Hydrogen Storage by Spillover Graduate Student Research Talk
Hydrogen Storage by Spillover Graduate Student Research Talk 10/2/09
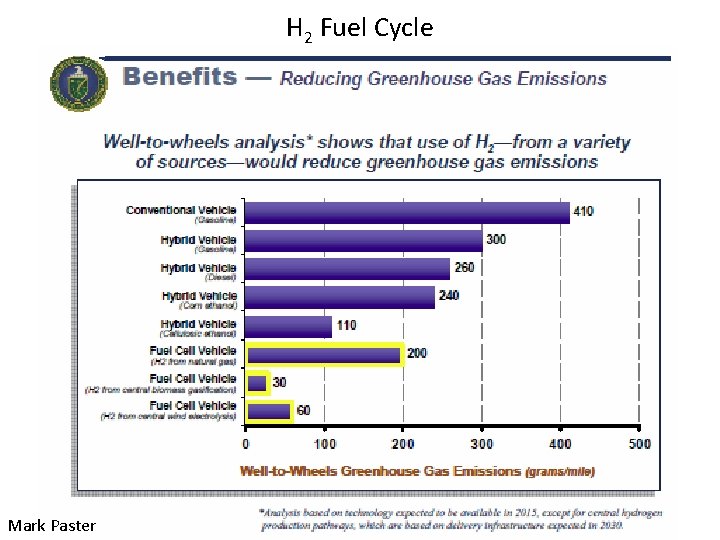
H 2 Fuel Cycle Mark Paster
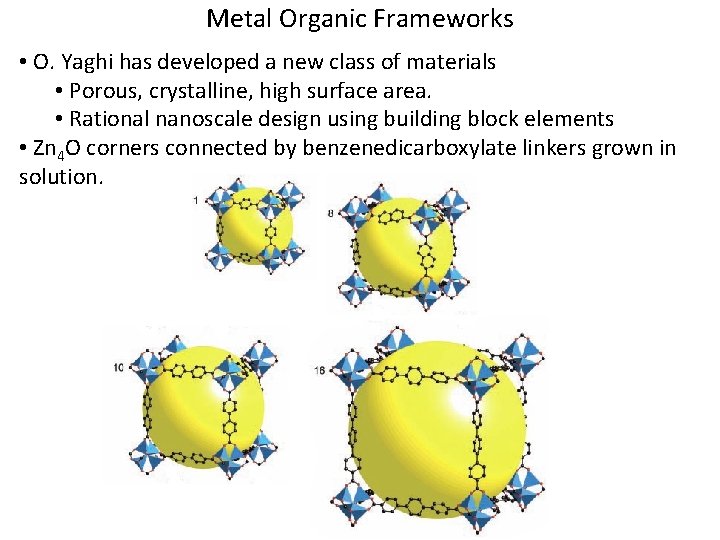
Metal Organic Frameworks • O. Yaghi has developed a new class of materials • Porous, crystalline, high surface area. • Rational nanoscale design using building block elements • Zn 4 O corners connected by benzenedicarboxylate linkers grown in solution.
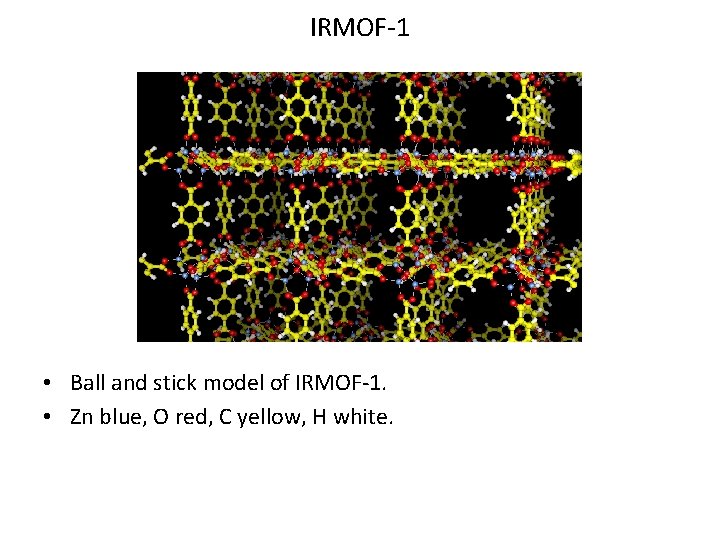
IRMOF-1 • Ball and stick model of IRMOF-1. • Zn blue, O red, C yellow, H white.
