Hydrocarbons David Martin City and Islington College A

Hydrocarbons David Martin City and Islington College A hydrocarbon is an organic compound consisting entirely of hydrogen and carbon

Topic Chemistry- Intro Hydrocarbons Aims Ø To help introduce students to Hydrocarbons Aimed at 16 -19 year olds Level 3 Method In-depth Power. Point slides, ALL can be hand-outs, some slides have questions and answers (could be done as a group discussion) Equipment Ø Ø Projector Laptop Pens Hand-Outs Duration >30 Mins

Learning Outcome • Define Hydrocarbons; Saturated and Unsaturated Hydrocarbons • Define alkanes, alkenes and aromatics • Name Hydrocarbons with single and double bond • Name Benzene and substitutes • Isomers • Physical Properties of Hydrocarbons • Chemical Reactions of Hydrocarbons
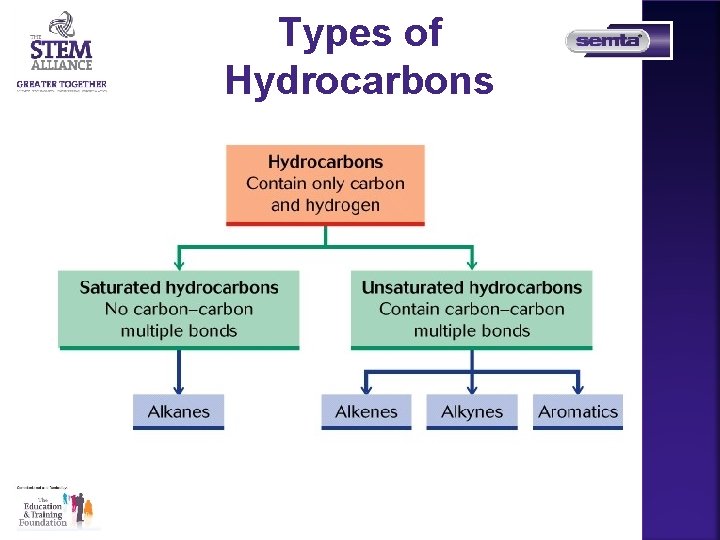
Types of Hydrocarbons

Saturated Hydrocarbons Alkanes • Only single bonds between carbons • Name ends in ___ane • General formula Cn. H 2 n+2

Structural Formula (show the way the atoms are arranged) • Carbon always has 4 bonds • Hydrogen always has 1 bond C or C H or C
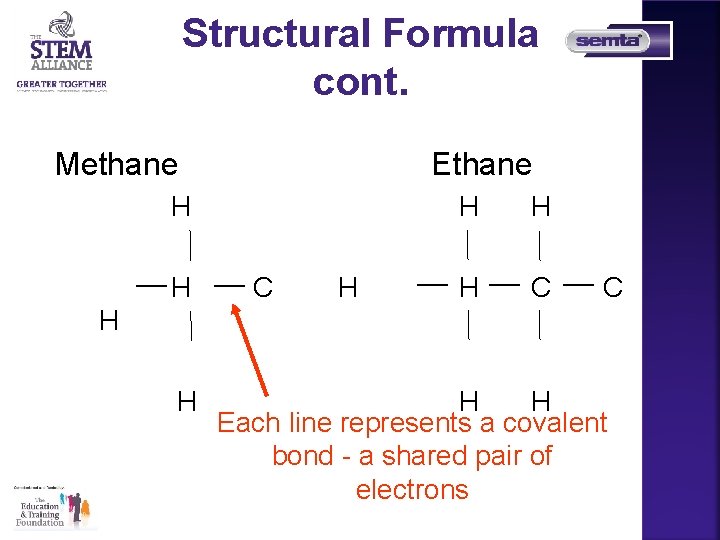
Structural Formula cont. Methane Ethane H H C C H H Each line represents a covalent bond - a shared pair of electrons

Structural Formulas Alkanes are written with structural formulas that are • expanded to show each bond. • condensed to show each carbon atom and its attached hydrogen atoms. Expanded H H Condensed CH 4 , methane
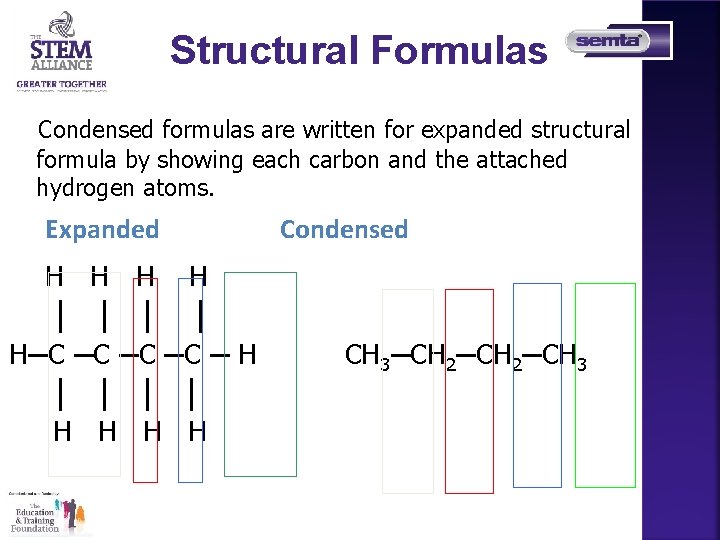
Structural Formulas Condensed formulas are written for expanded structural formula by showing each carbon and the attached hydrogen atoms. Expanded H H │ │ H─C ─C ─ H │ │ H H Condensed CH 3─CH 2─CH 3

Naming Alkanes Copyright © 2008 by Pearson Education, Inc. Publishing as Benjamin Cummings
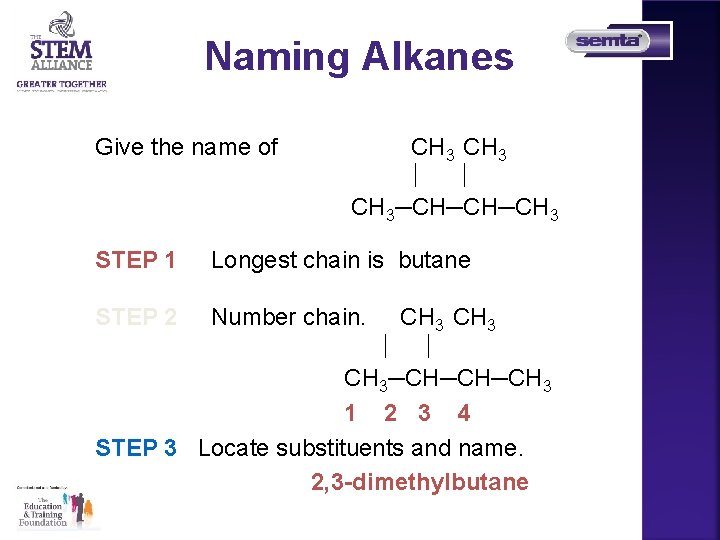
Naming Alkanes Give the name of CH 3─CH─CH─CH 3 STEP 1 Longest chain is butane STEP 2 Number chain. CH 3─CH─CH─CH 3 1 2 3 4 STEP 3 Locate substituents and name. 2, 3 -dimethylbutane
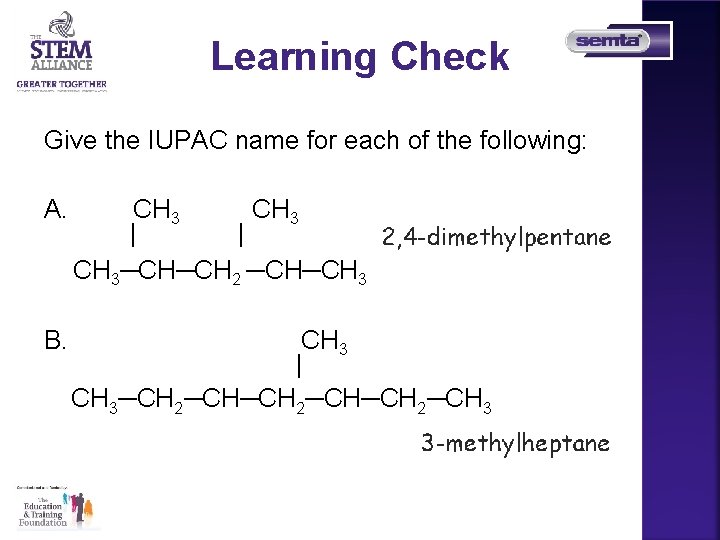
Learning Check Give the IUPAC name for each of the following: A. CH 3 | | CH 3─CH─CH 2 ─CH─CH 3 B. CH 3 | CH 3─CH 2─CH─CH 2─CH 3

