Hydrodynamic instabilities of autocatalytic reaction fronts A De

Hydrodynamic instabilities of autocatalytic reaction fronts: A. De Wit Unité de Chimie Physique non Linéaire Université Libre de Bruxelles, Belgium 1

Scientific questions n n n Can chemical reactions be at the origin of hydrodynamic instabilities (and not merely be passively advected by the flow) ? What are the properties of the new patterns that can then arise ? Influence on transport and yield of reaction ? 2

Outline I. Vertical set-ups • • Convective deformation of chemical fronts Experiments in Hele-Shaw cells Model of hydrodynamic instabilities of fronts Rayleigh-Bénard, Rayleigh-Taylor and doublediffusive instabilities • Reactive vs non-reactive system II. Horizontal set-ups • Marangoni vs buoyancy-driven flows 3

Buoyancy-driven instability of a chemical front in a vertical set-up Heavy A Light B Hydrodynamic Rayleigh. Taylor instability of autocatalytic IAA fronts ascending in the gravity field in a capillary tube because reactant A is heavier than the product B Bazsa and Epstein, 1985; Nagypal, Bazsa and Epstein, 1986 Pojman, Epstein, Mc. Manus and Showalter, 1991 Heavy A: stable descending front Light B: unstable ascending front (Courtesy D. Salin) 4
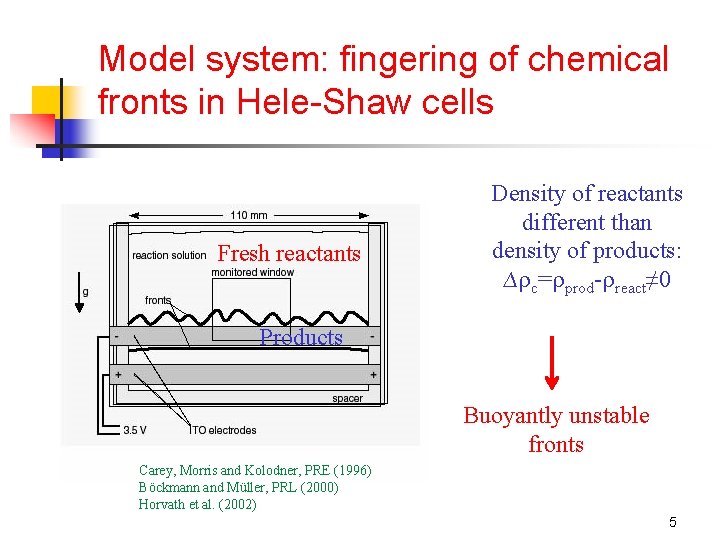
Model system: fingering of chemical fronts in Hele-Shaw cells Fresh reactants Density of reactants different than density of products: ∆ c= prod- react≠ 0 Products Buoyantly unstable fronts Carey, Morris and Kolodner, PRE (1996) Böckmann and Müller, PRL (2000) Horvath et al. (2002) 5
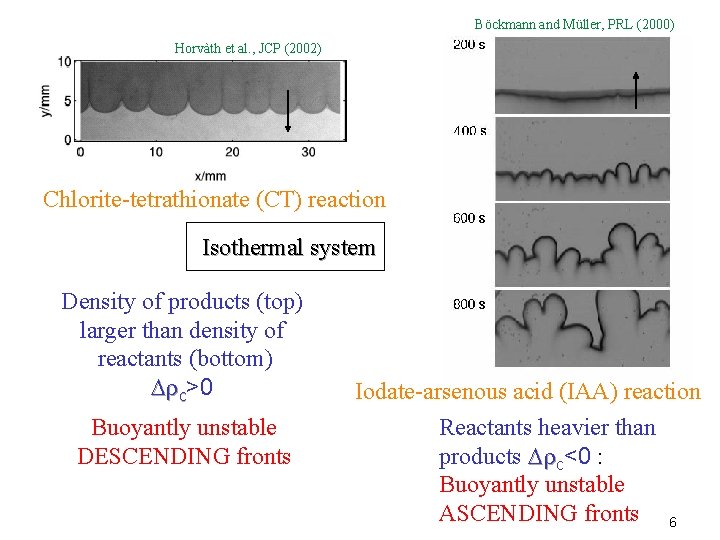
Böckmann and Müller, PRL (2000) Horvàth et al. , JCP (2002) Chlorite-tetrathionate (CT) reaction Isothermal system Density of products (top) larger than density of reactants (bottom) D c>0 Buoyantly unstable DESCENDING fronts Iodate-arsenous acid (IAA) reaction Reactants heavier than products D c<0 : Buoyantly unstable ASCENDING fronts 6