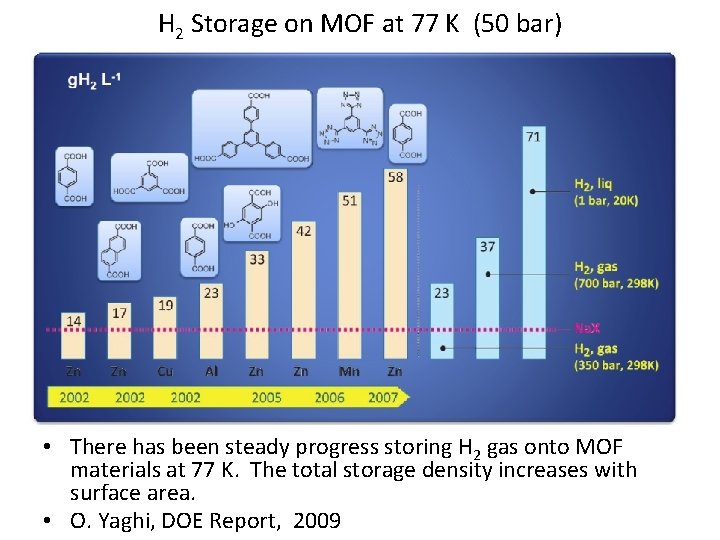
H 2 Storage on MOF at 77 K (50 bar) • There has been steady progress storing H 2 gas onto MOF materials at 77 K. The total storage density increases with surface area. • O. Yaghi, DOE Report, 2009
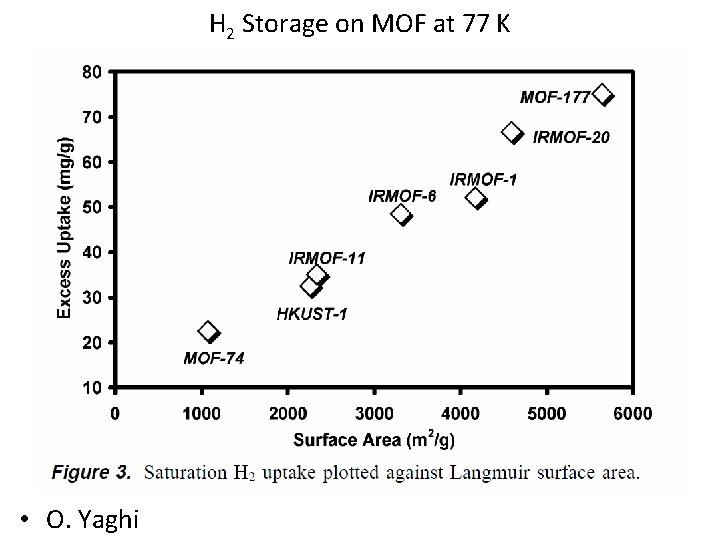
H 2 Storage on MOF at 77 K • O. Yaghi
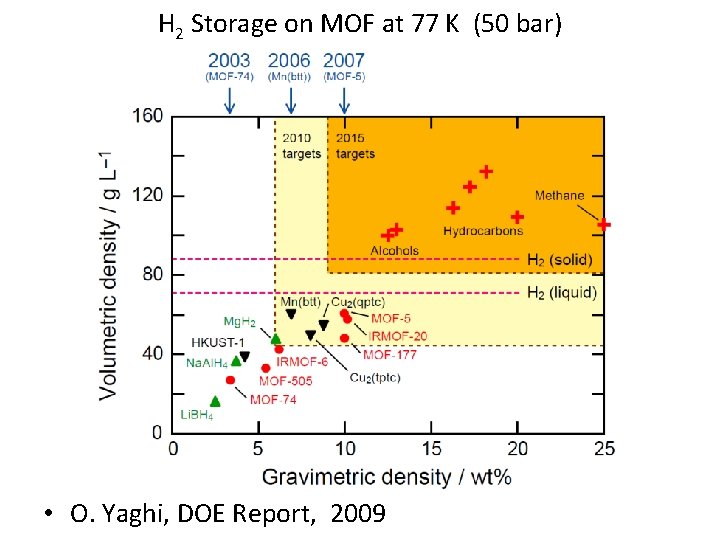
H 2 Storage on MOF at 77 K (50 bar) • O. Yaghi, DOE Report, 2009
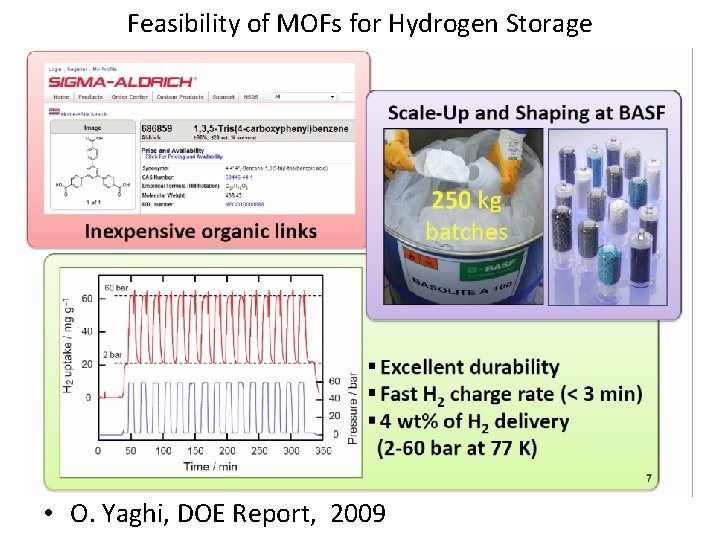
Feasibility of MOFs for Hydrogen Storage • O. Yaghi, DOE Report, 2009
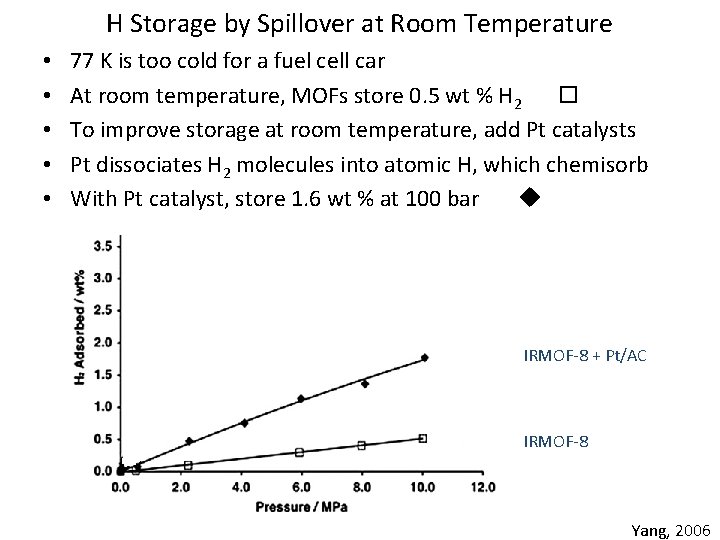
H Storage by Spillover at Room Temperature • • • 77 K is too cold for a fuel cell car At room temperature, MOFs store 0. 5 wt % H 2 To improve storage at room temperature, add Pt catalysts Pt dissociates H 2 molecules into atomic H, which chemisorb With Pt catalyst, store 1. 6 wt % at 100 bar IRMOF-8 + Pt/AC IRMOF-8 Yang, 2006
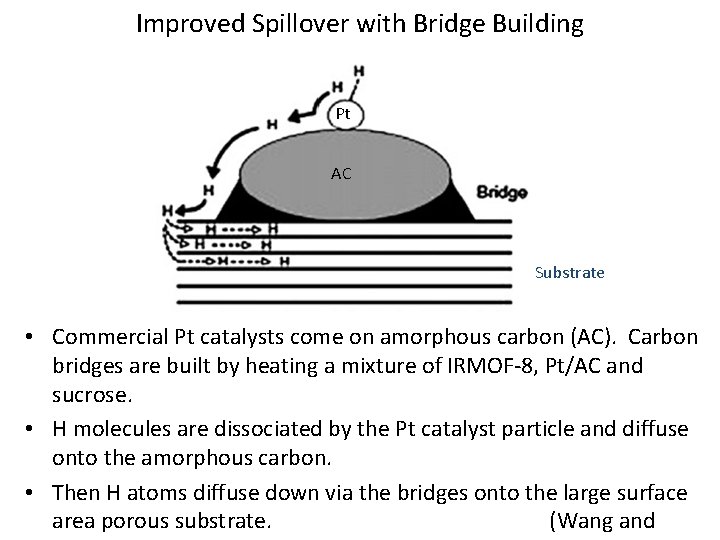