Unsaturated Hydrocarbons Alkenes • Contain one double bond • Name ends in ___ene • General formula Cn. H 2 n
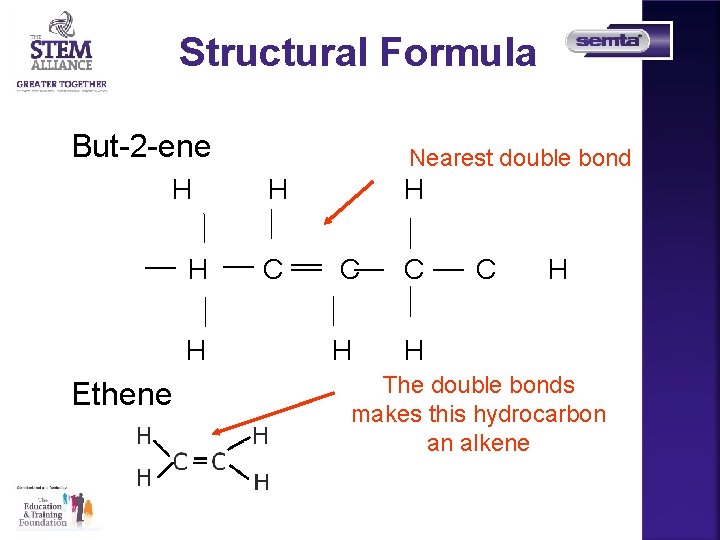
Structural Formula But-2 -ene H H H Ethene Nearest double bond H C C H H C H The double bonds makes this hydrocarbon an alkene

Naming Alkenes and Alkynes Copyright © 2008 by Pearson Education, Inc. Publishing as Benjamin Cummings
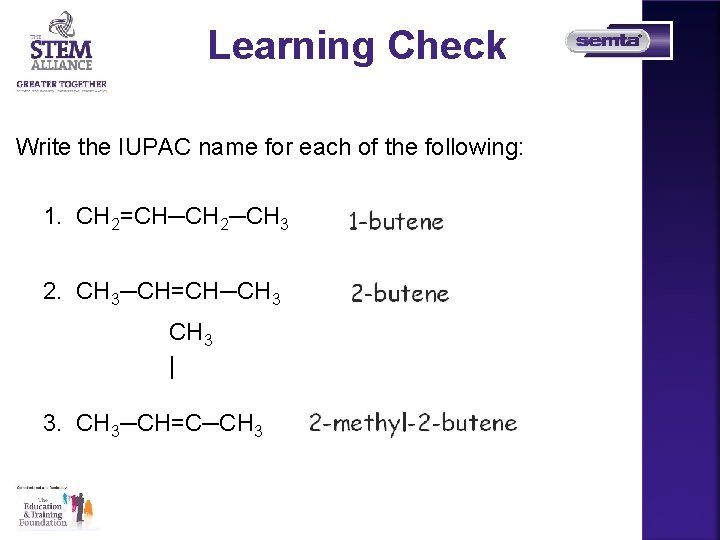
Learning Check Write the IUPAC name for each of the following: 1. CH 2=CH─CH 2─CH 3 2. CH 3─CH=CH─CH 3 | 3. CH 3─CH=C─CH 3

Example 1. How many carbon atoms is the longest chain? 4; but 2. Is there a side chain? If yes what is the name? 3. Is it an alkane, alkene or aromatic? 4. What number carbon atom the side chain is attached? 5. Give the final name of the hydrocarbon. Yes. Methyl Alkane; -ane 2; lowest number in the chain 2 -methyl butane
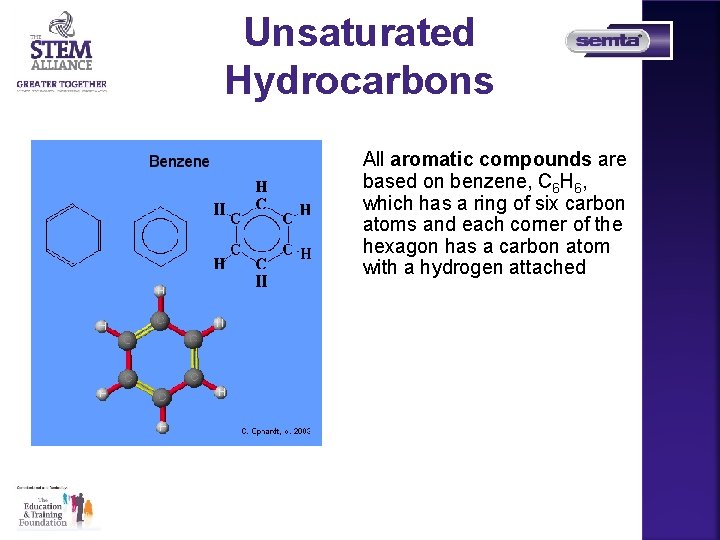
Unsaturated Hydrocarbons All aromatic compounds are based on benzene, C 6 H 6, which has a ring of six carbon atoms and each corner of the hexagon has a carbon atom with a hydrogen attached
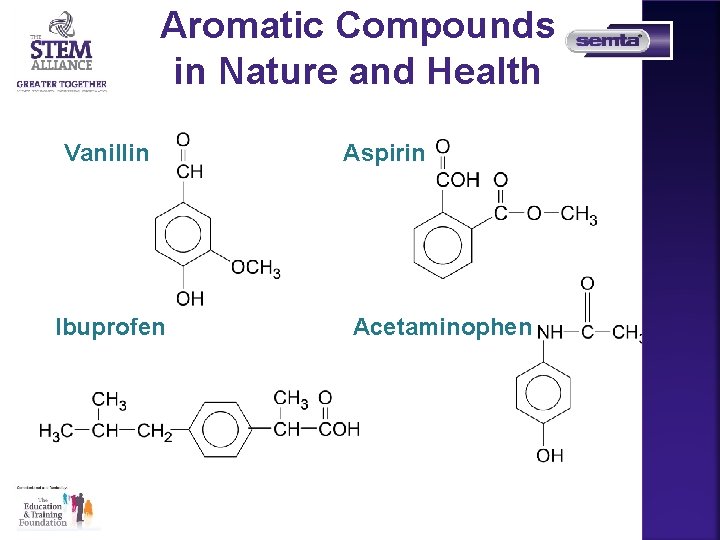
Aromatic Compounds in Nature and Health Vanillin Ibuprofen Aspirin Acetaminophen 19
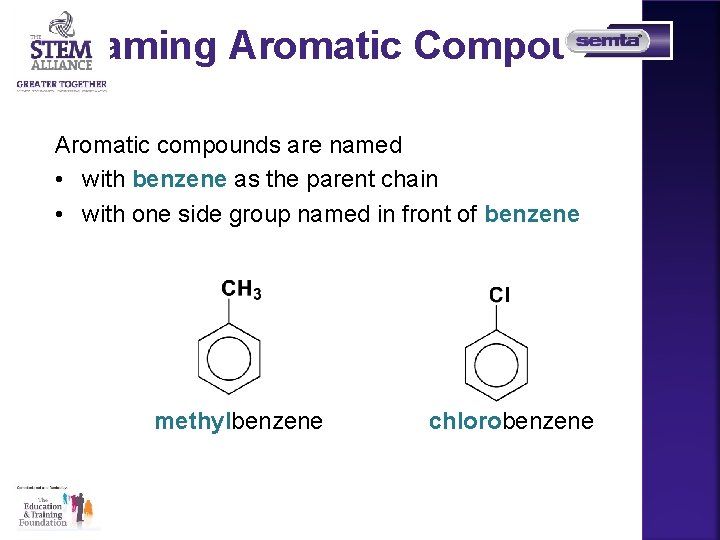
Naming Aromatic Compounds Aromatic compounds are named • with benzene as the parent chain • with one side group named in front of benzene methylbenzene chlorobenzene

Some Common Names Some substituted benzene rings • have common names that have been in use for many years • with a single substituent use a common name or are named as a benzene derivative toluene (methylbenzene) aniline (benzenamine) phenol (hydroxybenzene)