J. A. Pojman and I. R. Epstein, “Convective effects on chemical waves: 1. Mechanisms and stability criteria”, J. Phys. Chem. , 94, 4966 (1990) = o[1 -a. T(T-To)+S ac i(Ci-Cio)] Across a chemical front: D = products- reactants=D c+D T D c<0: reactants are heavier than products (IAA) D c>0: products are heavier than reactants (CT) D T<0: exothermic reaction, products are hotter than reactants 7
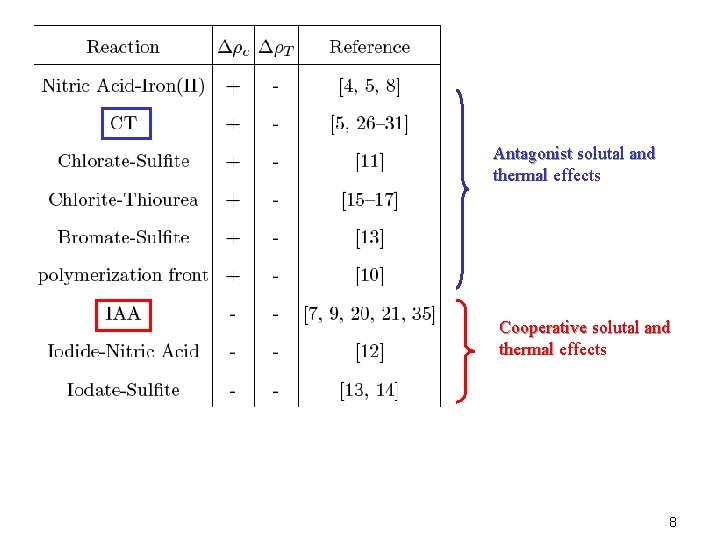
Antagonist solutal and thermal effects Cooperative solutal and thermal effects 8

Questions • Which kind of hydrodynamic instabilities can the competition or cooperative effects between solutal and thermal density effects generate across a chemical front ? • Are there new instabilities possible with regard to the non reactive case ? 9

- (C, T)g Theoretical model with Le=DT/D Rayleigh numbers Lewis number 10

Linear stability analysis of pure hydrodynamic instabilities F(C) = 0 Base state : linear concentration and temperature gradients with a corresponding density gradient 11
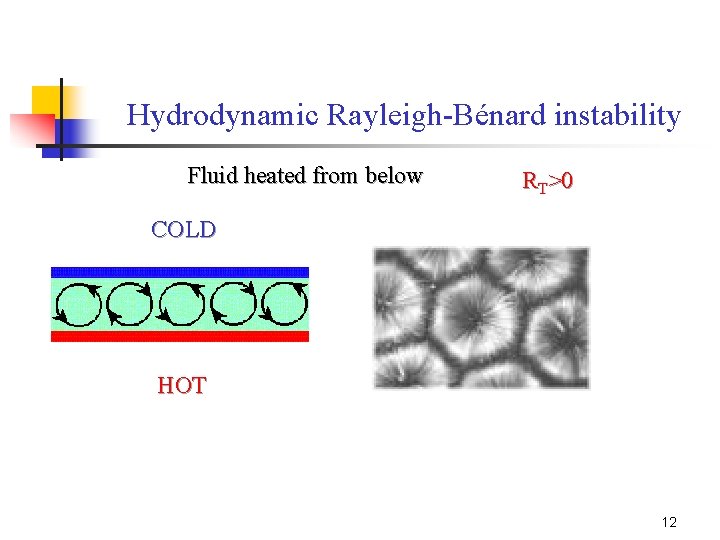
Hydrodynamic Rayleigh-Bénard instability Fluid heated from below RT>0 COLD HOT 12

