Hydrocarbons Read pp 180 190 Hydrocarbons Compounds that

Hydrocarbons Read pp. 180 -190

Hydrocarbons Compounds that contain ONLY hydrogen and carbon atoms. There are three kinds of hydrocarbons: Alkanes – single bond between carbon atoms Alkenes – one or more of the carbons have double bond(s) Alkynes – one or more of the carbons have triple bond(s)
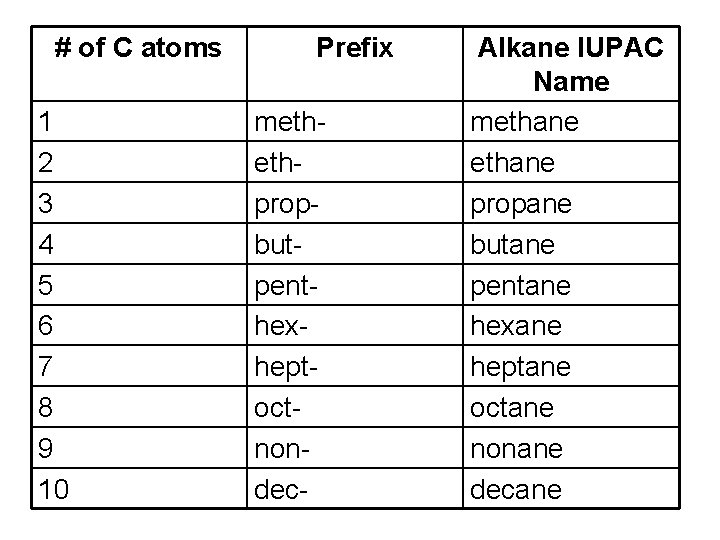
# of C atoms 1 2 3 4 5 6 7 8 9 10 Prefix methethpropbutpenthexheptoctnondec- Alkane IUPAC Name methane propane butane pentane hexane heptane octane nonane decane
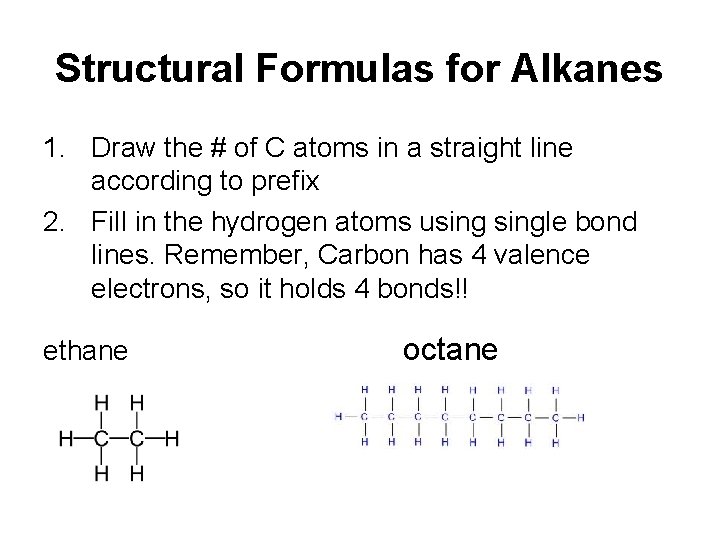
Structural Formulas for Alkanes 1. Draw the # of C atoms in a straight line according to prefix 2. Fill in the hydrogen atoms usingle bond lines. Remember, Carbon has 4 valence electrons, so it holds 4 bonds!! ethane octane
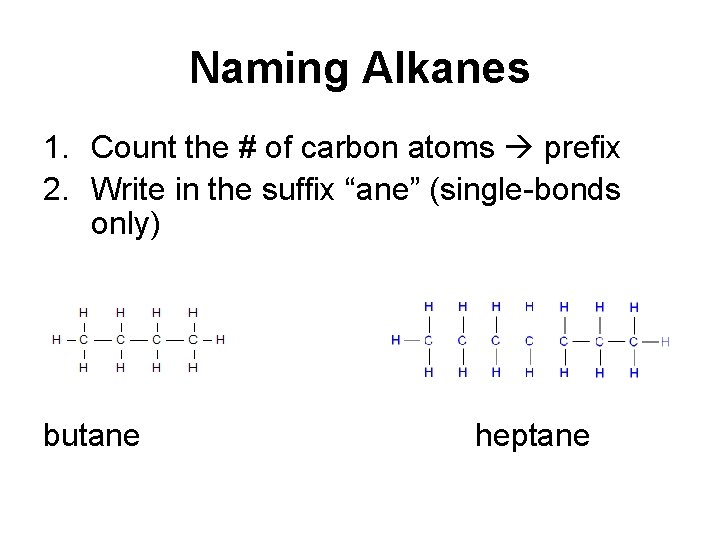
Naming Alkanes 1. Count the # of carbon atoms prefix 2. Write in the suffix “ane” (single-bonds only) butane heptane