Naming Aromatic Compounds When two groups are attached to the benzene ring, the ring is numbered to give the lowest numbers to the side groups. 3 -chlorotoluene 1, 3 -dichlorobenzene 2 -chlorophenol
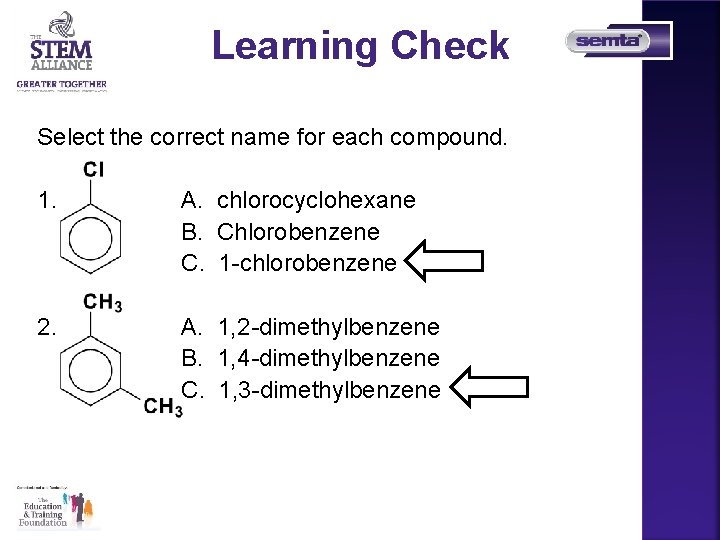
Learning Check Select the correct name for each compound. 1. A. chlorocyclohexane B. Chlorobenzene C. 1 -chlorobenzene 2. A. 1, 2 -dimethylbenzene B. 1, 4 -dimethylbenzene C. 1, 3 -dimethylbenzene 23
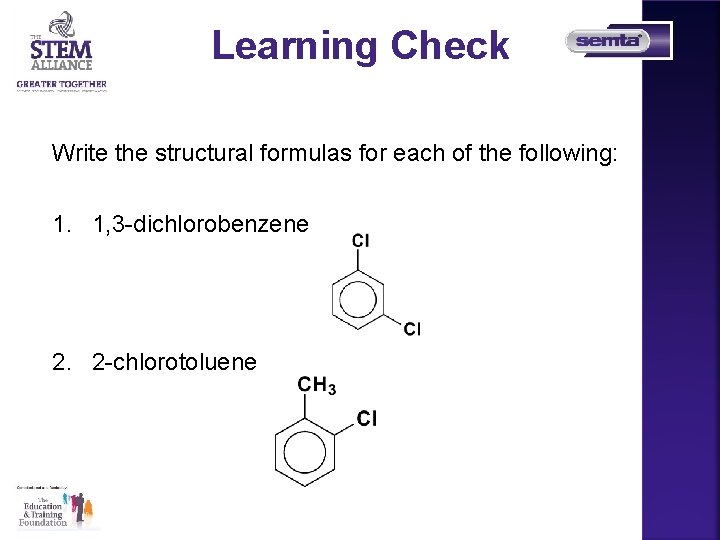
Learning Check Write the structural formulas for each of the following: 1. 1, 3 -dichlorobenzene 2. 2 -chlorotoluene
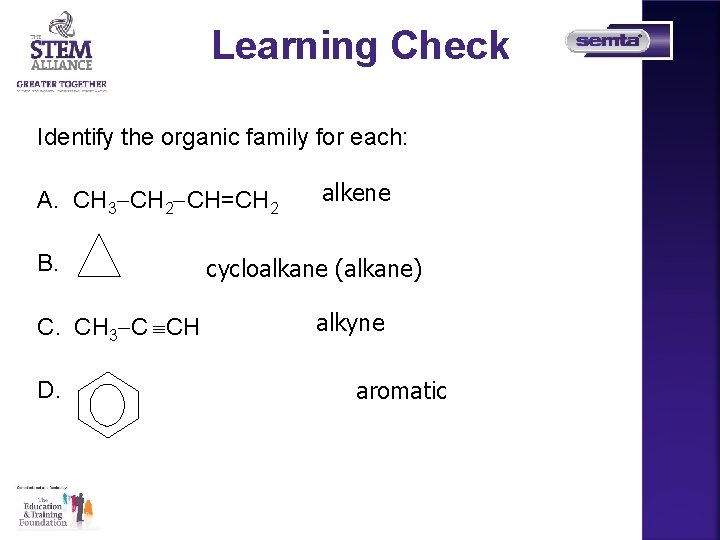
Learning Check Identify the organic family for each: A. CH 3 CH 2 CH=CH 2 B. C. CH 3 C CH D. alkene cycloalkane (alkane) alkyne aromatic
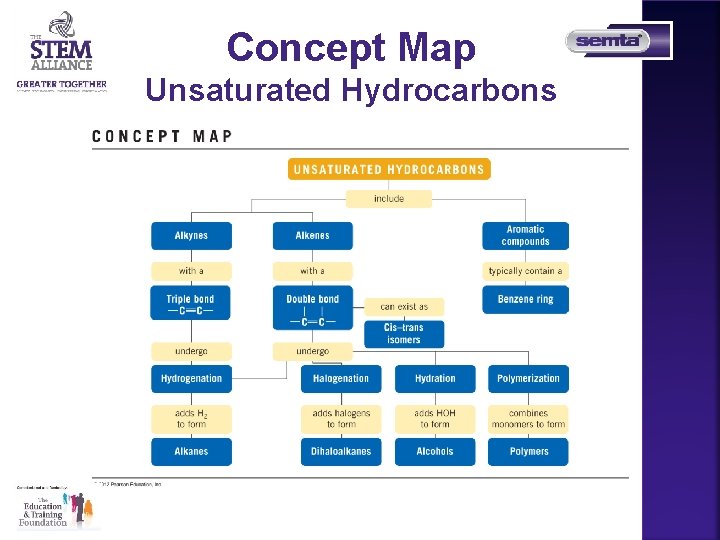
Concept Map Unsaturated Hydrocarbons

What are Isomers? Isomers are molecules that have the same molecular formula, but have a different arrangement of the atoms in space. That excludes any different arrangements which are simply due to the molecule rotating as a whole, or rotating about particular bonds.
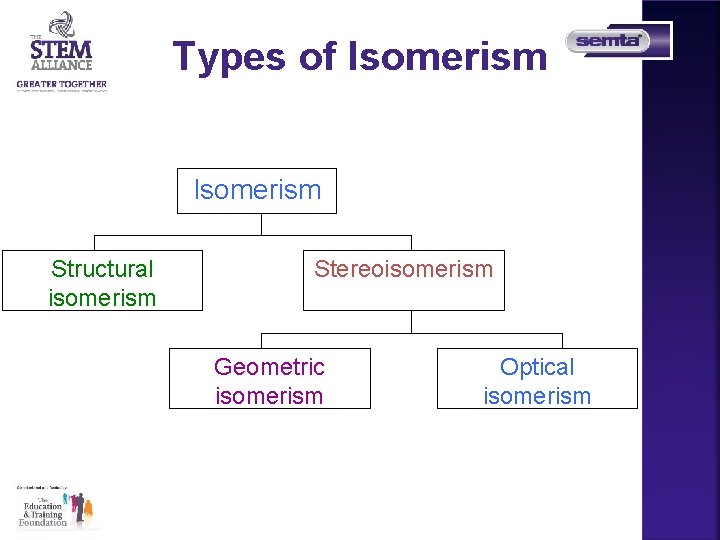
Types of Isomerism Structural isomerism Stereoisomerism Geometric isomerism Optical isomerism

What are Structural Isomers? In structural isomerism, the atoms are arranged in a completely different order.