Improved Spillover with Bridge Building Pt AC Substrate • Commercial Pt catalysts come on amorphous carbon (AC). Carbon bridges are built by heating a mixture of IRMOF-8, Pt/AC and sucrose. • H molecules are dissociated by the Pt catalyst particle and diffuse onto the amorphous carbon. • Then H atoms diffuse down via the bridges onto the large surface area porous substrate. (Wang and
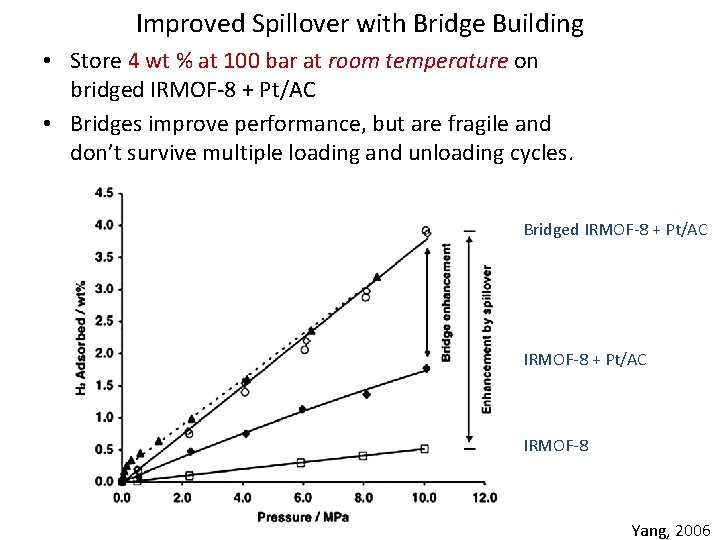
Improved Spillover with Bridge Building • Store 4 wt % at 100 bar at room temperature on bridged IRMOF-8 + Pt/AC • Bridges improve performance, but are fragile and don’t survive multiple loading and unloading cycles. Bridged IRMOF-8 + Pt/AC IRMOF-8 Yang, 2006
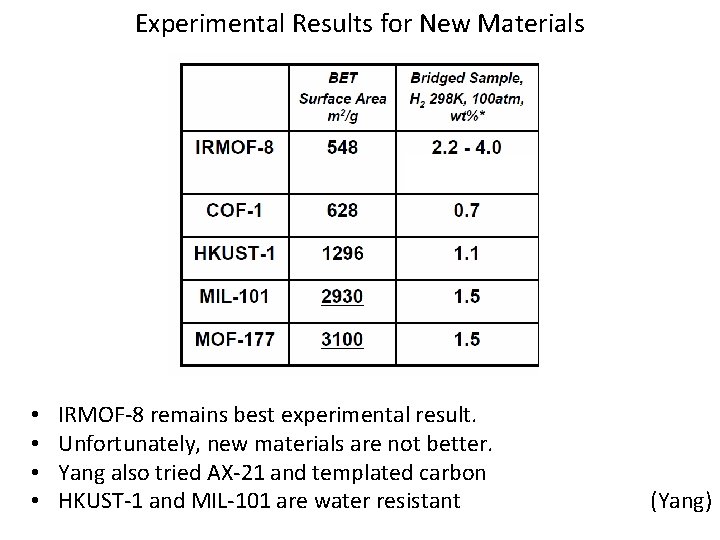
Experimental Results for New Materials • • IRMOF-8 remains best experimental result. Unfortunately, new materials are not better. Yang also tried AX-21 and templated carbon HKUST-1 and MIL-101 are water resistant (Yang)
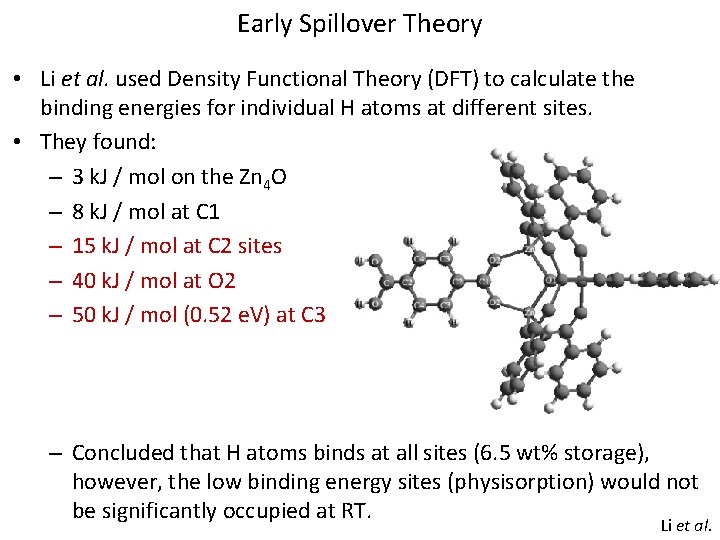
Early Spillover Theory • Li et al. used Density Functional Theory (DFT) to calculate the binding energies for individual H atoms at different sites. • They found: – 3 k. J / mol on the Zn 4 O – 8 k. J / mol at C 1 – 15 k. J / mol at C 2 sites – 40 k. J / mol at O 2 – 50 k. J / mol (0. 