , and Thierry, B. J. Eur. Ceram. Soc. Res. 1999, 19, 973 -977.

In-situ crystallization • Non-dissolved Ti. O 2 reacts with barium and hydroxide ions Eckert, J. O. , Hung-Houston, C. C. , Gersten, B. L. , Lencka, M. M. , Riman, R. E. , J. Am. Ceram. Soc. 1996, 79, 2939.

Why Nano? • For in-situ transformation, formation of a porous product facilitates the transfer of the reacting species, leading to a facile reaction • For dissolution-precipitation reaction, dissolution (and subsequent precipitation) must be fast to ensure a steady flow of reactants Eckert, J. O. , Hung-Houston, C. C. , Gersten, B. L. , Lencka, M. M. , Riman, R. E. , J. Am. Ceram. Soc. 1996, 79, 2939.
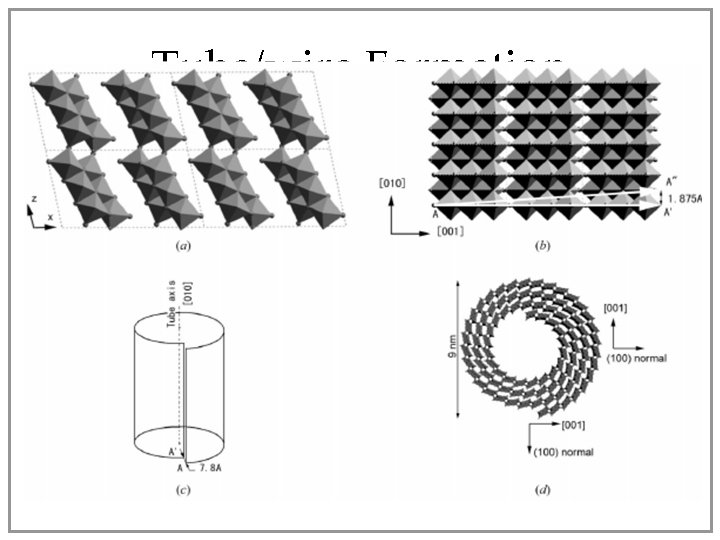
Tube/wire Formation • Ti. O 2 +Na. OH titanate nanomaterial • Crystal flakes rolled along [001] direction Chen, Q. , Du, G. H. , Zhang, S. , Peng, L. M. Acta Cryst. 2002, B 58, 587 -593.

Why nanowires/tubes? • Two-dimensional crystal flakes have low resistance to bending: hydrothermal energy curls these flakes • Why tubes? – When diameter grows, the strain in the tubes is outweighed by the minimizing of energy with nanotubes’ self-closed layers (no dangling bonds) Remskar, M. Adv. Mater. 2004, 16, 1497 -1502.
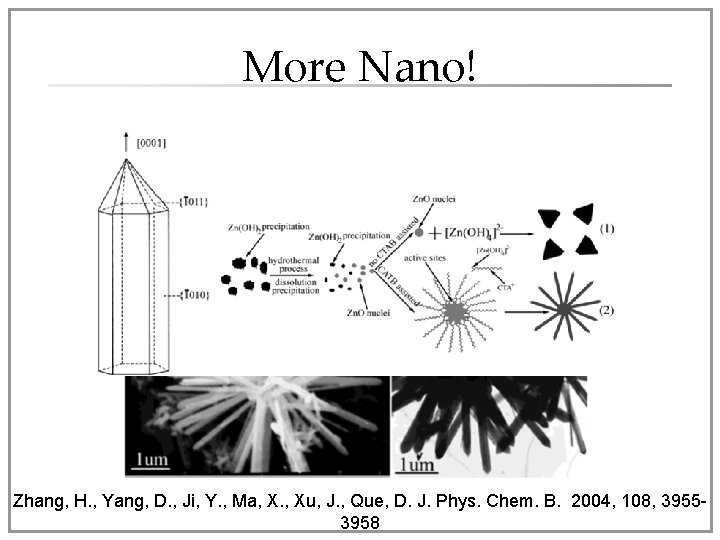
More Nano! • Nanoflower Zn. O- CTAB assisted hydrothermal reaction of Zn. O (in Na. OH solution) Zhang, H. , Yang, D. , Ji, Y. , Ma, X. , Xu, J. , Que, D. J. Phys. Chem. B. 2004, 108, 39553958

Hydrothermal Route • Pros: – New materials – Easy, relatively cheap • Cons: – Difficult to control morphology, size – Not for all materials – May obtain variation in size

Summary • Hydrothermal synthesis involves the chemical reaction of materials in aqueous solution heated (usually above BP) in a sealed vessel (bomb) • Alkaline solution used to increase solubility (for a dissolution-precipitation reaction) • It is difficult to predict the resultant morphology of the product

Summary • Nanomaterials are apt to form in HT synthesis as the formation of these structures allows for facile diffusion of reactants • Hydrothermal synthesis is used for oxide nanoparticle synthesis as the solubility is high in the alkaline medium
- Slides: 16