Hydrodynamic Rayleigh-Taylor instability Heavy fluid on top of a light one Rc>0 Fernandez et al. , J. Fluid Mech. (2002) 13

Double diffusive instability Salt fingers: Hot salty water lies over cold fresh water of a higher density. The stratification is kept gravitationally stable. The key to the instability is the fact that heat diffuses much more rapidly than salt (hence the term double-diffusion). A downward moving finger of warm saline water cools off via quick diffusion of heat, and therefore becomes more dense. This provides the downward buoyancy force that drives the finger. Similarly, an upward-moving finger gains heat from the surrounding fingers, becomes lighter, and rises. RT<0, Rc>0 Salt fingers: Instability even if light solution on top of a heavy solution (statistically stable density gradient) ! 14

Pure double diffusion (without chemistry): Le=20 Heated from below UNSTABLE OSCILLATING Heavy at bottom Thermal Rayleigh-Bénard Solutal Rayleigh-Taylor Salt fingers STABLE Light at the bottom Cooled from below Turner 15

Chemical fronts F( C ) = - C (C-1) (C+d) 16
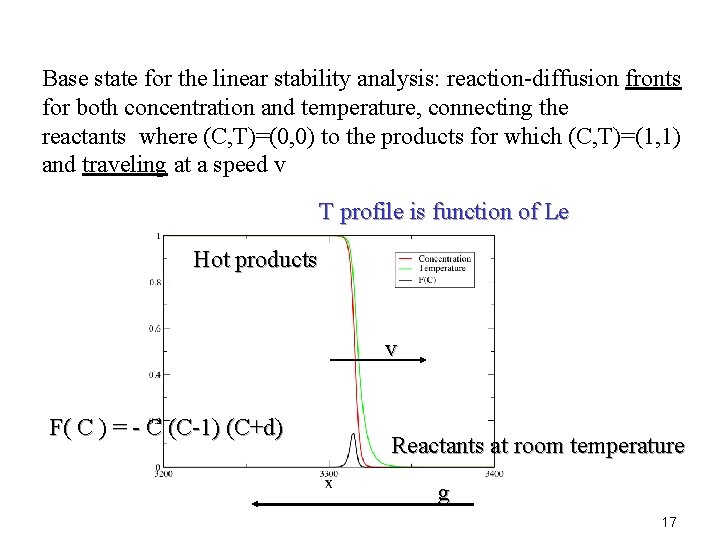
Base state for the linear stability analysis: reaction-diffusion fronts for both concentration and temperature, connecting the reactants where (C, T)=(0, 0) to the products for which (C, T)=(1, 1) and traveling at a speed v T profile is function of Le Hot products v F( C ) = - C (C-1) (C+d) Reactants at room temperature g 17
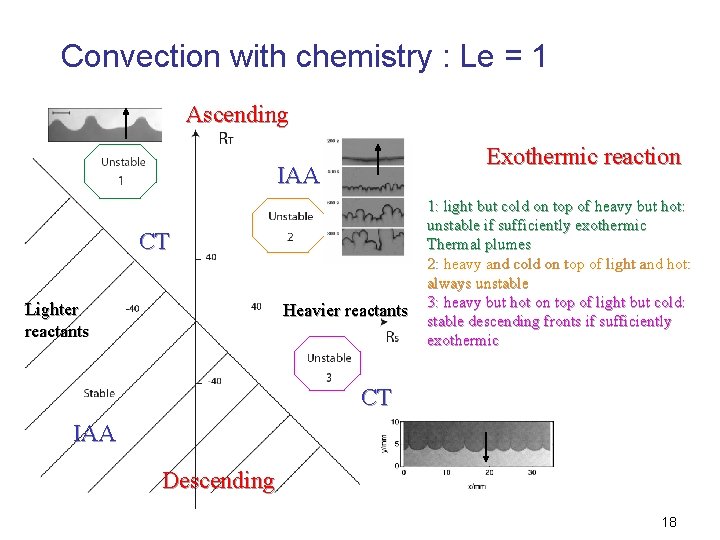
Convection with chemistry : Le = 1 Ascending Exothermic reaction IAA CT Lighter reactants Heavier reactants 1: light but cold on top of heavy but hot: unstable if sufficiently exothermic Thermal plumes 2: heavy and cold on top of light and hot: always unstable 3: heavy but hot on top of light but cold: stable descending fronts if sufficiently exothermic CT IAA Descending 18
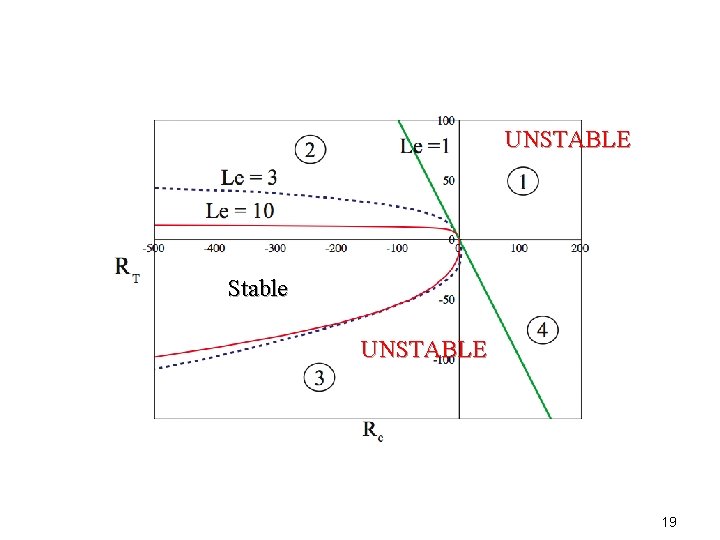
UNSTABLE Stable UNSTABLE 19

