Hydroelectrolytic disturbances Homeostasis Body fluids are in constant

Hydroelectrolytic disturbances

Homeostasis � Body fluids are in constant motion transporting nutrients, electrolytes, and oxygen to cells while carrying away waste products � The maintenance of normal volume and normal composition of the extracellular fluid is vital to life � Homeostasis: the various physiologic arrangements which serve to restore the normal state, once it has been disturbed ◦ ◦ Fluid balance Electrolyte balance Osmotic balance Acid-base balance

Distribution of body fluids total body water ═ 0, 6/0, 5 x(kg body weight) § intracellular � extracellular -interstitial (lymph) and transcellular (third-space, water distributed in the digestive tract, pleural, peritoneal, spinal fluid) -intravascular Intracellular and extracellular water move back and forth through the cell membrane (osmotic change). The intracellular water in adult males averages 57% of the total body water.
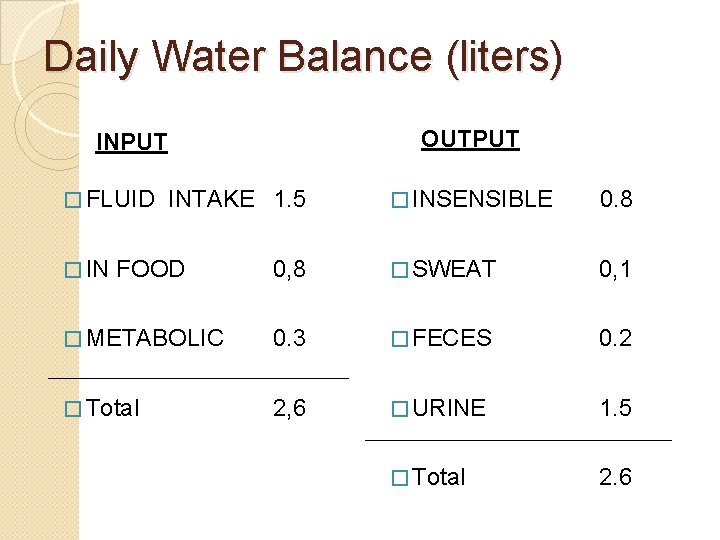
Daily Water Balance (liters) OUTPUT INPUT � FLUID INTAKE 1. 5 � INSENSIBLE 0. 8 � IN FOOD 0, 8 � SWEAT � METABOLIC 0. 3 � FECES 0. 2 � Total 2, 6 � URINE 1. 5 � Total 2. 6 0, 1