52 e. V) at C 3 – Concluded that H atoms binds at all sites (6. 5 wt% storage), however, the low binding energy sites (physisorption) would not be significantly occupied at RT. Li et al.
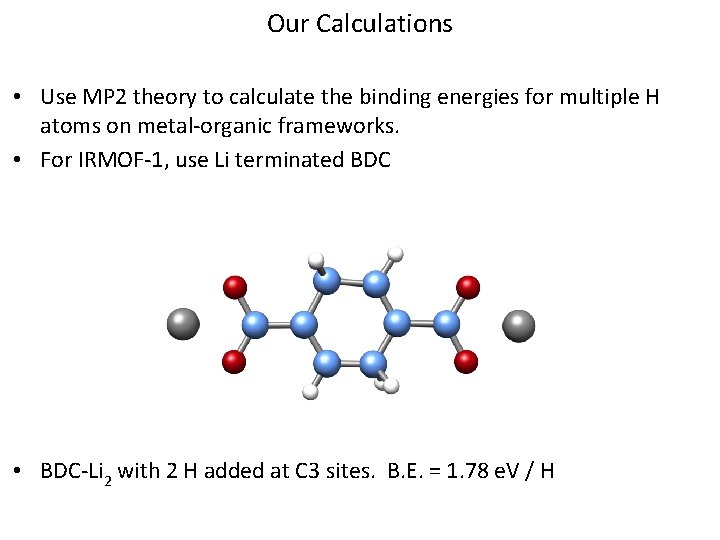
Our Calculations • Use MP 2 theory to calculate the binding energies for multiple H atoms on metal-organic frameworks. • For IRMOF-1, use Li terminated BDC • BDC-Li 2 with 2 H added at C 3 sites. B. E. = 1. 78 e. V / H
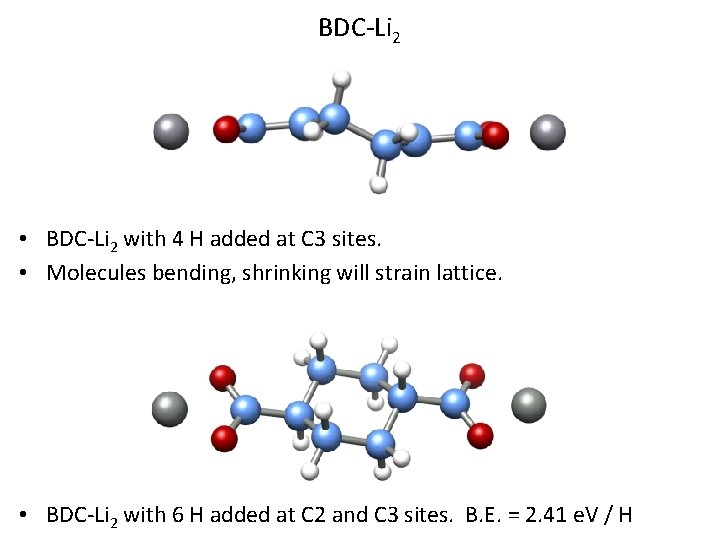
BDC-Li 2 • BDC-Li 2 with 4 H added at C 3 sites. • Molecules bending, shrinking will strain lattice. • BDC-Li 2 with 6 H added at C 2 and C 3 sites. B. E. = 2. 41 e. V / H
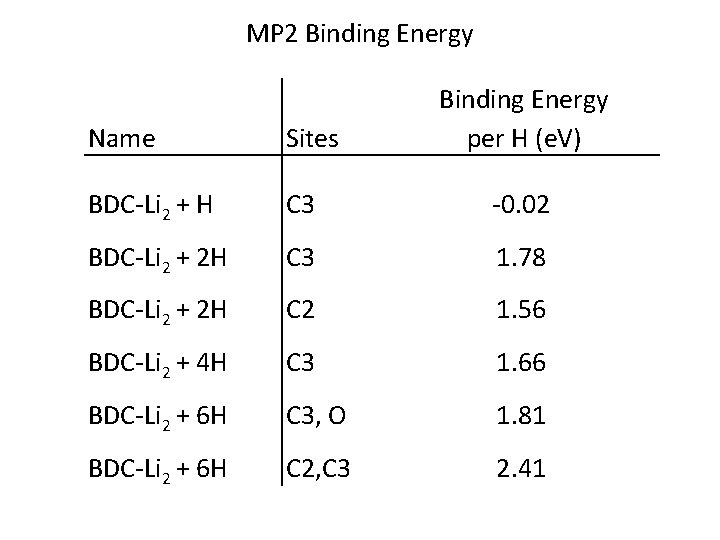
MP 2 Binding Energy per H (e. V) Name Sites BDC-Li 2 + H C 3 -0. 02 BDC-Li 2 + 2 H C 3 1. 78 BDC-Li 2 + 2 H C 2 1. 56 BDC-Li 2 + 4 H C 3 1. 66 BDC-Li 2 + 6 H C 3, O 1. 81 BDC-Li 2 + 6 H C 2, C 3 2. 41
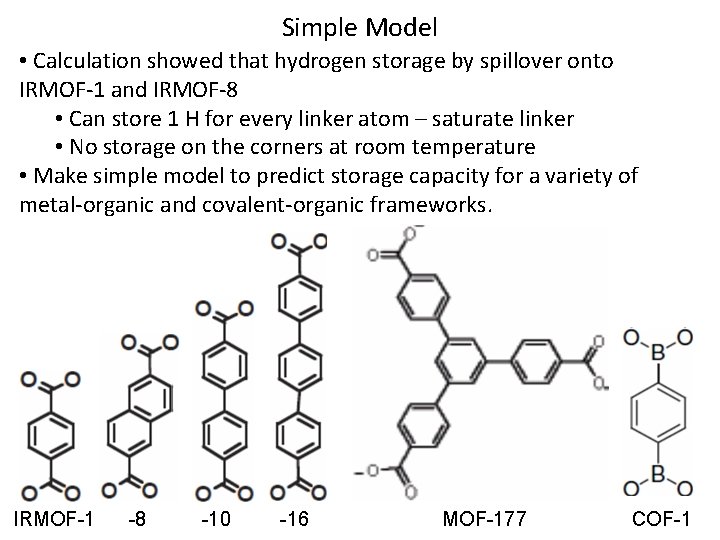
