Hydrocarbons S 2 Chemistry Hydrocarbons Compounds which contain

Hydrocarbons S 2 Chemistry


Hydrocarbons • Compounds which contain carbon and hydrogen only. • Examples? • Where do they come from? – Crude oil


Families • Alkanes • Alkenes • Cycloalkanes

Naming hydrocarbons • • • Meth. Eth. Prop. But. Pent. Hex. Hept. Oct. Non. Dec-

Homologous series • A family of compounds which all have the same general formula and similar chemical properties.

Alkanes • What is their general formula? – Cn. H 2 n+2 • What are their similar chemical properties? – Relatively unreactive – Flammable

Titan

Alkenes • What is their general formula? – Cn. H 2 n

Addition reactions Use the SG Chemistry textbook to find out: • What do the words “saturated” and “unsaturated” mean? • Which family of hydrocarbons is “unsaturated”? • Which family of hydrocarbons take part in addition reactions? • Illustrate two examples of addition reactions: – Using full structural formulae – Using word equations

• Complete Assessment Tests: – 6. 1 – 6. 2 – 6. 4

Cycloalkanes and isomers Use the SG Chemistry textbook to find out… • The names and structures of the first six members of the cycloalkane family. • The general formula of the cycloalkanes. • The definition of the term “isomer”. • The structures of three isomers of pentane. • The structures of three isomers of butene, including one from a different homologous series.

Isomers • Molecules with the same molecular formula but a different structural formula.

Isomers? • • C 4 H 10 and C 4 H 8 Hexane and hexene Hexane and cyclohexane Hexene and cyclohexane • Complete Assessment Test 6. 5
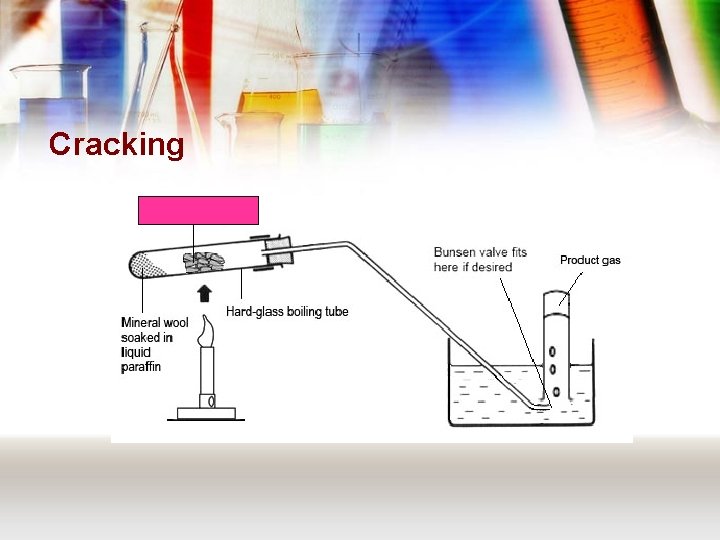
Cracking Aluminium oxide

Cracking

• Complete Assessment Test 6. 3
- Slides: 18