Hydrosilylation of Alkenes and Alkynes Catalyzed by NHC

Hydrosilylation of Alkenes and Alkynes Catalyzed by NHC Platinum(0) Complexes The Ph. D. Students’ Day June 1, 2012 Louvain-la-Neuve Steve Dierick István E. Markó

Outline 1. What is the hydrosilylation reaction? 2. Hydrosilylation of C−C multiple bonds with Platinum(0) carbene complexes a) 1 st generation catalyst in alkenes hydrosilylation The breakthrough b) 1 st and 2 nd generation catalysts in alkynes hydrosilylation More challenging substrates c) 3 rd generation catalyst in alkenes and alkynes hydrosilylation How serendipity, intuitions and detailed mechanistic studies lead to discovery 3. Conclusions and Perspectives 1

Hydrosilylation Addition of a Si−H unit across multiple bonds 1 Straightforward and atom-economical formation of Si−C bonds 2 → second most important reaction in the silicone industry → used for the production of silicone-based polymers, fluids, surfactants, adhesives etc… Since 1953, transition metals, particularly platinum, are used extensively as catalysts for this transformation 3 1. Marciniec, B. ; Maciejewski, H. ; Pietraszuk, C. ; Pawluć, P. In Hydrosilylation: A Comprehensive Review on Recent Advances; Marciniec, B. , Ed. ; Springer: New York, 2009 2. Troegel, D. ; Stohrer, J. Coord. Chem. Rev. 2011, 255, 1440 3. Wagner, G. H. (Union Carbide) U. S. Patent 2632013, 1953 2
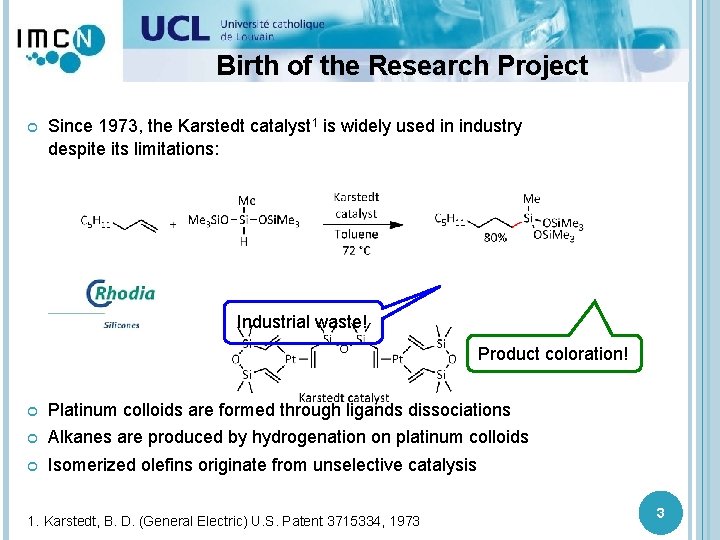
Birth of the Research Project Since 1973, the Karstedt catalyst 1 is widely used in industry despite its limitations: Industrial waste! Product coloration! Platinum colloids are formed through ligands dissociations Alkanes are produced by hydrogenation on platinum colloids Isomerized olefins originate from unselective catalysis 1. Karstedt, B. D. (General Electric) U. S. Patent 3715334, 1973 3