Simple Model • Calculation showed that hydrogen storage by spillover onto IRMOF-1 and IRMOF-8 • Can store 1 H for every linker atom – saturate linker • No storage on the corners at room temperature • Make simple model to predict storage capacity for a variety of metal-organic and covalent-organic frameworks. IRMOF-1 -8 -10 -16 MOF-177 COF-1
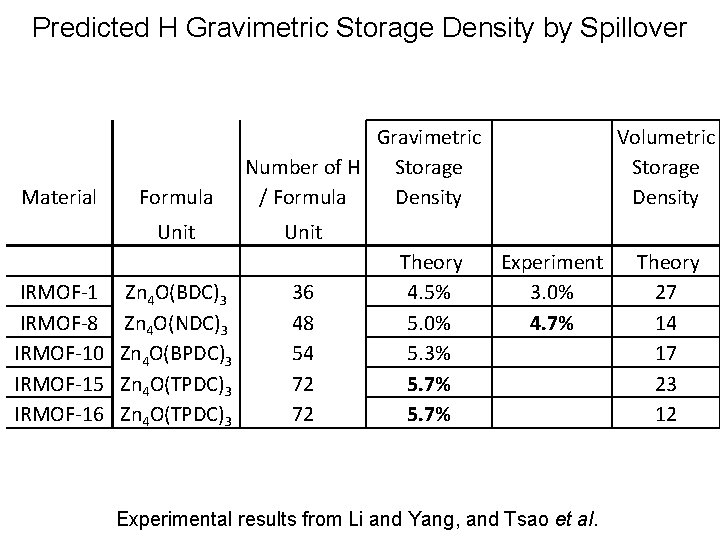
Predicted H Gravimetric Storage Density by Spillover Material Formula Unit IRMOF-1 IRMOF-8 IRMOF-10 IRMOF-15 IRMOF-16 Zn 4 O(BDC)3 Zn 4 O(NDC)3 Zn 4 O(BPDC)3 Zn 4 O(TPDC)3 Gravimetric Number of H Storage / Formula Density Volumetric Storage Density Unit 36 48 54 72 72 Theory 4. 5% 5. 0% 5. 3% 5. 7% Experiment 3. 0% 4. 7% Experimental results from Li and Yang, and Tsao et al. Theory 27 14 17 23 12
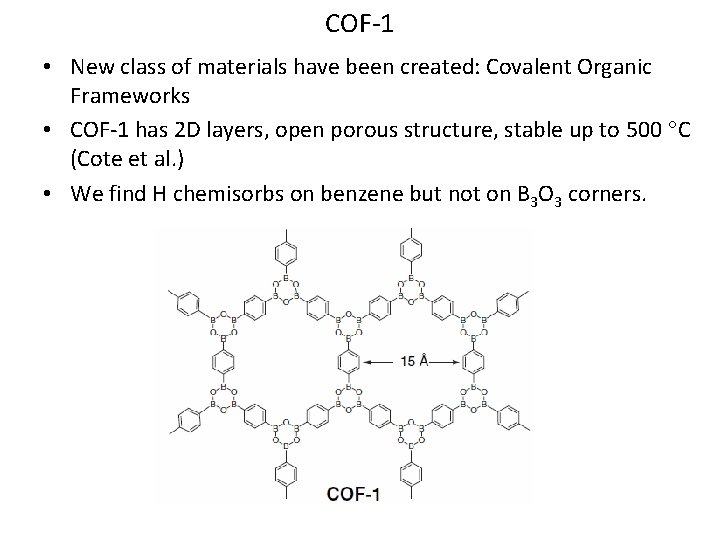
COF-1 • New class of materials have been created: Covalent Organic Frameworks • COF-1 has 2 D layers, open porous structure, stable up to 500 C (Cote et al. ) • We find H chemisorbs on benzene but not on B 3 O 3 corners.
