Hydrogen Fuel Cell Trends in the use of












- Slides: 23

Hydrogen Fuel Cell

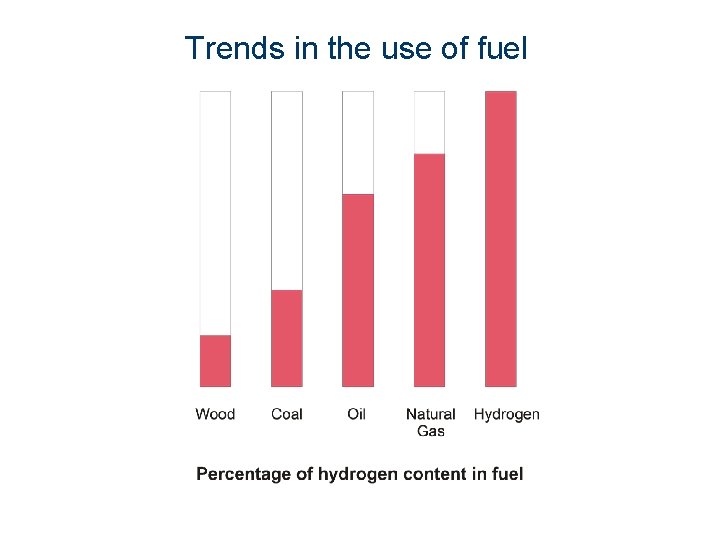
Trends in the use of fuel

19 th century: steam engine 20 th century: internal combustion engine 21 st century: fuel cells

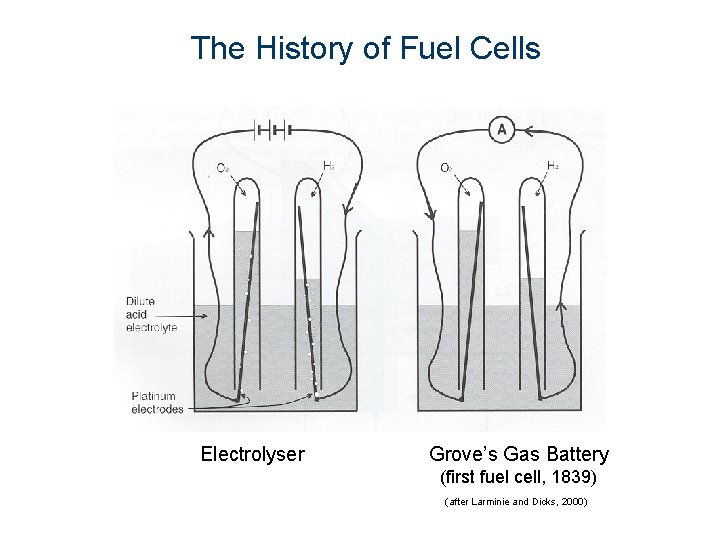
The History of Fuel Cells Electrolyser Grove’s Gas Battery (first fuel cell, 1839) (after Larminie and Dicks, 2000)

Photo courtesy of University of Cambridge Bacon’s laboratory in 1955

Photo courtesy of NASA Space Shuttle fuel cell

Applications for Fuel Cells Transportation vehicles Photo courtesy of Daimler. Chrysler NECAR 5

Applications. Distributed for Fuel Cells power stations Photo courtesy of Ballard Power Systems 250 k. W distributed cogeneration power plant

Applications for Fuel Cells Home power Photo courtesy of Plug Power 7 k. W home cogeneration power plant

Portable power Applications for Fuel Cells 50 W portable fuel cell with metal hydride storage

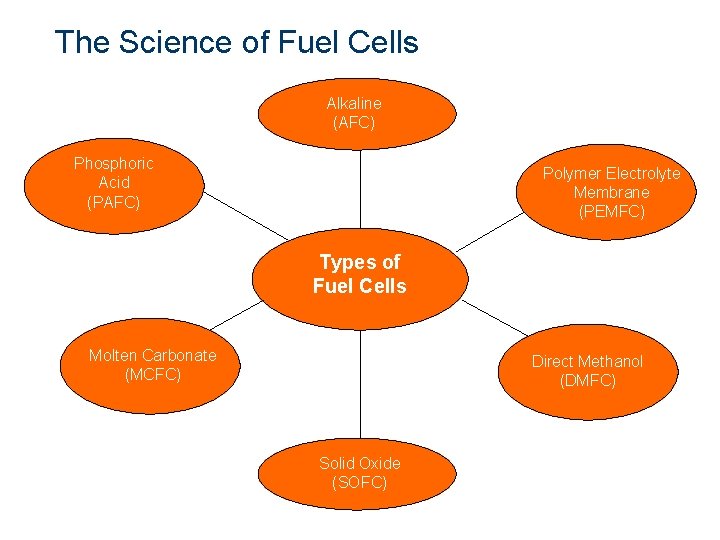
The Science of Fuel Cells Alkaline (AFC) Polymer Electrolyte Membrane (PEMFC) Phosphoric Acid (PAFC) Types of Fuel Cells Molten Carbonate (MCFC) Direct. Methanol Direct (DMFC) Solid. Oxide Solid (SOFC)