Instability due to thermal diffusion and chemistry Descending exothermic front g Light and hot products F(C 1) < F(C 2) T 1> T 2 C 1, T 1 C 2, T 2 Heavy and cold reactants Le>1 C 1, T 2 The little protrusion reaches rapid thermal equilibrium but still reacts at a rate F(C 1) smaller than the rate F(C 2) of its surroundings. It gains thus less heat (the reaction being exothermic) and it thus continues to sink. 20
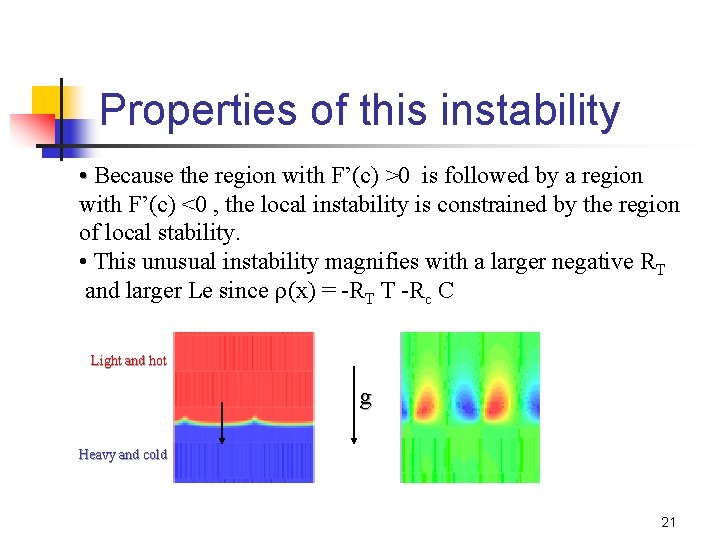
Properties of this instability • Because the region with F’(c) >0 is followed by a region with F’(c) <0 , the local instability is constrained by the region of local stability. • This unusual instability magnifies with a larger negative RT and larger Le since (x) = -RT T -Rc C Light and hot g Heavy and cold 21
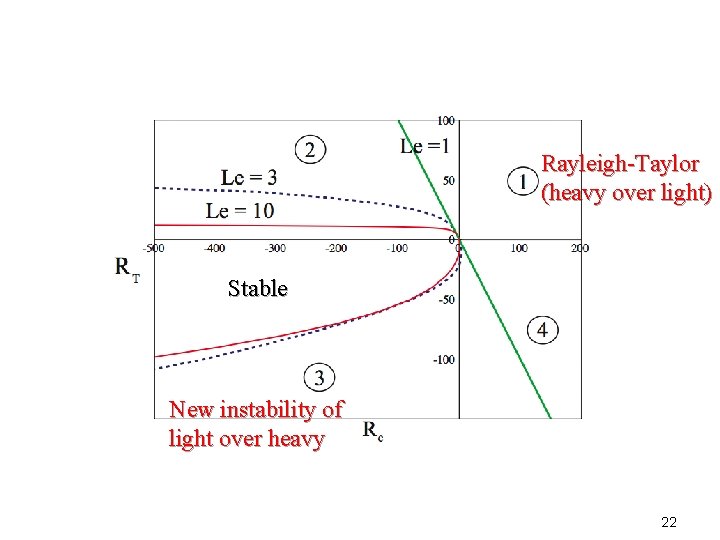
Rayleigh-Taylor (heavy over light) Stable New instability of light over heavy 22
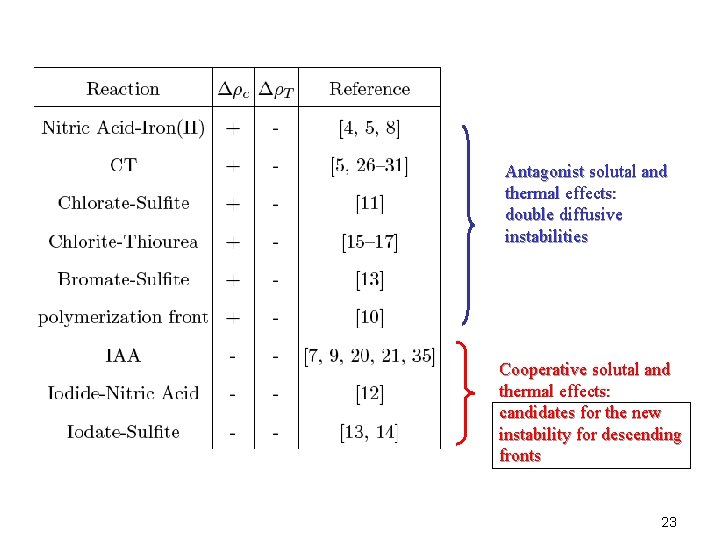
Antagonist solutal and thermal effects: double diffusive instabilities Cooperative solutal and thermal effects: candidates for the new instability for descending fronts 23

Conclusions and perspectives • Classification of the various hydrodynamic instabilities of exothermic reaction-diffusion fronts in the (RT, Rc) plane • Double-diffusive instabilities of chemical fronts have some differences with pure hydrodynamic DD instabilities: ü Different base state ü stability boundaries depend on the kinetics and on Le ü different nonlinear dynamics: frozen fingers • Uncovering of a new instability due to the coupling between thermal diffusion and spatial variations in reaction rate: should be observed in families of exothermic reactions for which D c and D T are both negative 24