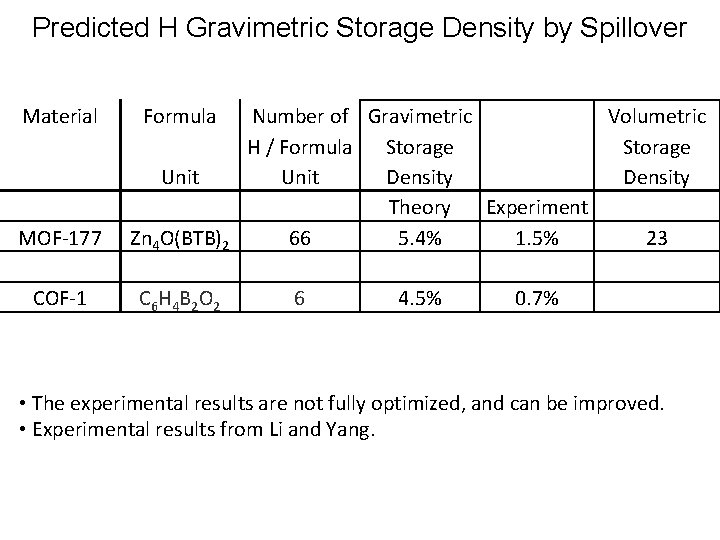
Predicted H Gravimetric Storage Density by Spillover Material Formula Unit MOF-177 Zn 4 O(BTB)2 COF-1 C 6 H 4 B 2 O 2 Number of Gravimetric Volumetric H / Formula Storage Unit Density Theory Experiment 66 5. 4% 1. 5% 23 6 4. 5% 0. 7% • The experimental results are not fully optimized, and can be improved. • Experimental results from Li and Yang.

Future Work • Design and evaluation of new materials for improved H storage. – Optimize gravimetric storage – Optimize volumetric storage – Detailed kinetic model including binding energies – Study strain and strength of crystal as H loaded – Tune binding energy to optimize kinetics – Design to improve durability – Explore modified graphene
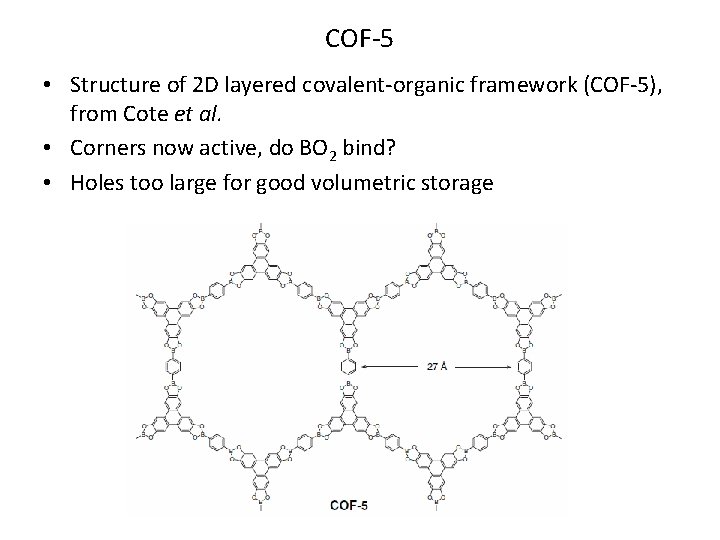
COF-5 • Structure of 2 D layered covalent-organic framework (COF-5), from Cote et al. • Corners now active, do BO 2 bind? • Holes too large for good volumetric storage

MIL-101 • MIL-101 is a new metal-organic framework material with lightweight Cu corners and open structure. Crystal structure of MIL-101 from Ferey

Predicted H Gravimetric Storage Density by Spillover Material Formula Unit MOF-177 HKUST-1 MIL-101 Zn 4 O(BTB)2 Cu 3(BTC)2 Cr 3 O(BDC)3 COF-1 COF-5 C 6 H 4 B 2 O 2 C 9 H 4 BO 2 IRMOF-993 Zn 4 O(ADC)3 Number of Gravimetric Volumetric H / Formula Storage Unit Density Theory Experiment 66 5. 4% 1. 5% 23 36 5. 6% 1. 1% 42 5. 9% 1. 5% 6 9 4. 5% 5. 5% 60 5. 3% 0. 7% 43 • The experimental results are not fully optimized, and can be improved. • The model results for HKUST-1, MIL-101 and COF-5 are preliminary. • Experimental results from Li and Yang.