PEM Fuel Cell Electrochemical Reactions Anode: H 2 2 H+ + 2 e- (oxidation) Cathode: 1/2 O 2 + 2 e- + 2 H+ H 2 O (l) (reduction) Overall Reaction: H 2 + 1/2 02 H 2 O (l) ΔH = - 285. 8 k. J/mole

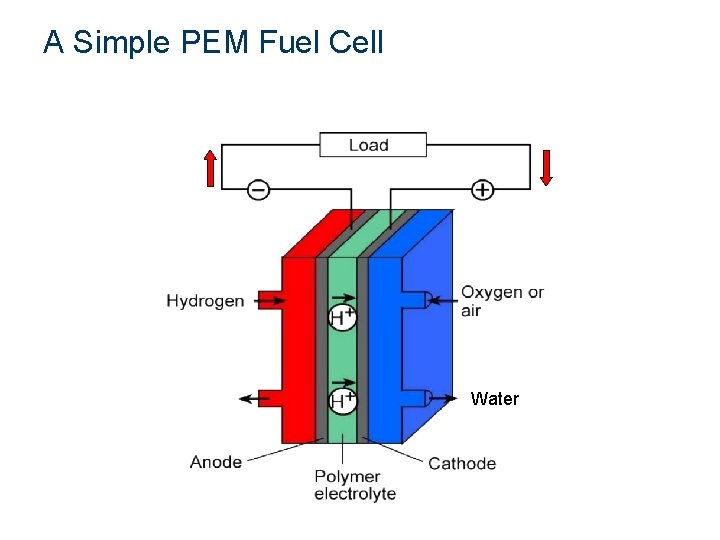
A Simple PEM Fuel Cell Hydrogen + Oxygen Electricity + Water

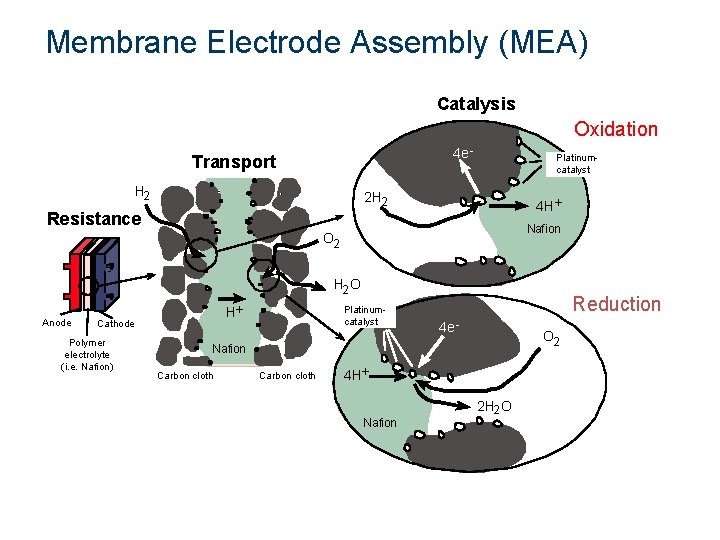
Membrane Electrode Assembly (MEA) Catalysis Oxidation 4 e - Transport H 2 Platinumcatalyst 2 H 2 4 H+ Resistance Nafion O 2 H 2 O Anode H+ Cathode Polymer electrolyte (i. e. Nafion) Platinum. Kcatalyst Reduction 4 e - O 2 Nafion Carbon cloth 4 H+ Nafion 2 H 2 O 2

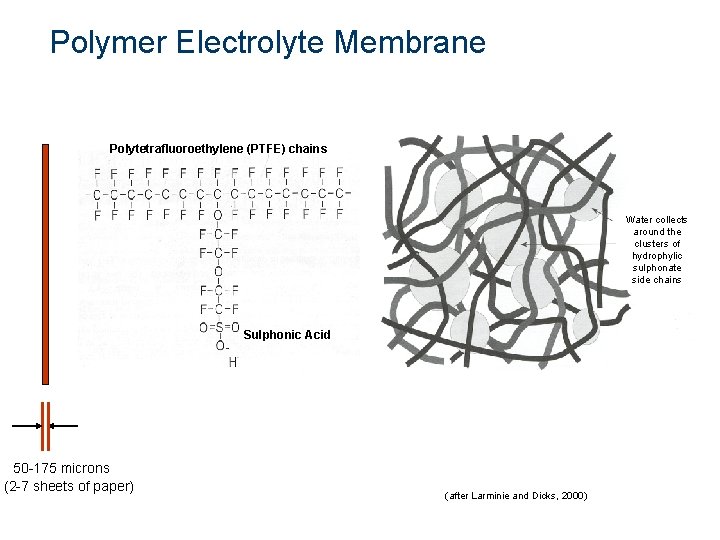
Polymer Electrolyte Membrane Polytetrafluoroethylene (PTFE) chains Water collects around the clusters of hydrophylic sulphonate side chains Sulphonic Acid 50 -175 microns (2 -7 sheets of paper) (after Larminie and Dicks, 2000)

