Hydrogen Peroxide Detection Using Biogenic and Synthetic Mn

Hydrogen Peroxide Detection Using Biogenic and Synthetic Mn. O 2 Shu Feng, Jim Nurmi, Paul Tratynek Satya Chinni, Brad Tebo Department of Environmental and Biomolecular Systems OGI School of Science and Engineering Oregon Health & Science University ddd
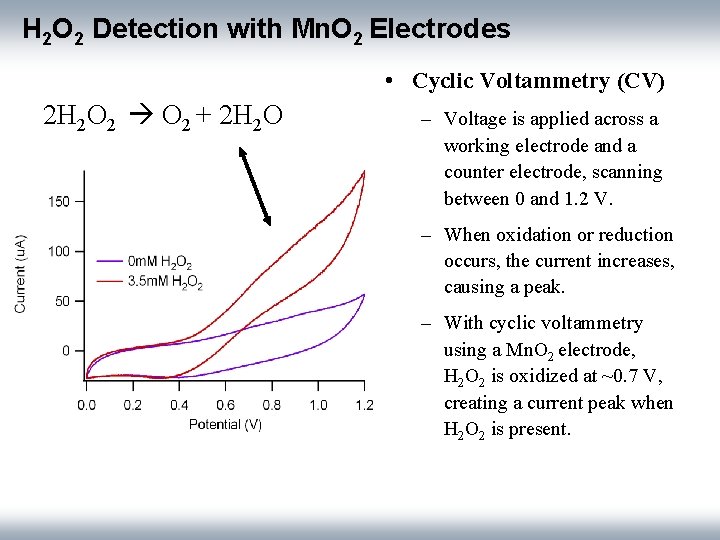
H 2 O 2 Detection with Mn. O 2 Electrodes • Cyclic Voltammetry (CV) 2 H 2 O 2 + 2 H 2 O – Voltage is applied across a working electrode and a counter electrode, scanning between 0 and 1. 2 V. – When oxidation or reduction occurs, the current increases, causing a peak. – With cyclic voltammetry using a Mn. O 2 electrode, H 2 O 2 is oxidized at ~0. 7 V, creating a current peak when H 2 O 2 is present.
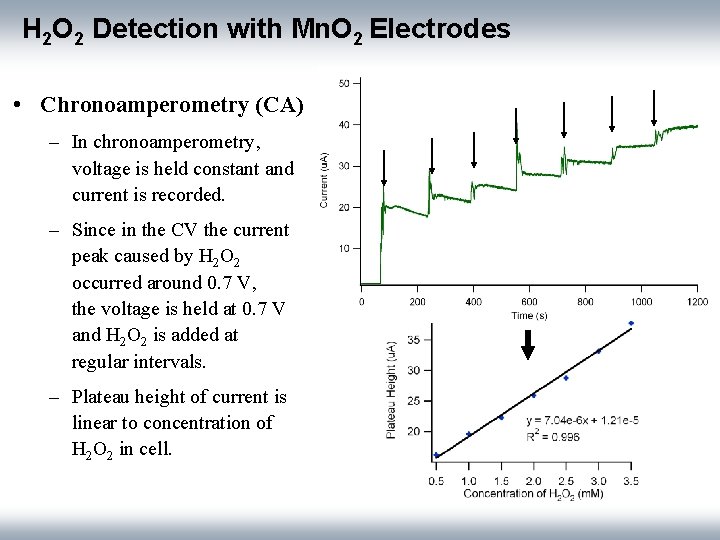
H 2 O 2 Detection with Mn. O 2 Electrodes • Chronoamperometry (CA) – In chronoamperometry, voltage is held constant and current is recorded. – Since in the CV the current peak caused by H 2 O 2 occurred around 0. 7 V, the voltage is held at 0. 7 V and H 2 O 2 is added at regular intervals. – Plateau height of current is linear to concentration of H 2 O 2 in cell.
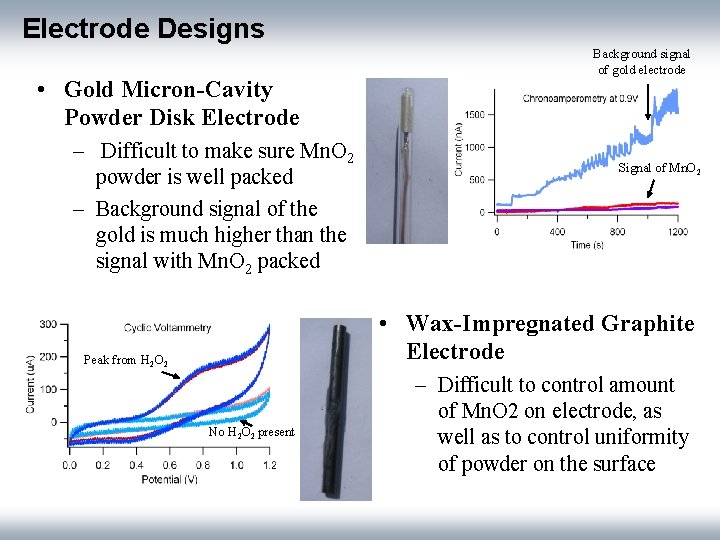
Electrode Designs • Gold Micron-Cavity Powder Disk Electrode – Difficult to make sure Mn. O 2 powder is well packed – Background signal of the gold is much higher than the signal with Mn. O 2 packed Background signal of gold electrode Signal of Mn. O 2 • Wax-Impregnated Graphite Electrode Peak from H 2 O 2 No H 2 O 2 present – Difficult to control amount of Mn. O 2 on electrode, as well as to control uniformity of powder on the surface