Take home message When chemical reactions are at the core of density gradients, the possible resulting hydrodynamic instabilities in the corresponding reaction-diffusion-convection system is not always the simple addition of the usual buoyancy related instabilities on a chemical pattern. New chemically-driven instabilities can arise ! References: J. D'Hernoncourt, A. Zebib and A. De Wit, Phys. Rev. Lett. 96, 154501 (2006). J. D'Hernoncourt, A. De Wit and A. Zebib, J. Fluid Mech. 576, 445 -456 (2007). J. D'Hernoncourt, A. Zebib and A. De Wit, Chaos, 17, 013109 (2007). 25
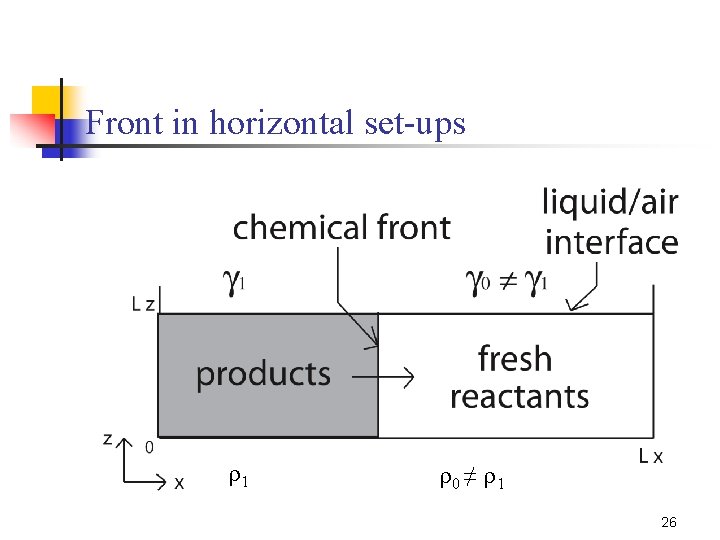
Front in horizontal set-ups 1 0 ≠ 1 26
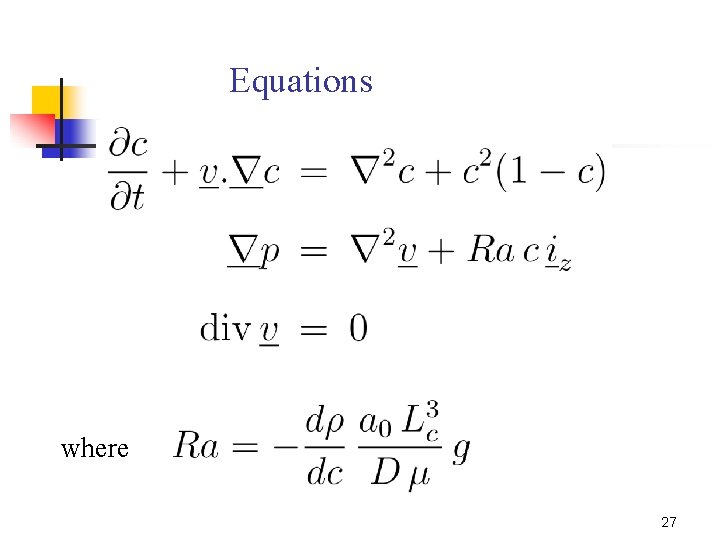
Equations where 27