Birth of the Research Project How could all these shortcomings be tackled? l Through stabilization of the platinum species involved → avoid platinum colloids and thus hydrogenation l Via the fine tuning of the catalyst properties → control the catalyst reactivity and selectivity Both objectives might be addressed by introducing a strongly binding ancillary ligand on platinum! 4
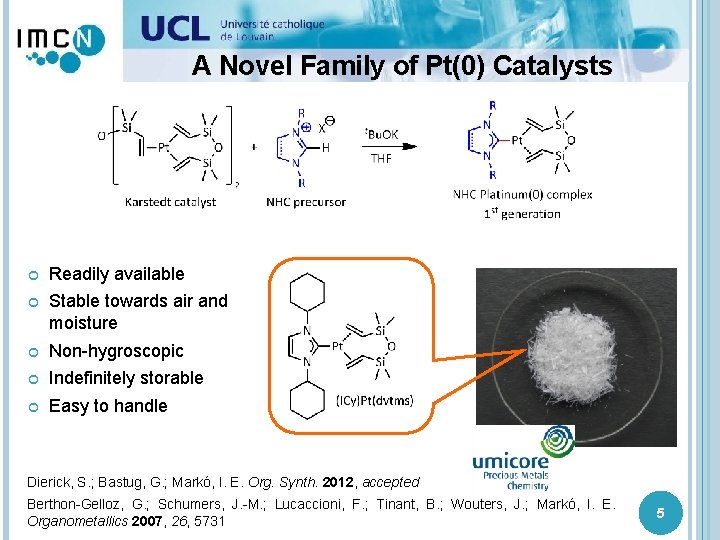
A Novel Family of Pt(0) Catalysts Readily available Stable towards air and moisture Non-hygroscopic Indefinitely storable Easy to handle Dierick, S. ; Bastug, G. ; Markó, I. E. Org. Synth. 2012, accepted Berthon-Gelloz, G. ; Schumers, J. -M. ; Lucaccioni, F. ; Tinant, B. ; Wouters, J. ; Markó, I. E. Organometallics 2007, 26, 5731 5
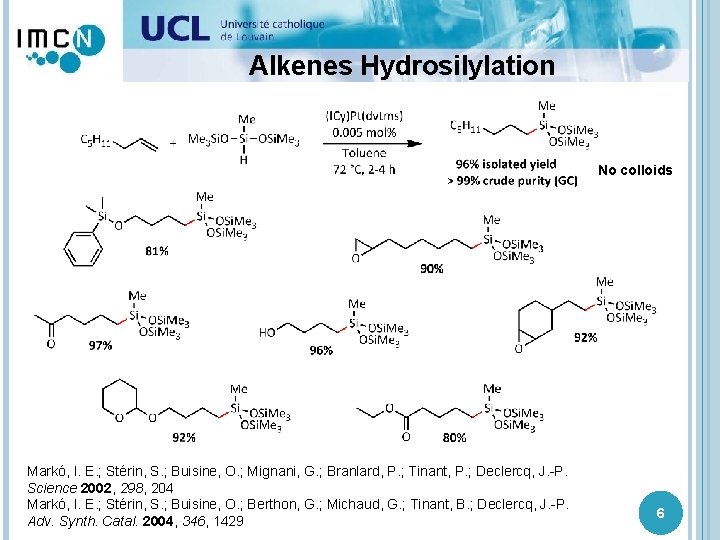
Alkenes Hydrosilylation No colloids Markó, I. E. ; Stérin, S. ; Buisine, O. ; Mignani, G. ; Branlard, P. ; Tinant, P. ; Declercq, J. -P. Science 2002, 298, 204 Markó, I. E. ; Stérin, S. ; Buisine, O. ; Berthon, G. ; Michaud, G. ; Tinant, B. ; Declercq, J. -P. Adv. Synth. Catal. 2004, 346, 1429 6
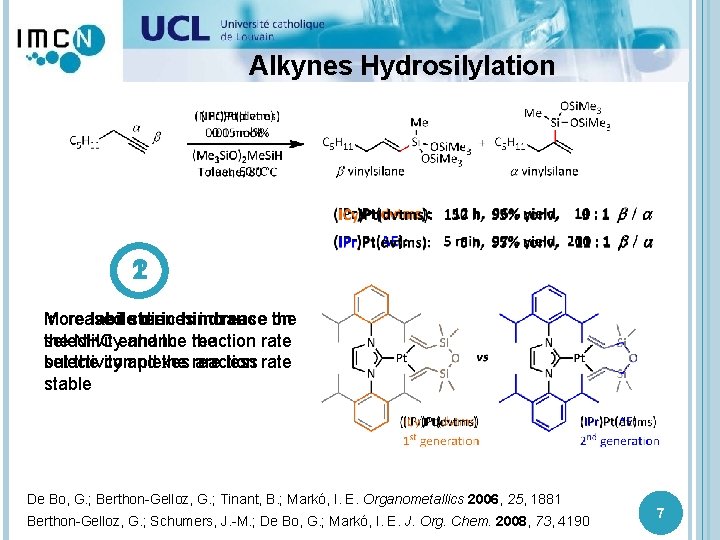
Alkynes Hydrosilylation 1 2 Increased More labilesteric dienes hindrance increase the on selectivity the NHC enhance and the reaction the rate but the complexes selectivity and the reaction are less rate stable De Bo, G. ; Berthon-Gelloz, G. ; Tinant, B. ; Markó, I. E. Organometallics 2006, 25, 1881 Berthon-Gelloz, G. ; Schumers, J. -M. ; De Bo, G. ; Markó, I. E. J. Org. Chem. 2008, 73, 4190 7
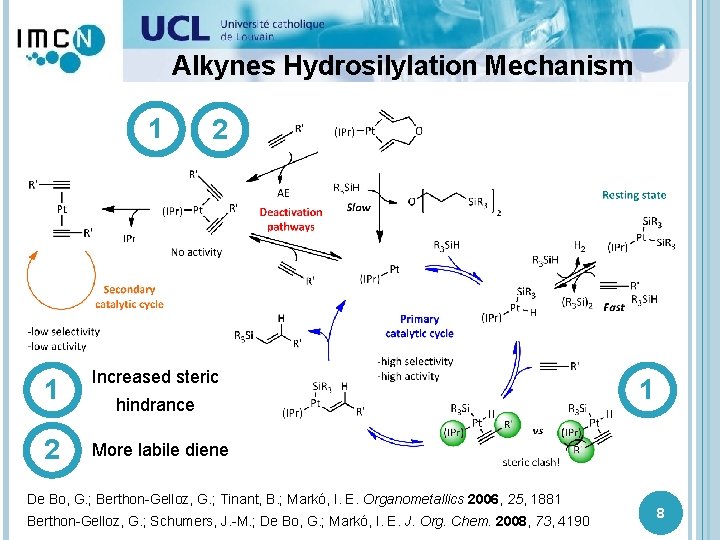
Alkynes Hydrosilylation Mechanism 1 2 1 Increased steric 2 More labile diene hindrance De Bo, G. ; Berthon-Gelloz, G. ; Tinant, B. ; Markó, I. E. Organometallics 2006, 25, 1881 Berthon-Gelloz, G. ; Schumers, J. -M. ; De Bo, G. ; Markó, I. E. J. Org. Chem. 2008, 73, 4190 1 8
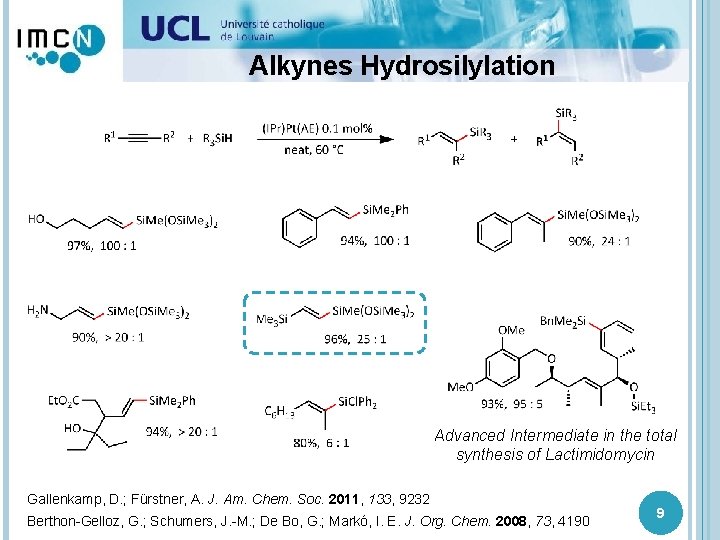
Alkynes Hydrosilylation Advanced Intermediate in the total synthesis of Lactimidomycin Gallenkamp, D. ; Fürstner, A. J. Am. Chem. Soc. 2011, 133, 9232 Berthon-Gelloz, G. ; Schumers, J. -M. ; De Bo, G. ; Markó, I. E. J. Org. Chem. 2008, 73, 4190 9