Chain Isomerism These isomers arise because of the possibility of branching in carbon chains. For example, there are two isomers of butane, C 4 H 10. In one of them, the carbon atoms lie in a "straight chain" whereas in the other the chain is branched.
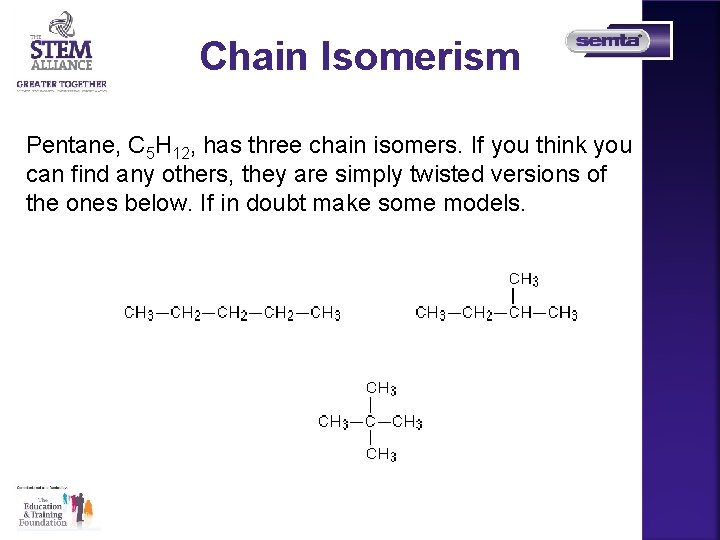
Chain Isomerism Pentane, C 5 H 12, has three chain isomers. If you think you can find any others, they are simply twisted versions of the ones below. If in doubt make some models.

Position Isomerism In position isomerism, the basic carbon skeleton remains unchanged, but important groups are moved around on that skeleton.

Stereoisomerism Geometric Isomerism In stereoisomerism, the atoms making up the isomers are joined up in the same order, but still manage to have a different spatial arrangement. Geometric isomerism is one form of stereoisomerism. Geometric isomerism (also known as cis-trans isomerism or E-Z isomerism) is a form of stereoisomerism
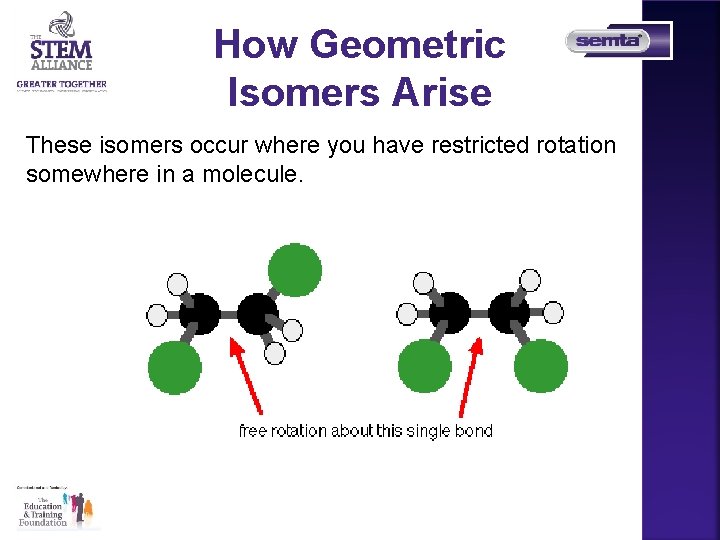
How Geometric Isomers Arise These isomers occur where you have restricted rotation somewhere in a molecule.
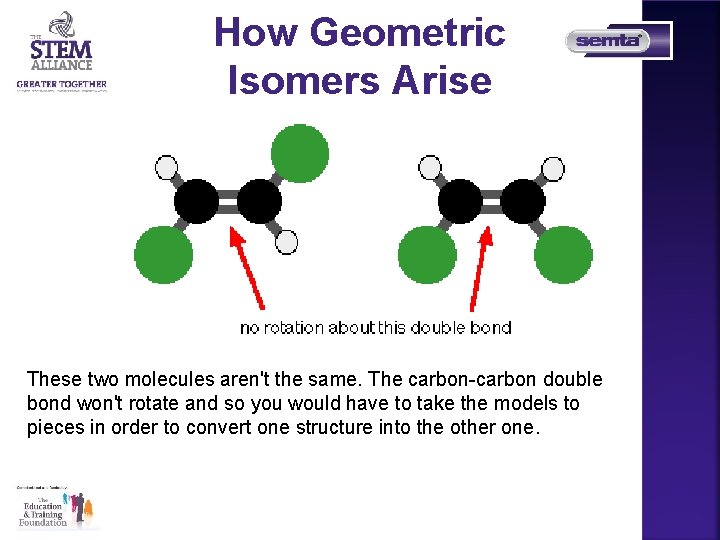
How Geometric Isomers Arise These two molecules aren't the same. The carbon-carbon double bond won't rotate and so you would have to take the models to pieces in order to convert one structure into the other one.
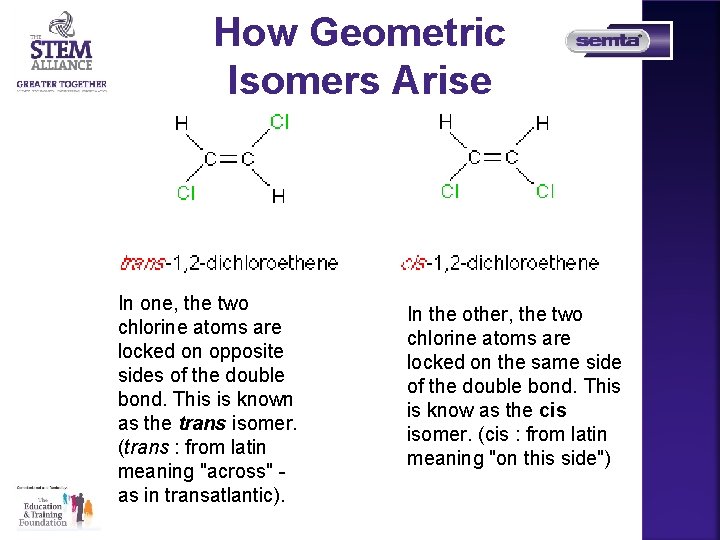
How Geometric Isomers Arise In one, the two chlorine atoms are locked on opposite sides of the double bond. This is known as the trans isomer. (trans : from latin meaning "across" as in transatlantic). In the other, the two chlorine atoms are locked on the same side of the double bond. This is know as the cis isomer. (cis : from latin meaning "on this side")
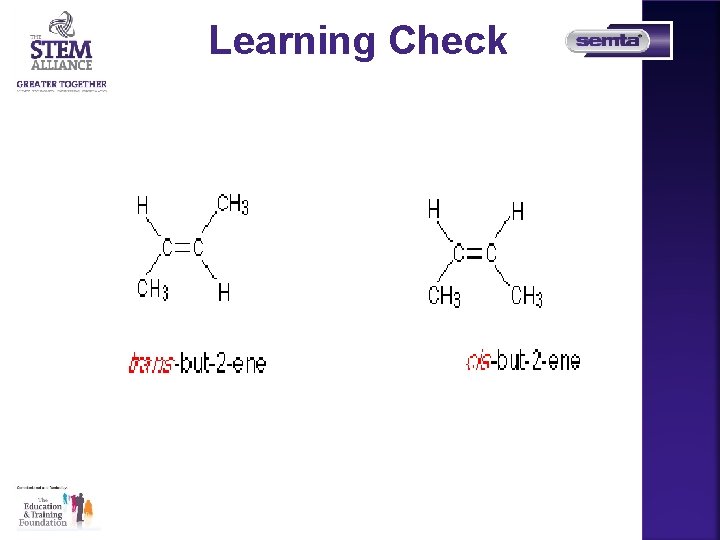
Learning Check

The E-Z System Could you name these two isomers using cis and trans? You look at what is attached to each end of the double bond in turn, and give the two groups a "priority" according to a set of rules
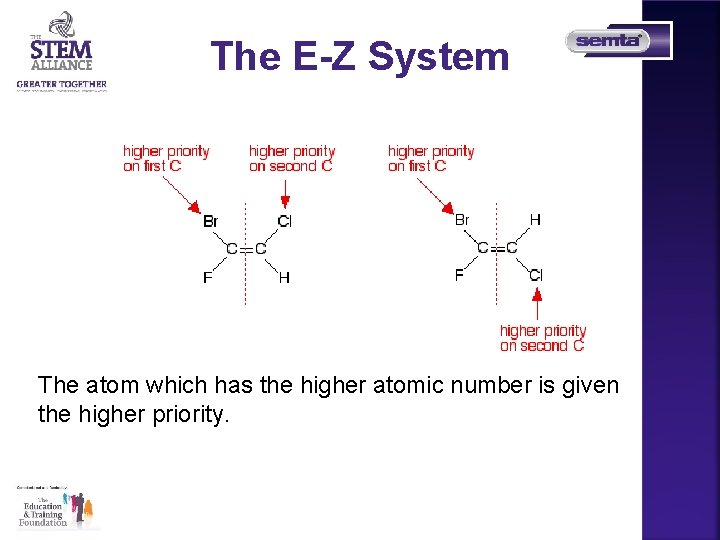
The E-Z System The atom which has the higher atomic number is given the higher priority.