Electrolytes (m. Eq) �Chemicals dissolved in the body fluid �Regulated by intake, output, acid-base balance, hormones, and cell integrity �Sodium -major extracellular electrolyte -controls and regulate water balance • Potassium -major intracellular electrolyte -helps maintain intracellular water balance -transmit nerve impulses to muscles
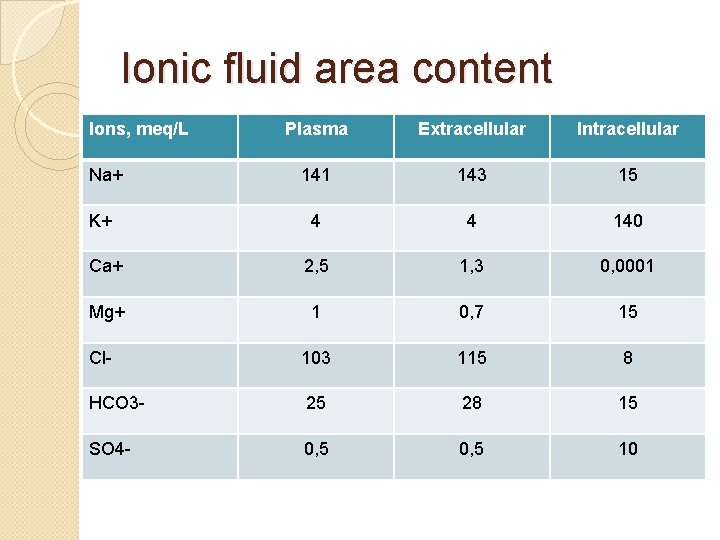
Ionic fluid area content Ions, meq/L Plasma Extracellular Intracellular 141 143 15 4 4 140 Ca+ 2, 5 1, 3 0, 0001 Mg+ 1 0, 7 15 Cl- 103 115 8 HCO 3 - 25 28 15 SO 4 - 0, 5 10 Na+ K+
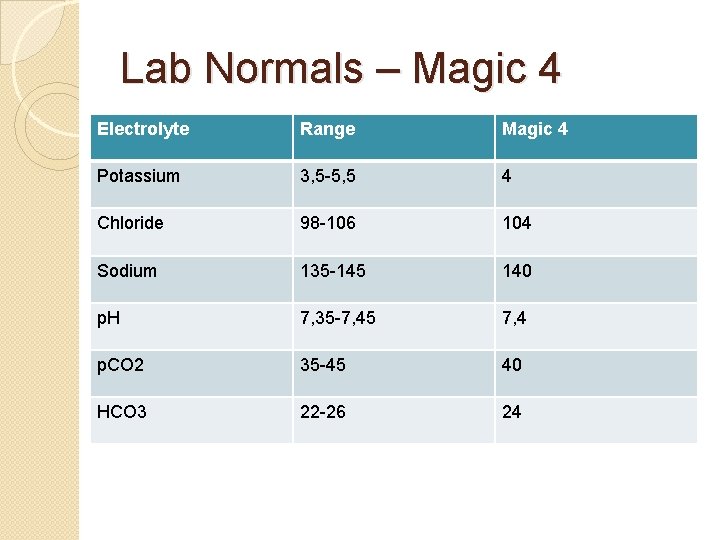
Lab Normals – Magic 4 Electrolyte Range Magic 4 Potassium 3, 5 -5, 5 4 Chloride 98 -106 104 Sodium 135 -145 140 p. H 7, 35 -7, 45 7, 4 p. CO 2 35 -45 40 HCO 3 22 -26 24

Water distribution of water and electrolytes between compartments is dependent on: �Osmolarity �Tonicity �Electroneutrality �Starling balance

Osmolarity and osmolality �Indicates the water balance of the body �Serum osmo is 285 -295 m. Osm/kg -high is water deficit (concentrated) -low is water excess (dilute) �Urine osmo is 50 -1200 m. Osm/kg �Together are used to determine what is causing a sodium imbalance �Urine osmo/serum osmo=1/3

Regulation of fluid and electrolyte movement �Filtration – the fluid passes through the membrane (hydrostatic pressure, oedema) �Diffusion is the net movement of molecules from a region of high concentration to a region of low concentration �Osmosis is the way water moves between two spaces, solutes create the concentration gradients that promote osmosis (Osmolarity of extracellular space depends on the sodium concentration in plasma and is equivalent to the osmolarity of the intracellular space which is given by the potassium. ) � Active transport (molecules have to move against to gradient, so they requires some energy and pump, Na/K/ATP)

Ionic balance provide: �the resting membrane potential, generated by intracellular and extracellular ion concentrations ratio �neuromuscular excitability

Regulation of water balance Kidneys (JG cells) Kidneys (adrenal cortex) Hypothalamus Heart • Sense low Na, low volume • Release renin • Converts angiotensinogen to angiotensin I which converts to angiotensin II • Stimulates release of aldosterone • Senses low serum osmo or low Na • Releases aldosterone • Reabsorbs Na into the blood, increases K excretion in the urine • Increases serum osmo • Senses high serum osmo or high Na • Stimulates thirst • Triggers release of ADH (vasopresine) from posterior pituitary • Retains water in the blood • Concentrates urine • Mildly constricts blood vessels • Decreases serum osmo • Senses high volume through stretch receptors in the right atrium • Secrets ANP, BNP • Inhibits ADH • Increases Na excretion through the urine • Dilates blood vessels • Decreases serum osmo

Fluid disorders Are represented by: �Volume and Osmolarity disorders �we refer primarily to extracellular space Ø Water lack or excess Ø Sodium lack or excess Intracellular space is determined by the volume and osmolarity of extracellular space.