Thermodynamics of PEM Fuel Cells Change in enthalpy (ΔH) = - 285, 800 J/mole Gibb’s free energy (ΔG) = ΔH - TΔS ΔG at 25° C: = - 285, 800 J - (298 K)(-163. 2 J/K) = - 237, 200 J Ideal cell voltage (Δ E) ΔE at 25º C = - ΔG/(n. F) = - [-237, 200 J/((2)(96, 487 J/V))] = 1. 23 V ΔG at operating temperature (80º C): = - 285, 800 J - (353 K)(163. 2 J/K) = - 228, 200 J ΔE at 80º C = - [-228, 200 J/((2)(96, 487 J/V))] = 1. 18 V

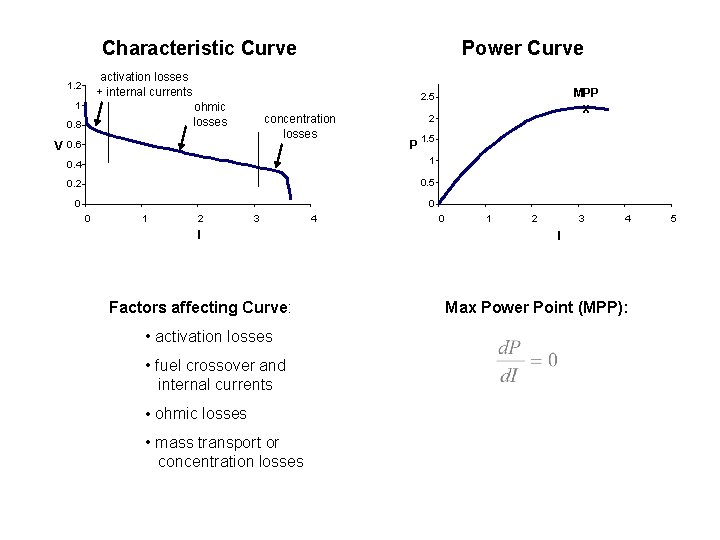
Characteristic Curve Power Curve activation losses + internal currents 1. 2 1 0. 8 concentration losses V 0. 6 P 1. 5 1 0. 2 0. 5 0 0 1 2 3 I Factors affecting Curve: • activation losses • fuel crossover and internal currents • ohmic losses • mass transport or concentration losses 4 x 2 0. 4 0 MPP 2. 5 ohmic losses 0 1 2 3 4 I Max Power Point (MPP): 5

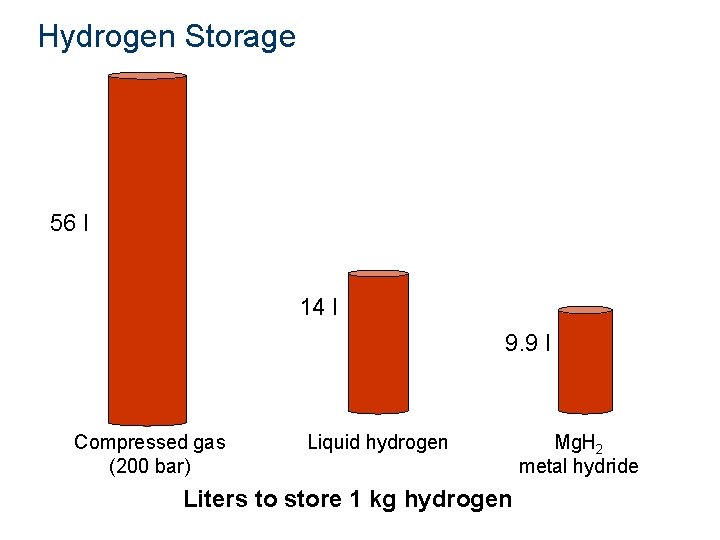
Hydrogen Storage 56 l 14 l 9. 9 l Compressed gas (200 bar) Liquid hydrogen Liters to store 1 kg hydrogen Mg. H 2 metal hydride