Electrode Designs • Glassy Carbon Electrode with Spin Coated Mn. O 2 – Excess Mn. O 2 solution is pipetted onto the electrode surface. – By controlling spin speed, an even coat can be achieved. – However, after a long period of time in solution, Mn. O 2 eventually falls off. Electrode Head Spin Coating Apparatus Close-up views of the electrode surface Uneven Mn. O 2 surface after time in solution
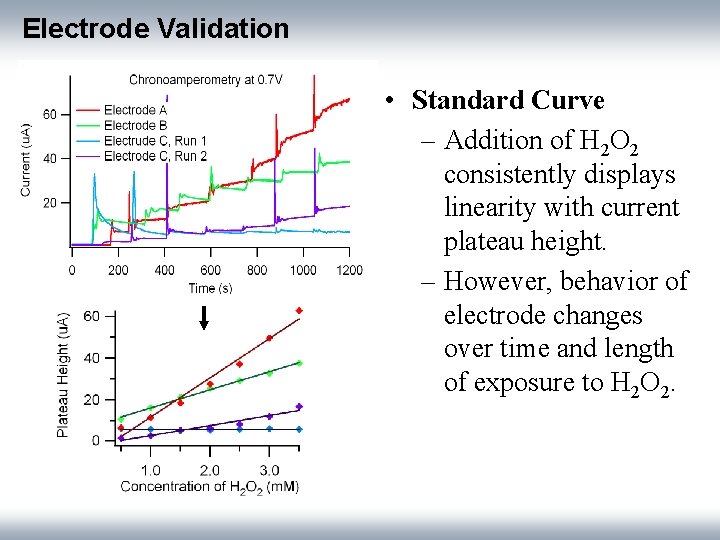
Electrode Validation • Standard Curve – Addition of H 2 O 2 consistently displays linearity with current plateau height. – However, behavior of electrode changes over time and length of exposure to H 2 O 2.
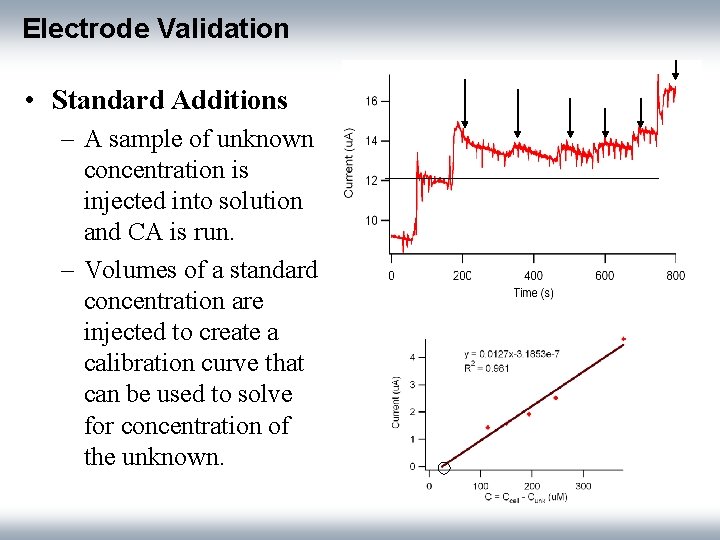
Electrode Validation • Standard Additions – A sample of unknown concentration is injected into solution and CA is run. – Volumes of a standard concentration are injected to create a calibration curve that can be used to solve for concentration of the unknown.

Conclusions • Gold micron-cavity powder disk electrode has a high background signal from gold. • Amount of Mn. O 2 is difficult to control for on the wax impregnated graphite electrode. • Spin coated carbon electrode produces a evenly coated electrode, but produces a varied response. – Standard addition allows these electrodes to be used as a sensor. • Biogenic and synthetic Mn. O 2 electrodes both have H 2 O 2 sensing capabilities, but the differences between them need to be further studied.

Goals For Next Summer’s Intern… (or me in the next week) • Determine effects of aging of the electrode and Mn. O 2 powder • Further characterize electrode behavior in different electrolyte conditions (p. H, concentration, etc) • More biogenic vs. synthetic Mn. O 2 comparison • Resolve problems with using sensor in seawater Acknowledgments Jim Nurmi Paul Tratynek Satya Chinni Brad Tebo Vanessa Green EBS Department
- Slides: 9