Structural Formulas – Alkenes and Alkynes 1. 2. 3. Draw the # of C atoms in straight line according to prefix The number in front of the IUPAC name tells you which C atom the double/triple bond is located. Fill in the H atoms. Remember, each C atom has 4 bonds, so double-check! 2 -hexene 1 -pentyne

Naming Alkenes and Alkynes 1. Look at the structural formula. A double bond tells you it’s an alkene. A triple bond tells you it’s an alkyne. 2. Count # of C atoms prefix 3. Read left-right and right-left. Take the lowest # that the C atom is located on. This # goes in front of prefix 4. Write ‘ene’ if alkene or ‘yne’ if alkyne.
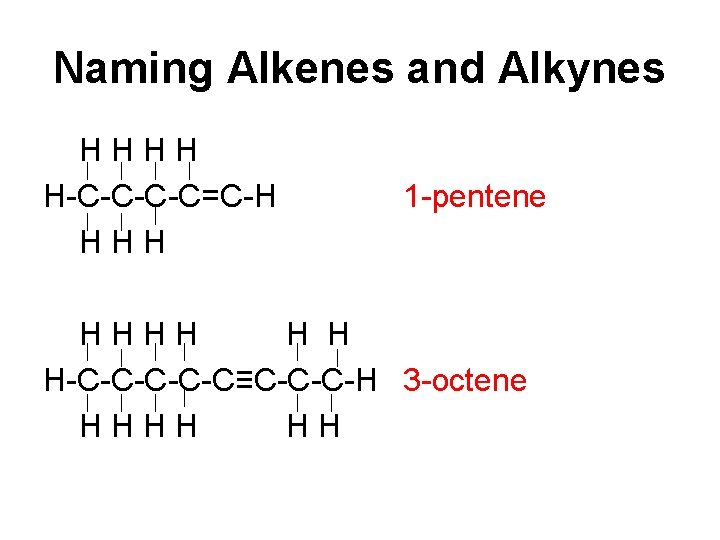
Naming Alkenes and Alkynes HHHH H-C-C=C-H HHH 1 -pentene HHHH H H H-C-C-C≡C-C-C-H 3 -octene HHHH HH

Isomers Compounds that have the SAME molecular formula (eg. C 4 H 8), but have different shapes. This leads to differences in chemical and/or physical properties. 1 -pentene BP: 30°C 2 -pentene BP: 37°C

Properties of Hydrocarbons • Alkanes are saturated. It cannot accept any more atoms/molecules. • Alkenes and alkynes are unsaturated. Their double/triple bonds can be broken down to accept more atoms/molecules (see Addition Reactions) • The longer the hydrocarbon (more C atoms), the more energy it takes to break it apart. • The more C atoms, the higher the boiling point.

Combustion Reactions of Hydrocarbons When a hydrocarbon reacts with oxygen gas to form two products: CO 2 and H 2 O. Write a balanced equation to show the combustion of 1 -pentane. C 5 H 12 + 8 O 2 5 CO 2 + 6 H 2 O

Addition Reactions of Alkenes and Alkynes When a compound is added across a double or triple bond. This compound can be: • A halogen (HCl, HBr, HI, HF) • Water (H 2 O) • Hydrogen gas (H 2) Note: The double bond becomes a single bond; the triple bond becomes a double bond.
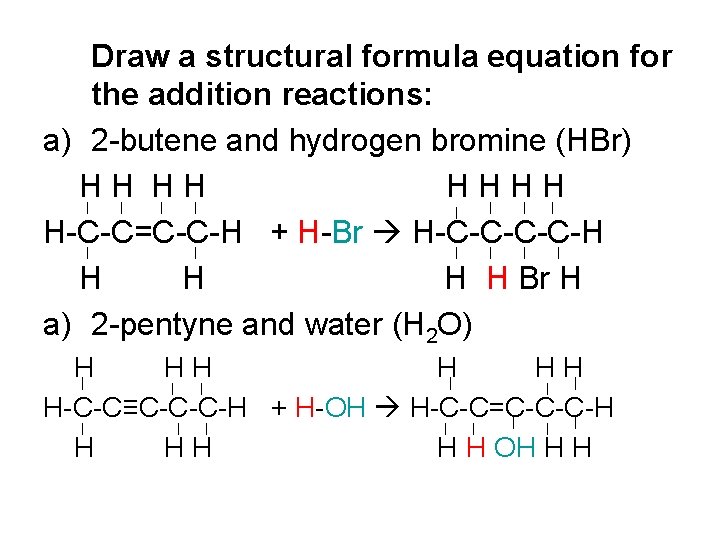
Draw a structural formula equation for the addition reactions: a) 2 -butene and hydrogen bromine (HBr) HH HH HHHH H-C-C=C-C-H + H-Br H-C-C-H H H Br H a) 2 -pentyne and water (H 2 O) H HH H-C-C≡C-C-C-H + H-OH H-C-C=C-C-C-H H H OH H H

c) 2 -heptene and hydrogen gas (H 2) HH HHHHHHH H-C-C=C-C-C-H + H-H H-C-C-C-C-H H HHHHHH
- Slides: 14