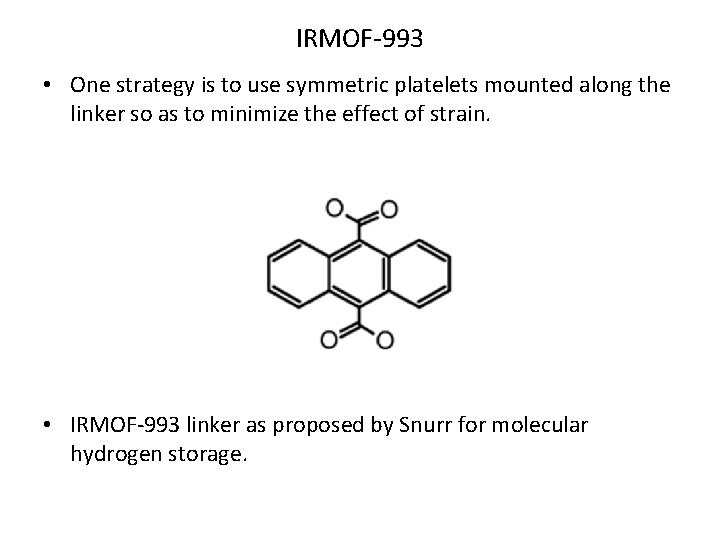
IRMOF-993 • One strategy is to use symmetric platelets mounted along the linker so as to minimize the effect of strain. • IRMOF-993 linker as proposed by Snurr for molecular hydrogen storage.
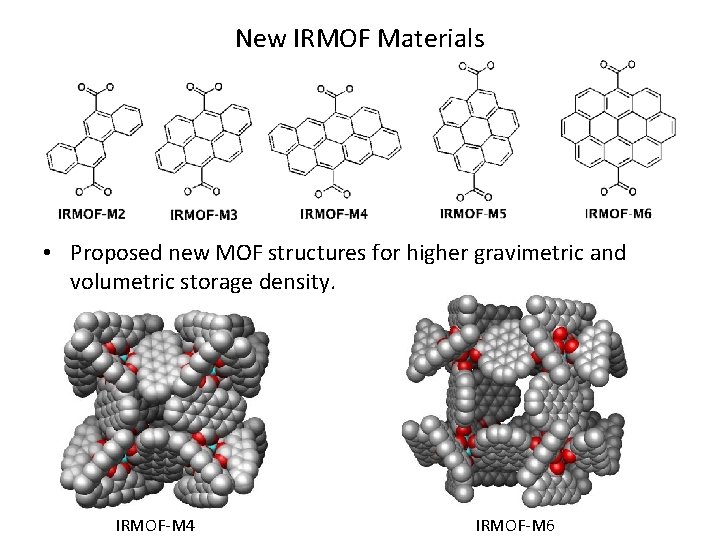
New IRMOF Materials • Proposed new MOF structures for higher gravimetric and volumetric storage density. IRMOF-M 4 IRMOF-M 6
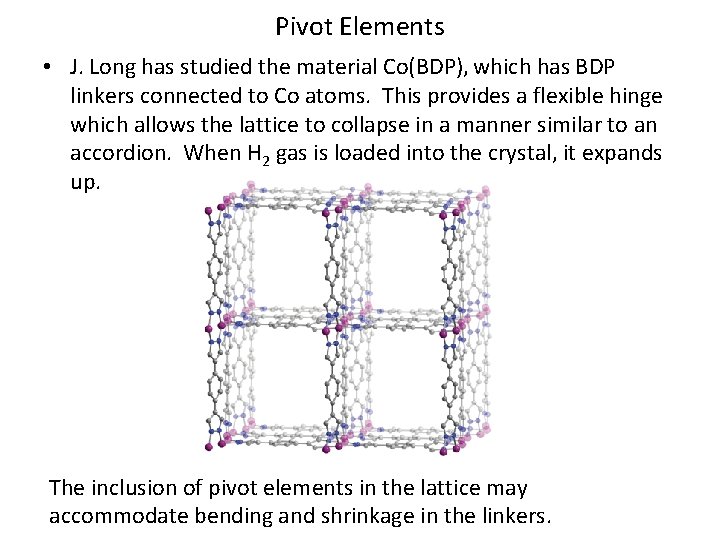
Pivot Elements • J. Long has studied the material Co(BDP), which has BDP linkers connected to Co atoms. This provides a flexible hinge which allows the lattice to collapse in a manner similar to an accordion. When H 2 gas is loaded into the crystal, it expands up. The inclusion of pivot elements in the lattice may accommodate bending and shrinkage in the linkers.