3. Boundary conditions and Marangoni boundary condition at the free surface : (4) M>0: C g M<0: C g with 28

Marangoni effects Open surface with no buoyancy effects (Ra=0) M = 0 : reaction-diffusion front M = 500 M = - 500 29
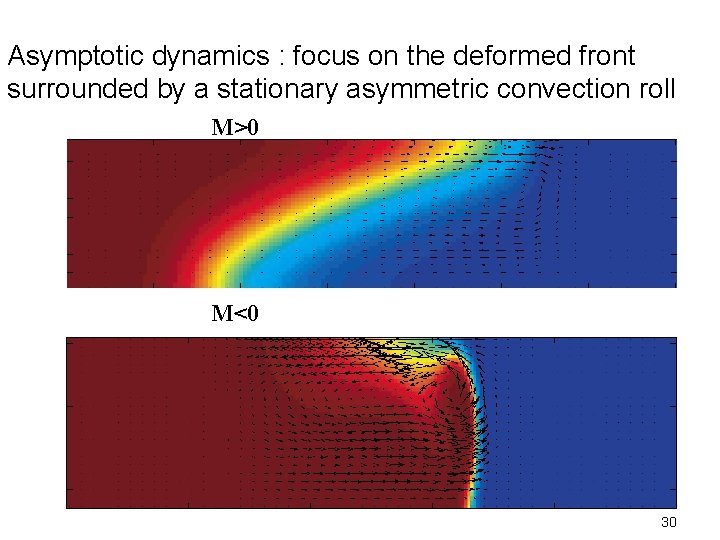
Asymptotic dynamics : focus on the deformed front surrounded by a stationary asymmetric convection roll M>0 M<0 30

Buoyancy effects Closed surface: no Marangoni effects (Ma=0) Ra = 0 : reaction-diffusion front Ra=100: p r : products lighter go on top Ra= - 100: p > r : products heavier sink 31
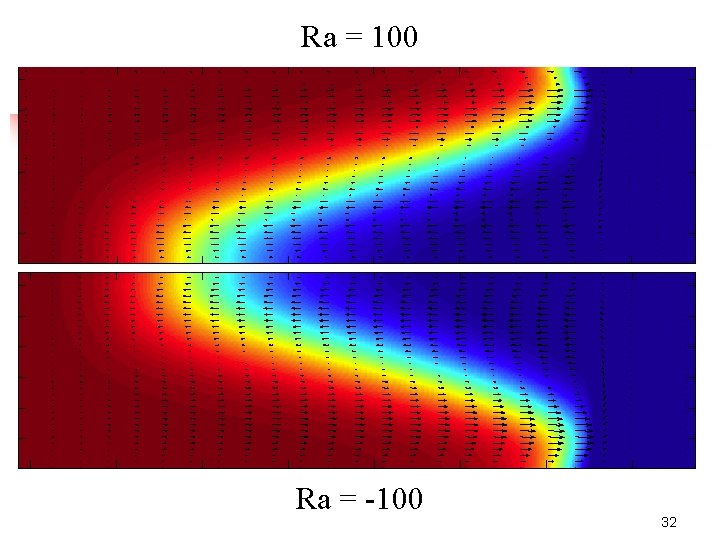
Ra = 100 Ra = -100 32