Fluid disorders The mechanisms involved in the pathology development of fluid and electrolyte are: �ingestion disorders �eliminate disorders �disorders of control mechanisms

Primary disorders �Water deficiency �Water excess �Sodium deficiency

Disturbances of fluid homeostasis Diagnosis: �Physical signs: skin turgor, oedema, mucous membranes, neck veins, puls, liver, level of consciousness, capillary refill, fontanel (children) �Clinical sings: blood pressure; heart rate; (respiratory rate); body temperature; CVP; urine output; serum and urine Na, osmolarity; Htk; serum total protein

Fluid volume deficit (no water, no salt, or both) �No water (hypertonic) -profuse sweating, hyperventilation, fevers, diarrhea, renal failure, DI �No salt (hypotonic) -water intoxication, chronic illness, malnutrition, renal failure �Both (isotonic) -NPO, poor intake, hemorrhage

Dehydration Isotonic Hypertonic Se Na Se osmolarity Hb Htk Blood volume Thirst n n + + - mod. increase + + - Hypotonic + + - increased no
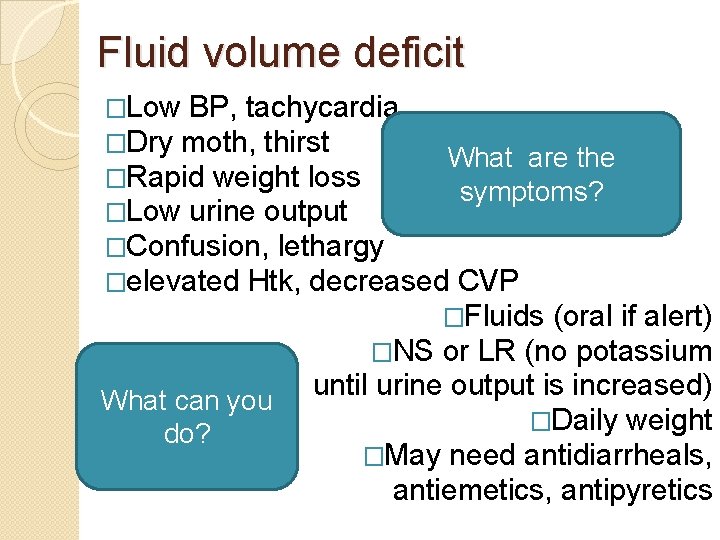
Fluid volume deficit �Low BP, tachycardia �Dry moth, thirst What are the �Rapid weight loss symptoms? �Low urine output �Confusion, lethargy �elevated Htk, decreased CVP �Fluids (oral if alert) �NS or LR (no potassium What can you do? until urine output is increased) �Daily weight �May need antidiarrheals, antiemetics, antipyretics

Fluid volume excess �Increased sodium and water �Causes: -hypervolemia (isotonic) • Too much IV fluid, kidney failure, corticosteroids -water intoxication (hypotonic) � SIADH, IV fluids, psych problems, wound irrigation -too much sodium intake (hypertonic) � Too much salt, 3%saline IV, too much Na. HCO 3

Fluid volume excess � Rapid weight gain What are the symptoms? � Edema � High BP, bounding pulses � May have increased urine output � Neck vein distention, crackles, dyspnea � Low Htc, low blood urea, high Na, low osmo �Diuretics �Fluid restriction (no IV fluids) What can you do? �Sodium restriction �Daily weights

Sodium �Major cation of ECF �Regulated by kidneys, ADH, aldosterone �GI tract absorbs sodium from food �Imbalances are typically associated with fluid volume problems �Foods high in sodium – processed meats, condiments, dairy

Hypernatremia � Water loss or excess sodium � Decreased Na excretion – renal failure, corticosteroids � Increased Na intake – eating too much salt, too much sodium in IV fluids � Increased water loss – fever, infection, sweating, diarrhea � over nasogastric feeding, hyperosmolar coma, dehydration secondary to fever or ambient temperature increase � diabetes insipidus � excessive administration of hypertonic solution and Sodium bicarbonate “You are fried” F Fever (low grade, flushed skin) R Restless (irritable) I Increased fluid retention and BP E Edema (peripheral) D Decreased urine output, dry mouth

Hypernatremia What can you do? Reduce sodium slowly! �Treat the underlying cause �Diuretics �Sodium restriction �Seizure precautions

Hyponatremia � Water excess or loss of sodium � Dilution – polydipsia, freshwater drowning, SIADH � Increased excretion – sweating, diuretics, GI wound drainage, renal disease � Decreased intake – NPO, law salt diet, severe vomiting/diarrhea � Symptoms: -confusion, headaches -seizures (can progress to coma) -abd. cramps