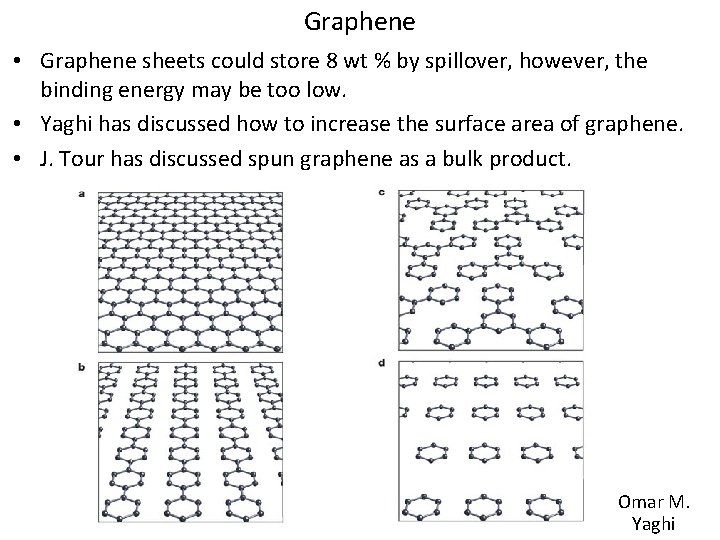
Graphene • Graphene sheets could store 8 wt % by spillover, however, the binding energy may be too low. • Yaghi has discussed how to increase the surface area of graphene. • J. Tour has discussed spun graphene as a bulk product. Omar M. Yaghi
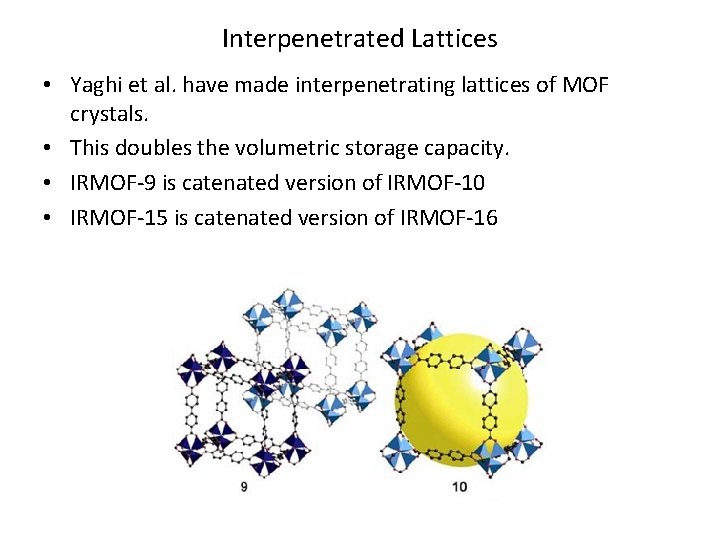
Interpenetrated Lattices • Yaghi et al. have made interpenetrating lattices of MOF crystals. • This doubles the volumetric storage capacity. • IRMOF-9 is catenated version of IRMOF-10 • IRMOF-15 is catenated version of IRMOF-16
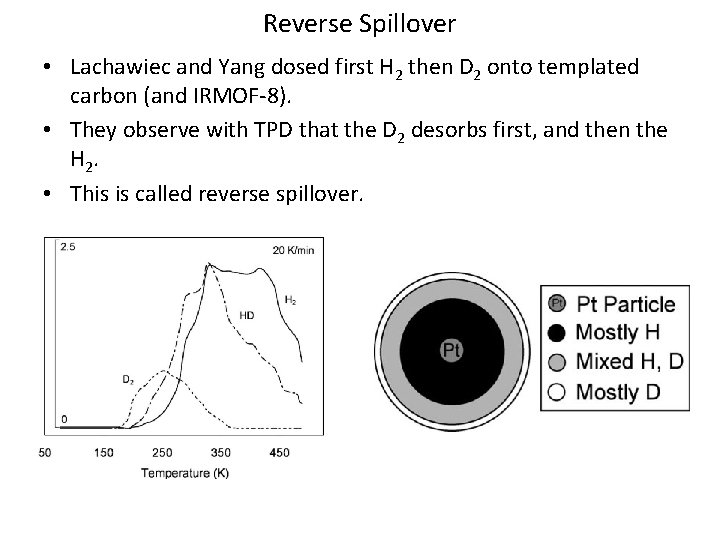
Reverse Spillover • Lachawiec and Yang dosed first H 2 then D 2 onto templated carbon (and IRMOF-8). • They observe with TPD that the D 2 desorbs first, and then the H 2. • This is called reverse spillover.

EXTRAS
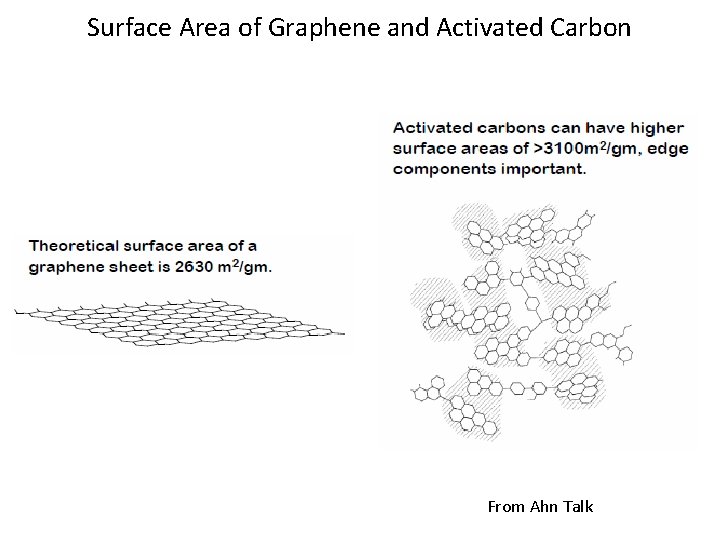
Surface Area of Graphene and Activated Carbon From Ahn Talk
Nature
- Slides: 33