Silylated Alkynes Hydrosilylation The reaction is sluggish and affords low selectivities: → silylated alkynes promote the deactivation pathways! Serendipity Optimisation reaction catalysts conditions: → 1 st and 2 ofndthe generation give the same results? ! catalyst loading, temperature, silane, dilution, slow addition, addition order, reagents purity, There must be a common intermediate ! Intuition inert atmosphere… Dierick, S. ; Markó, I. E. Unpublished results 10
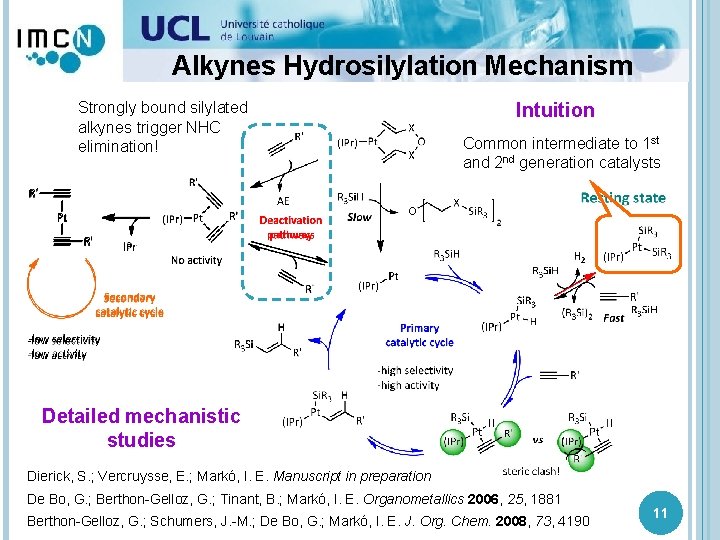
Alkynes Hydrosilylation Mechanism Strongly bound silylated alkynes trigger NHC elimination! Intuition Common intermediate to 1 st and 2 nd generation catalysts Detailed mechanistic studies Dierick, S. ; Vercruysse, E. ; Markó, I. E. Manuscript in preparation De Bo, G. ; Berthon-Gelloz, G. ; Tinant, B. ; Markó, I. E. Organometallics 2006, 25, 1881 Berthon-Gelloz, G. ; Schumers, J. -M. ; De Bo, G. ; Markó, I. E. J. Org. Chem. 2008, 73, 4190 11
![3 rd Generation Hydrosilylation Catalyst [Pt 0] Time (min) Ratio β : α Yield 3 rd Generation Hydrosilylation Catalyst [Pt 0] Time (min) Ratio β : α Yield](http://slidetodoc.com/presentation_image_h2/ecf70622c624419bb285a6e9ef680e06/image-13.jpg)
3 rd Generation Hydrosilylation Catalyst [Pt 0] Time (min) Ratio β : α Yield (%) (IPr)Pt(dvtms) 420 12 : 1 95 (IPr)Pt(AE) 240 16 : 1 95 (IPr)Pt(Si. Me 2 Ph)2 < 15 23 : 1 96 → best catalyst developed to date! Berthon-Gelloz, G. ; de Bruin, B. ; Tinant, B. ; Markó, I. E. Angew. Chem. , Int. Ed. 2009, 48, 3161 Dierick, S. ; Vercruysse, E. ; Markó, I. E. Manuscript in preparation 12
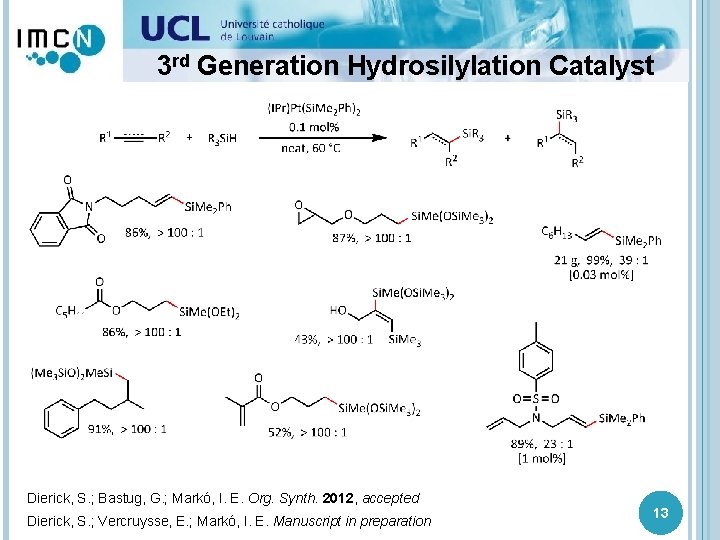
3 rd Generation Hydrosilylation Catalyst Dierick, S. ; Bastug, G. ; Markó, I. E. Org. Synth. 2012, accepted Dierick, S. ; Vercruysse, E. ; Markó, I. E. Manuscript in preparation 13