Buoyancy effects: Comparison with experiments Experiments in capillary tubes with the Bromate-Sulfite reaction : products heavier than reactants => Ra < 0 products reactants A. Keresztessy et al. , Travelling Waves in the Iodate-Sulfite and Bromate-Sulfite Systems , J. Phys. Chem. 99, 5379 -5384, 1995. Qualitative agreement between experiments and theoretical model 33
![Experiments in capillary tubes with the Iodate-Arsenous Acid reaction : d /d[I-] = -1, Experiments in capillary tubes with the Iodate-Arsenous Acid reaction : d /d[I-] = -1,](http://slidetodoc.com/presentation_image_h/c32ebbcc60deb89b12da7d8c7ac23b5a/image-34.jpg)
Experiments in capillary tubes with the Iodate-Arsenous Acid reaction : d /d[I-] = -1, 7. 10 -2 g/cm 3 M Numerical front velocities (10 -3 cm/s) Vnum = 3. 24 Vnum = 4. 84 Vnum = 6. 44 J. Pojman et al. , Convective Effects on Chemical Waves, J. Phys. Chem. 95, 1299 -1306, 1991. Quantitative agreement between experiments and theoretical model 34
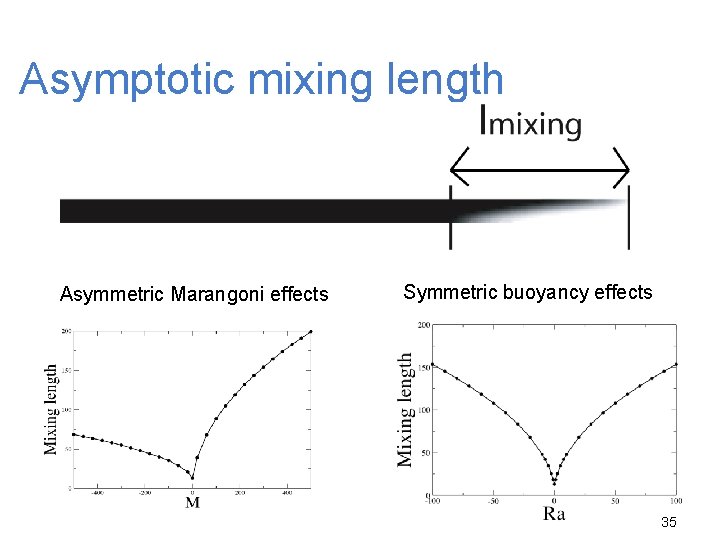
Asymptotic mixing length Asymmetric Marangoni effects Symmetric buoyancy effects 35
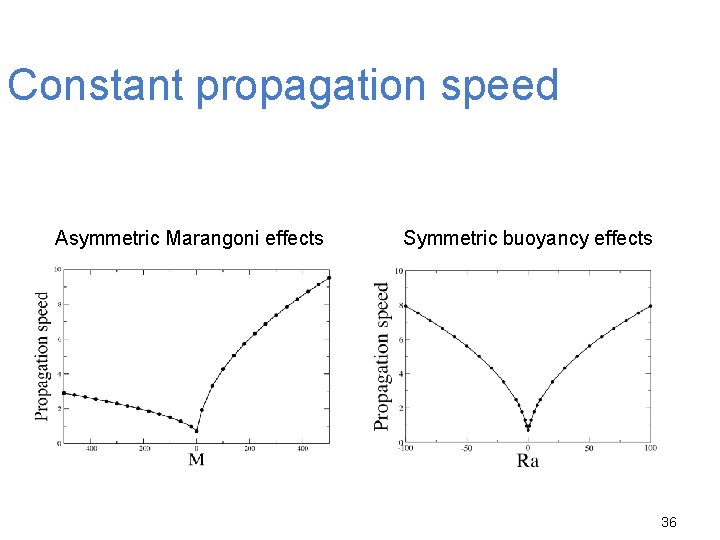
Constant propagation speed Asymmetric Marangoni effects Symmetric buoyancy effects 36

Marangoni effects : Conclusions Asymmetry between the results for M > 0 and M < 0 Increase of the front deformation, the propagation speed and the convective motions with M and Lz Buoyancy effects : Symmetry between the results for Ra > 0 and Ra < 0 Increase of the front deformation, the propagation speed and the convective motions with Ra and Lz 37

References Marangoni effects: - L. Rongy and A. De Wit, ``Steady Marangoni flow traveling with a chemical front", J. Chem. Phys. 124, 164705 (2006). - L. Rongy and A. De Wit, ``Marangoni flow around chemical fronts traveling in thin solution layers: influence of the liquid depth", J. Eng. Math. 59, 221 -227 (2007). Buoyancy effects: - L. Rongy, N. Goyal, E. Meiburg and A. De Wit, ``Buoyancy-driven convection around chemical fronts traveling in covered horizontal solution layers", J. Chem. Phys. 127, 114710 (2007). 38

2008 Gordon conference “Oscillations and dynamic instabilities in chemical systems” July 13 -18, 2008 Colby College, Waterville, USA Chair: Anne De Wit Vice Chair: Oliver Steinbock http: //www. grc. org/programs. aspx? year=2008&program=oscillat 39
- Slides: 39