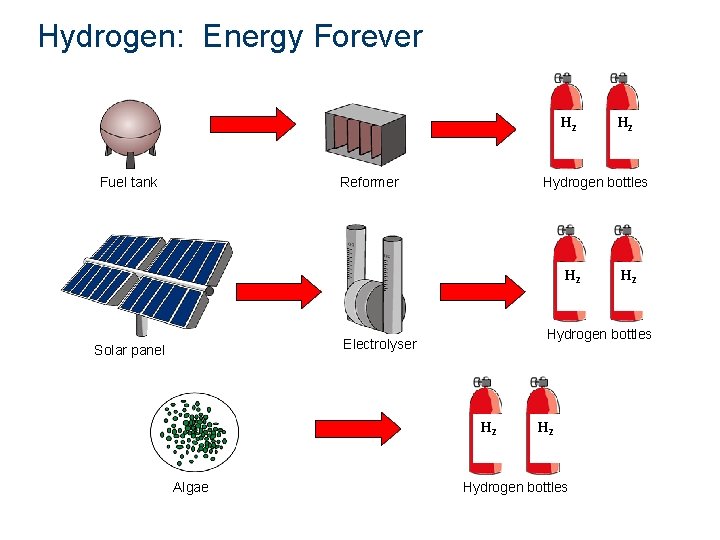
Hydrogen: Energy Forever H 2 Fuel tank Reformer Hydrogen bottles H 2 Algae H 2 Hydrogen bottles Electrolyser Solar panel H 2 Hydrogen bottles

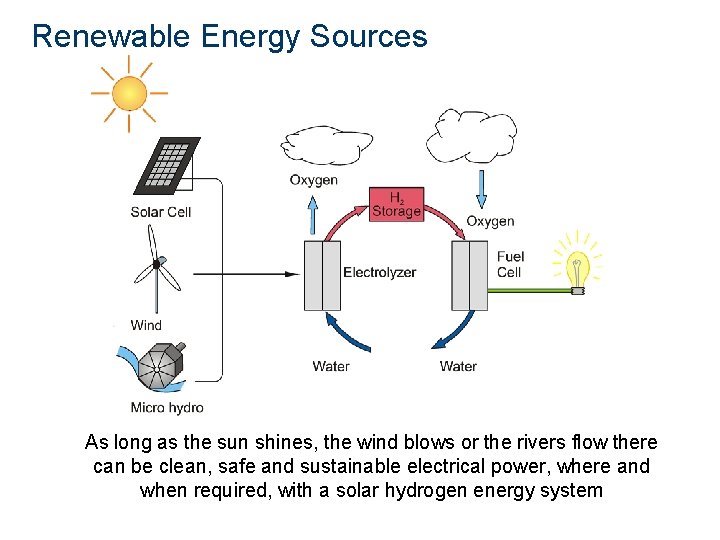
Renewable Energy Sources As long as the sun shines, the wind blows or the rivers flow there can be clean, safe and sustainable electrical power, where and when required, with a solar hydrogen energy system

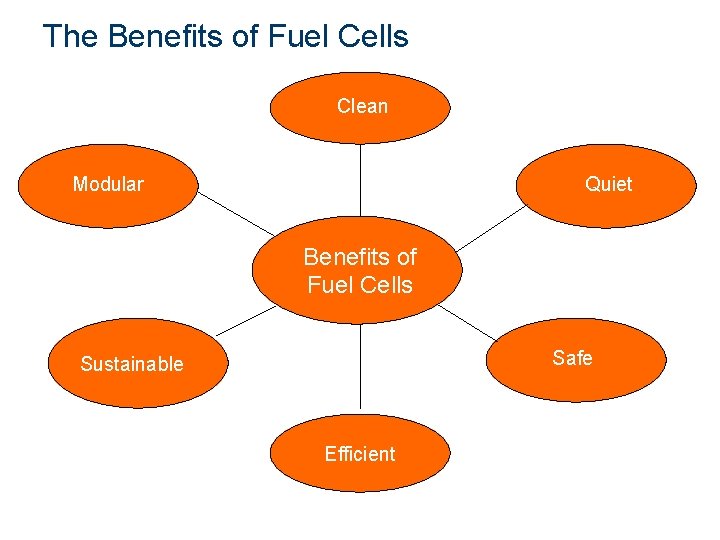
The Benefits of Fuel Cells Clean Modular Quiet Benefits of Fuel Cells Safe Sustainable Efficient

Our Fragile Planet. We have the responsibility to mind the planet, so that the extraordinary natural beauty of the Earth is preserved for generations to come. Heliocentris: Science education through fuel cells 22 Photo courtesy of NASA

Presentation courtesy of Heliocentris