Hyponatremia What can you do? Replace sodium slowly! � 3%normal saline �If caused by fluid excess, will need fluid restriction �Usually can not be fixed by adding sodium to the diet

Sodium deficiency-hyponatremia � Hyponatremia with euvolaemia is Syndrome of inappropriate antidiuretic hormone secretion (SIADH) � The diagnostic criteria are: - hyponatremia with hypo-osmolarity - permanent removal of sodium (elimination is proportional to ingestion. ) - urinary hyperosmolarity compared with plasma - absence of other causes to decrease the dilution capacity of the kidney - absence of hyponatraemia and hypo osmolarity after restriction of water

Potassium �Major cation of Intracellular Fluid, helps maintain Intracellular Fluid volume �Key role in the resting membrane potential and action potential of neurons and muscle fibers �the inter-compartment gradient is maintained by the activity of Na/K/ATP-ase, that pumps Sodium out of cells while pumping Potassium into cells, both against their concentration gradients �K+ level is controlled by aldosterone (increased Na and decreased K by reabsorbtion or not in kidney). Primary route of loss – kidney

Hyperkalemia �Causes ◦ Impaired excretion �Renal failure �Use of salt or potassium supplements �Receiving old blood �Hypoxia �Exercise (catabolic state) �Drugs (potassium sparing diuretics) ◦ Shifts of K out of cells �Tissue breakdown (Cell destruction) �acidosis, insulin deficiency

Hyperkalemia ”MURDER” M muscle weakness U urine, oliguria, anuria R respiratory distress D decreased cardiac contractility E ECG changes R reflexes, hyperreflexia, or areflexia

Hyperkalemia Therapy: �Cardiac monitor �Kayexalate ◦ Movement of extracellular K into intracellular compartment �Insulin (+glucose) �Sodium bicarbonate �Calcium gluconate ◦ Removal K from the body �Diuretics �Dialysis if severe -stop potassium in IV fluids -avoid foods high in potassium

Hypokalemia �Causes: ◦ Decreased K intake ◦ Increased excretion: vomiting, diarrhea, renal losses, diuretics ◦ Shifts of K into cells: drugs (Insulin, β 2 -adrenergic agonists, Theophylline, Caffeine), alkalosis
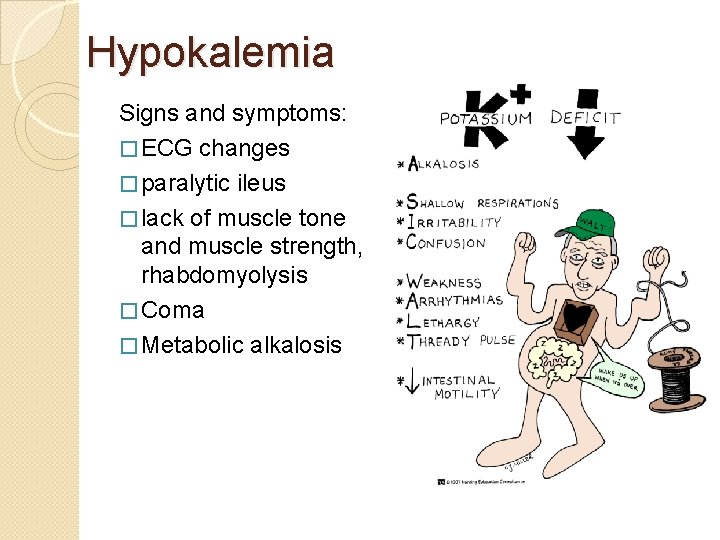
Hypokalemia Signs and symptoms: � ECG changes � paralytic ileus � lack of muscle tone and muscle strength, rhabdomyolysis � Coma � Metabolic alkalosis

Hypokalemia What can you do? � Foods high in potassium � Watch for Digoxin toxicity � Potassium IV (only if good urine output) � Spironolactone � Treat constipation � Potassium administration • Must have urine output • Never give IV push • Must be on cardiac monitor • Assess IV site (prefer CVC) • Always dilute and give no more than 20 m. Eq, no faster than 1 hr • Max concentration in IV fluids is 40 m. Eq/l Cardiac monitor
- Slides: 34