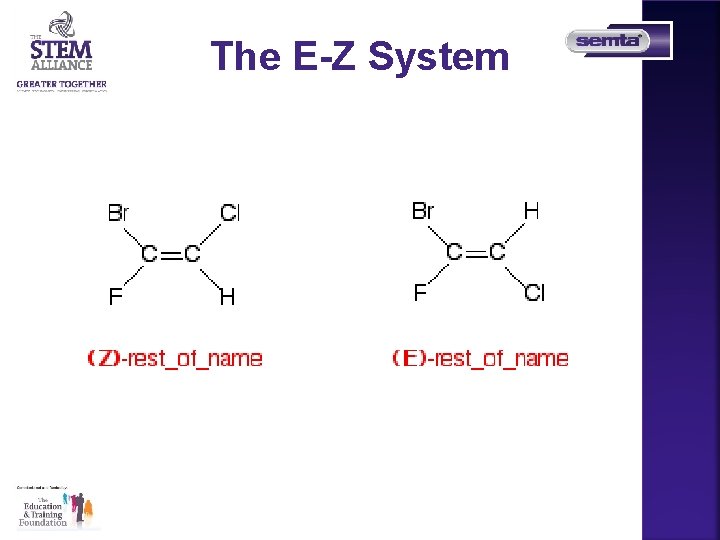
The E-Z System If the two groups with the higher priorities are on the same side of the double bond, that is described as the (Z)- isomer. So you would write it as (Z)-name of compound. The symbol Z comes from a German word (zusammen) which means together. If the two groups with the Higher priorities are on opposite sides of the double bond, then this is The (E)- isomer. E comes from the German entgegen which means opposite.
The E-Z System
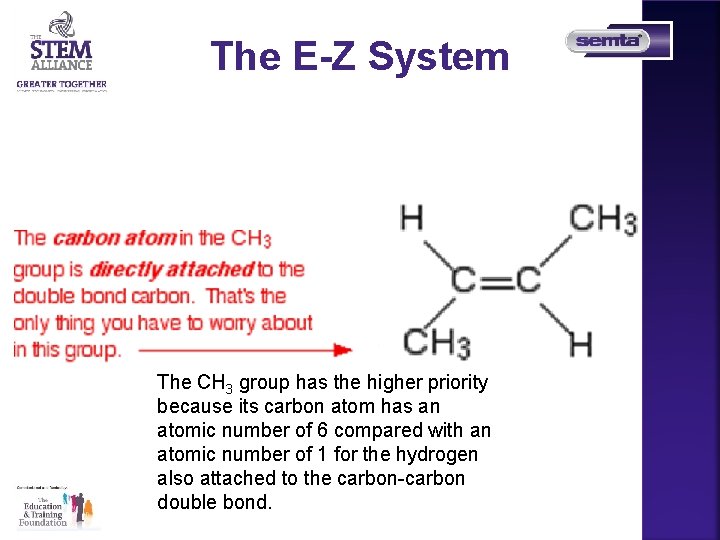
The E-Z System The CH 3 group has the higher priority because its carbon atom has an atomic number of 6 compared with an atomic number of 1 for the hydrogen also attached to the carbon-carbon double bond.
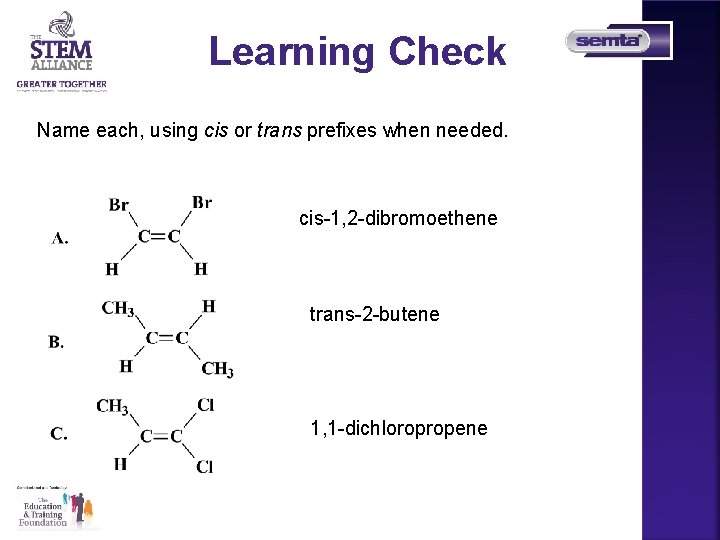
Learning Check Name each, using cis or trans prefixes when needed. cis-1, 2 -dibromoethene trans-2 -butene 1, 1 -dichloropropene

Some Uses of Alkanes with one to four carbons are gases at room temperature and are widely used as heating fuels. methane, propane, butane Butane has four carbons: Alkanes with five to eight carbons are highly volatile liquids at room temperature, making them useful in fuels such as gasoline. pentane, hexane, heptane, octane Octane has eight carbons:

Some Uses of Alkanes with 9− 17 carbons are liquids with higher boiling points and are found in motor oils, mineral oil, kerosene, diesel, and jet fuels. Decane has 10 carbons:

Some Uses of Alkanes with 18 or more carbon atoms • have high molar masses • are waxy solids at room temperature • are used in waxy coatings of fruits and vegetables

Solubility and Density of Alkanes are • nonpolar • insoluble in water • less dense than water • flammable in air The crude oil in oil spills floats on top of the water, forming a thin layer on the surface because it is less dense than water.
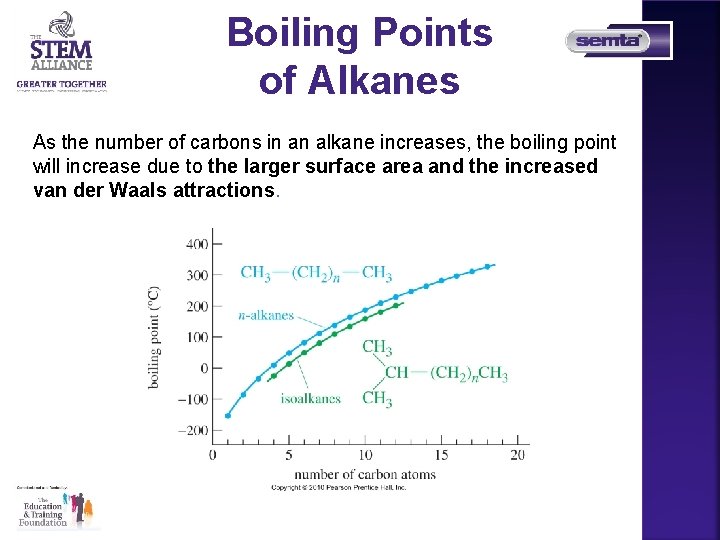
Boiling Points of Alkanes As the number of carbons in an alkane increases, the boiling point will increase due to the larger surface area and the increased van der Waals attractions.
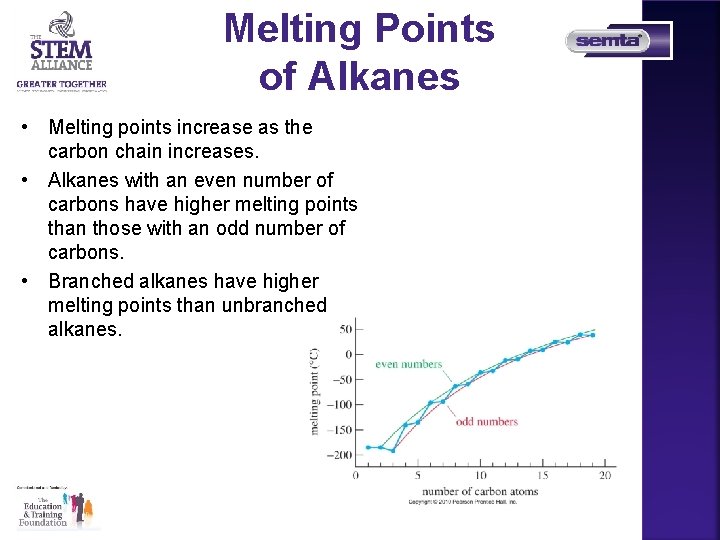
Melting Points of Alkanes • Melting points increase as the carbon chain increases. • Alkanes with an even number of carbons have higher melting points than those with an odd number of carbons. • Branched alkanes have higher melting points than unbranched alkanes.