Conclusions & Perspectives NHC Platinum(0) complexes are the ideal user-friendly reagents for the hydrosilylation of alkenes and alkynes: High activity Readily available High selectivity Indefinitely storable Low catalyst loading Easy to handle Exploration of the selectivity between C−C multiple bonds Hydrosilylation of challenging allyl substrates Asymmetric version of the process Catalysts samples are available on request! We are always open to new collaborations! For reviews on NHC Pt(0) complexes, see: a) Dierick, S. ; Markó, I. E. “(IPr)Pt(dvtms)” In Encyclopedia of reagents for Organic Synthesis (e-EROS); Wiley, ASAP 2012. b) Berthon. Gelloz, G. ; Markó, I. E. In N-Heterocyclic Carbenes in Synthesis; Nolan, S. P. , Ed. ; Wiley, 2006 14

Acknowledgment Hydrosilylation family: Dr. Buisine, O. Dr. Stérin, S. Dr. Michaud, G. Dr. Berthon, G. Dr. De Bo, G. Dr. Schumers, J. -M. Flamant, M. Vercruysse E. Prof. Markó group: Himself Lucaccioni, F. All my colleagues ! Financial support: UCL, FRIA, FNRS, ARC Rhodia Silicones, Umicore The organising committee and YOU !
- Slides: 16