Physical Properties of Alkenes • • • Physical state The first lower member like ethene, propene and butene are colorless gases. Alkenes with five to fifteen carbon atoms are liquids and higher ones are solids at ordinary temperatures. Alkenes have characteristic smell. Density Alkenes are lighter than water. Solubility Alkenes are insoluble in water and soluble in organic solvents such as benzene, ether etc. Boiling point The boiling points of alkenes gradually increase with an increase in the molecular mass (or chain length). This indicates that inter-molecular attractions become stronger with an increase in the size of the molecule. Branched chain alkenes have lower boiling points than the corresponding straight chain isomers. This is because cis-isomers are polar molecules and have higher boiling points when compared to the trans-isomers. . cis-2 -butene (b. p. = 3. 7°C) and trans-2 -butene (b. p. = 1°C) Melting point The melting points of alkenes increase with an increase in the molecular mass. The trans-isomers fit well into the crystal lattice due to their symmetrical structure. This results in their melting at higher temperatures as compared to the cis form.

Uses of Alkenes are very useful compounds. Major uses are: • Lower alkenes are used as fuel and illuminate. These may be obtained by the cracking of kerosene or petrol. • For the manufacture of a wide variety of polymers, e. g. , polyethene, polyvinylchloride (PVC) and teflon etc. • As a raw material for the manufacture of industrial Chemicals such as alcohols, aldehydes, etc. • For producing lamp black.

Properties of Benzene • Benzene is a colourless liquid (melting point 5. 5°C, boiling point 80. 4°C) with a characteristic smell of petrol. • It is immiscible with water, but dissolves in organic solvents. • It is lighter than water: density 0. 87 g cm-3 • Its vapours are toxic.

Uses of Benzene • As an industrial solvent for fats and oils, rubber, resins etc. • As a starting material for dyes, drugs, perfumes and explosives and polymers • For dry-cleaning of woollen clothes. • As a motor fuel along with petrol, under the name benzol.

Combustion of Alkanes • have strong C C bonds • react with oxygen gas to make carbon dioxide and water in combustion reactions • release energy when C C bonds are broken in combustion reactions CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(g) + energy

Trends • The hydrocarbons become harder to ignite as the molecules get bigger. This is because the bigger molecules don't vaporise so easily - the reaction is much better if the oxygen and the hydrocarbon are well mixed as gases. If the liquid isn't very volatile, only those molecules on the surface can react with the oxygen. • Bigger molecules have greater Van der Waals attractions which makes it more difficult for them to break away from their neighbours and turn to a gas. • Provided the combustion is complete, all the hydrocarbons will burn with a blue flame. However, combustion tends to be less complete as the number of carbon atoms in the molecules rises. That means that the bigger the hydrocarbon, the more likely you are to get a yellow, smoky flame.

Reaction with Chlorine or Bromine • • • Requires heat or light for initiation. The most effective wavelength is blue, which is absorbed by chlorine gas. Many molecules of product are formed from absorption of only one photon of light (chain reaction).

Substitution Reaction • In the presence of light alkanes undergo substitution reaction with halogens. • RH + Br 2 RBr + HBr • In a substitution reaction, one atom of a molecule is removed and replaced or substituted by another atom or group of atoms. • Mechanism of substitution reaction involves free radicals.

Free Radical Substitution • Free radicals are atoms or groups of atoms which have a single unpaired electron. A free radical substitution reaction is one involving these radicals. • Free radicals are formed if a bond splits evenly - each atom getting one of the two electrons.

The Free-Radical Chain Reaction • Initiation generates a radical intermediate. • Propagation: The intermediate reacts with a stable molecule to produce another reactive intermediate (and a product molecule). • Termination: Side reactions that destroy the reactive intermediate.
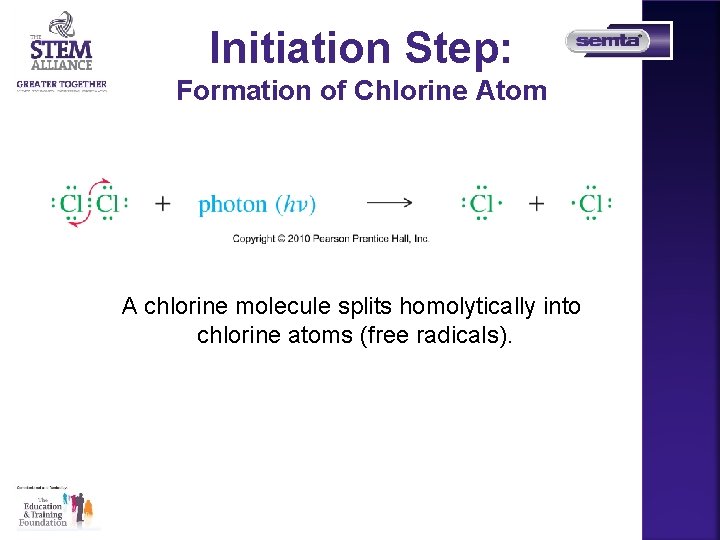
Initiation Step: Formation of Chlorine Atom A chlorine molecule splits homolytically into chlorine atoms (free radicals).

Propagation Step: Carbon Radical The chlorine atom collides with a methane molecule and abstracts (removes) an H, forming another free radical and one of the products (HCl).

Propagation Step: Product Formation The methyl free radical collides with another chlorine molecule, producing the organic product (methyl chloride) and regenerating the chlorine radical.
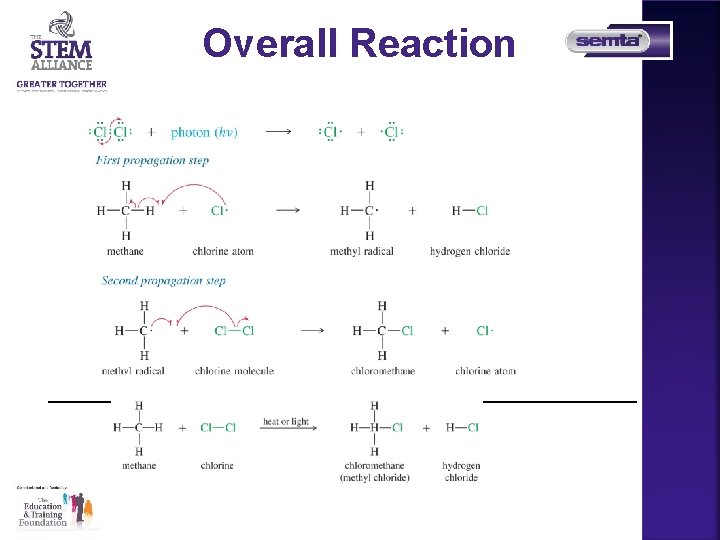
Overall Reaction

Cracking Alkanes What is cracking? Cracking is the name given to breaking up large hydrocarbon molecules into smaller and more useful bits. This is achieved by using high pressures and Temperatures without a catalyst, or lower temperatures and pressures in the presence of a catalyst.
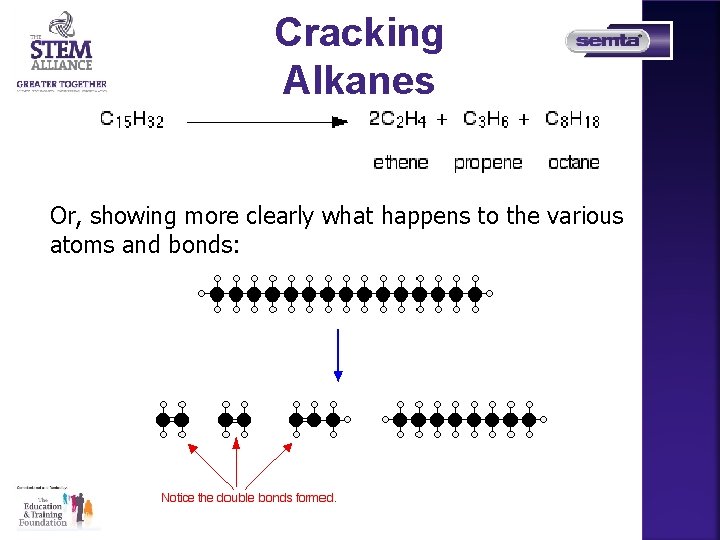
Cracking Alkanes Or, showing more clearly what happens to the various atoms and bonds:

Chemical Reactivity Bonding in the Alkenes Ethene is often modelled like this: The double bond between the carbon atoms is, of course, two pairs of shared electrons. What the diagram doesn't show is that the two pairs aren't the same as each other.
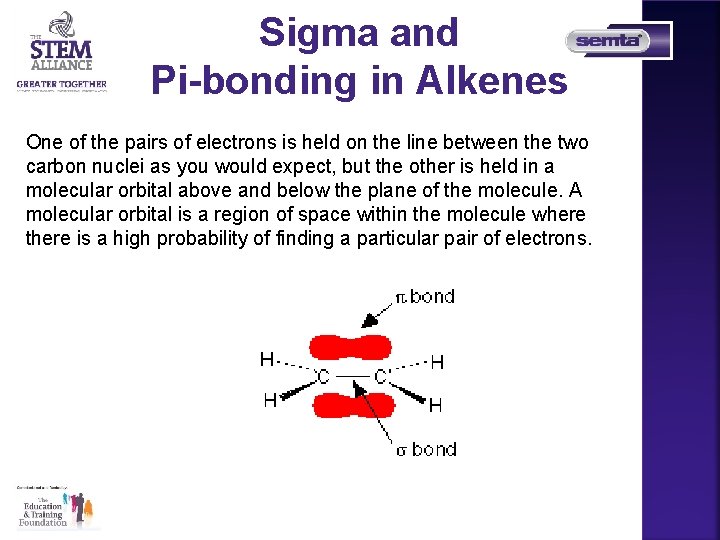
Sigma and Pi-bonding in Alkenes One of the pairs of electrons is held on the line between the two carbon nuclei as you would expect, but the other is held in a molecular orbital above and below the plane of the molecule. A molecular orbital is a region of space within the molecule where there is a high probability of finding a particular pair of electrons.

Sigma and Pi-bonding in Alkenes • The line between the two carbon atoms represents a normal bond the pair of shared electrons lies in a molecular orbital on the line between the two nuclei where you would expect them to be. This sort of bond is called a sigma bond. • The other pair of electrons is found somewhere in the shaded part above and below the plane of the molecule. This bond is called a pi bond. The electrons in the pi bond are free to move around anywhere in this shaded region and can move freely from one half to the other.

The reactions of Alkenes Like any other hydrocarbons, alkenes burn in air or oxygen, but these reactions are unimportant. Alkenes are too valuable to waste in this way. The important reactions all centre around the double bond. Typically, the pi bond breaks and the electrons from it are used to join the two carbon atoms to other things. Alkenes undergo addition reactions.

Addition Reactions • An addition reaction is a reaction in which two molecules join together to make a bigger one. Nothing is lost in the process. All the atoms in the original molecules are found in the bigger one. • Most addition reactions to alkenes follow the mechanism of electrophilic addition

Electrophilic Addition An electrophilic addition reaction is an addition reaction where, in a chemical compound, a pi bond is broken and two new sigma bonds are formed. The substrate of an electrophilic addition reaction must have a double bond or triple bond!

Understanding The Electrophilic Addition Mechanism It is very unlikely that any two different atoms joined together will have the same electronegativity. We are going to assume that Y is more electronegative than X, so that the pair of electrons is pulled slightly towards the Y end of the bond. That means that the X atom carries a slight positive charge.

Understanding The Electrophilic Addition Mechanism The slightly positive X atom is an electrophile and is attracted to the exposed pi bond in the ethene. Now imagine what happens as they approach each other. You are now much more likely to find the electrons in the half of the pi bond nearest the XY. As the process continues, the two electrons in the pi bond move even further towards the X until a covalent bond is made.
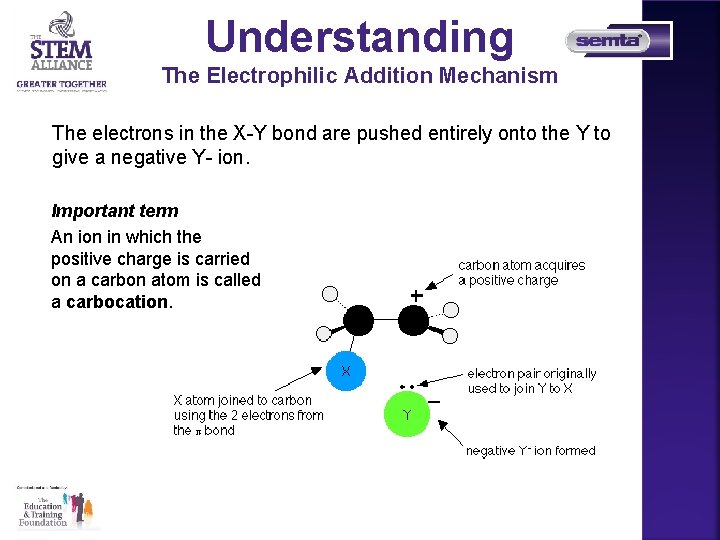
Understanding The Electrophilic Addition Mechanism The electrons in the X-Y bond are pushed entirely onto the Y to give a negative Y- ion. Important term An ion in which the positive charge is carried on a carbon atom is called a carbocation.
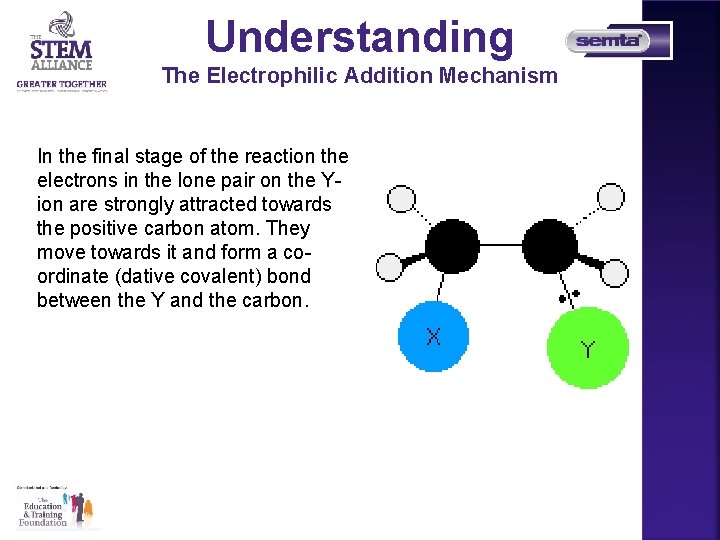
Understanding The Electrophilic Addition Mechanism In the final stage of the reaction the electrons in the lone pair on the Yion are strongly attracted towards the positive carbon atom. They move towards it and form a coordinate (dative covalent) bond between the Y and the carbon.

Understanding The Electrophilic Addition Mechanism Watch this animation for better understanding http: //www. youtube. com/watch? v=v. H--i. R 5 jw. Sk

The Halogenation Of Alkenes Reaction of the carbon-carbon double bond in alkenes such as ethene with halogens such as chlorine, bromine and iodine.
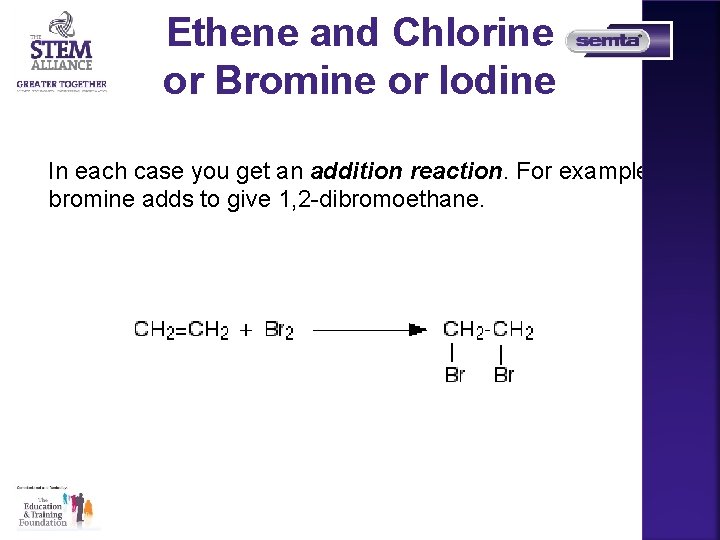
Ethene and Chlorine or Bromine or Iodine In each case you get an addition reaction. For example, bromine adds to give 1, 2 -dibromoethane.

Ethene and Chlorine or Bromine or Iodine • The reaction with bromine happens at room temperature. If you have a gaseous alkene like ethene, you can bubble it through either pure liquid bromine or a solution of bromine in an organic solvent like tetrachloromethane. The reddish-brown bromine is decolourised as it reacts with the alkene. • A liquid alkene (like cyclohexene) can be shaken with liquid bromine or its solution in tetrachloromethane. • Chlorine reacts faster than bromine, but the chemistry is similar. Iodine reacts much, much more slowly, but again the chemistry is similar. You are much more likely to meet the bromine case than either of these.

Ethene and Fluorine Ethene reacts explosively with fluorine to give carbon and hydrogen fluoride gas. This isn't a useful reaction

Alkenes and Hydrogen Halides Reaction of the carbon-carbon double bond in alkenes such as ethene with hydrogen halides e. g. hydrogen chloride (HCl) or hydrogen bromide (HBr). a. Addition to symmetrical alkenes b. Addition to unsymmetrical alkenes

Addition to Symmetrical Alkenes All alkenes undergo addition reactions with the hydrogen halides. A hydrogen atom joins to one of the carbon atoms originally in the double bond, and a halogen atom to the other. For example, with ethene and hydrogen chloride, you get chloroethane: With but-2 -ene you get 2 -chlorobutane:

Conditions • The alkenes react with gaseous hydrogen halides at room temperature. If the alkene is also a gas, you can simply mix the gases. If the alkene is a liquid, you can bubble the hydrogen halide through the liquid. • Alkenes will also react with concentrated solutions of the gases in water. A solution of hydrogen chloride in water is, of course, hydrochloric acid. A solution of hydrogen bromide in water is hydrobromic acid - and so on. • There are, however, problems with this. The water will also get involved in the reaction and you end up with a mixture of products.

Reaction Rates Variation of rates when you change the halogen • Reaction rates increase in the order HF - HCl - HBr - HI. Hydrogen fluoride reacts much more slowly than the other three, and is normally ignored in talking about these reactions. • When the hydrogen halides react with alkenes, the hydrogen-halogen bond has to be broken. The bond strength falls as you go from HF to HI, and the hydrogenfluorine bond is particularly strong. Because it is difficult to break the bond between the hydrogen and the fluorine, the addition of HF is bound to be slow.

Reaction Rates cont. Variation of rates when you change the alkene • This applies to unsymmetrical alkenes as well as to symmetrical ones. • Reaction rates increase as the alkene gets more complicated in the sense of the number of alkyl groups (such as methyl groups) attached to the carbon atoms at either end of the double bond.

Addition to Unsymmetrical Aalkenes The problem comes with the orientation of the addition - in other words, which way around the hydrogen and the halogen add across the double bond.
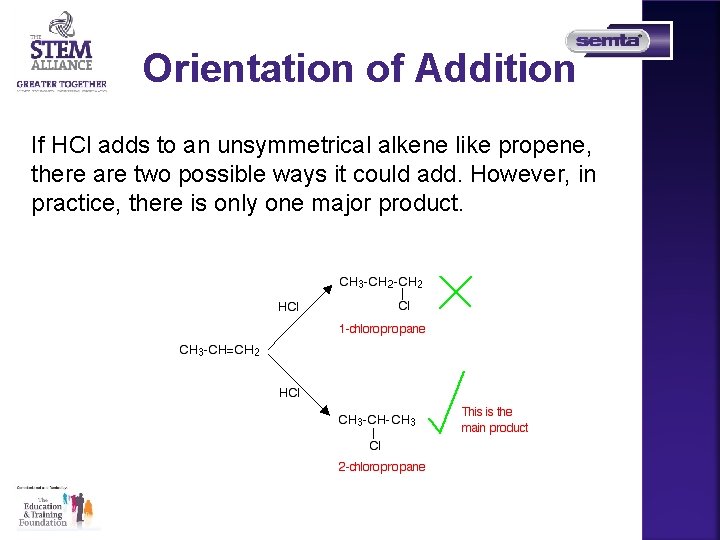
Orientation of Addition If HCl adds to an unsymmetrical alkene like propene, there are two possible ways it could add. However, in practice, there is only one major product.

Markovnikov's Rule When a compound HX is added to an unsymmetrical alkene, the hydrogen becomes attached to the carbon with the most hydrogens attached to it already. • In this case, the hydrogen becomes attached to the CH 2 group, because the CH 2 group has more hydrogens than the CH group. • Notice that only the hydrogens directly attached to the carbon atoms at either end of the double bond count. The ones in the CH 3 group are totally irrelevant.

Alkenes And Sulphuric Acid Reaction of the carbon-carbon double bond in alkenes such as ethene with concentrated sulphuric acid. It includes the conversion of the product into an alcohol.
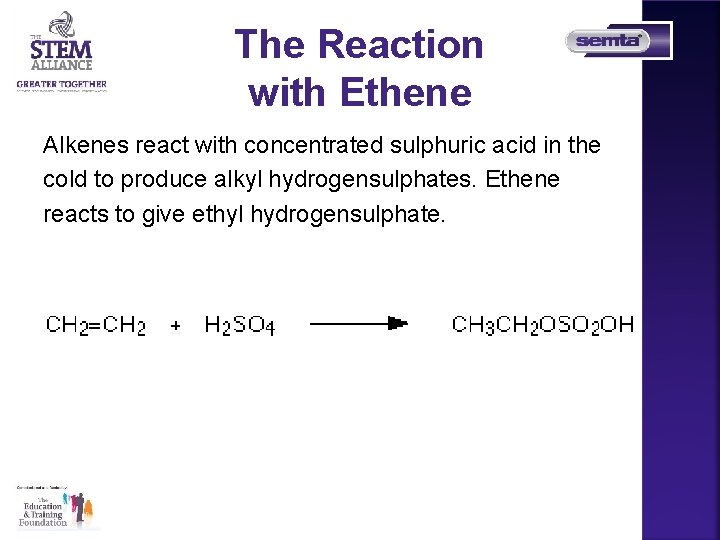
The Reaction with Ethene Alkenes react with concentrated sulphuric acid in the cold to produce alkyl hydrogensulphates. Ethene reacts to give ethyl hydrogensulphate.

The Reaction with Propene If sulphuric acid adds to an unsymmetrical alkene like propene, there are two possible ways it could add. You could end up with one of two products depending on which carbon atom the hydrogen attaches itself to. However, in practice, there is only one major product. Markovnikov's Rule

Using these Reactions to make Alcohols Making Ethanol Ethene is passed into concentrated sulphuric acid to make ethyl hydrogensulphate (as above). The product is diluted with water and then distilled. The water reacts with the ethyl hydrogensulphate to produce ethanol which distils off.

Using these Reactions to make Alcohols cont. Making propan-2 -ol More complicated alkyl hydrogensulphates react with water in exactly the same way. For example: Notice that the position of the -OH group is determined by where the HSO 4 group was attached. You get propan-2 -ol rather than propan-1 -ol because of the way the sulphuric acid originally added across the double bond in propene.

The direct Hydration of Alkenes The production of alcohols by the direct hydration of alkenes adding water directly to the carbon-carbon double bond. The reaction is reversible.
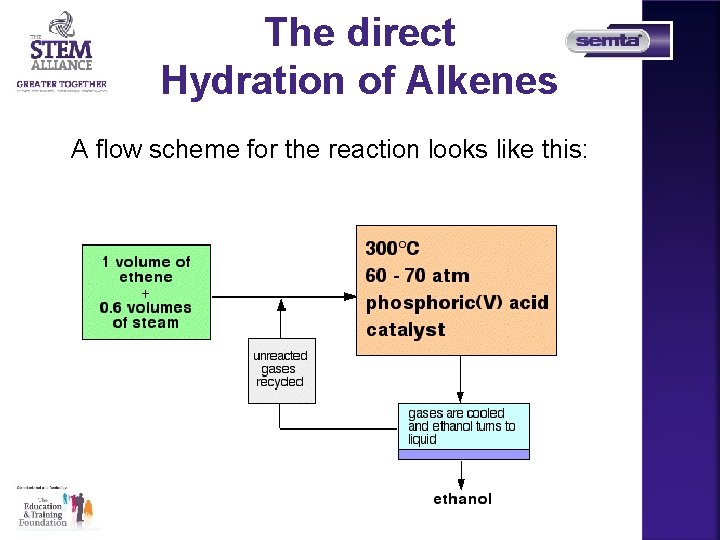
The direct Hydration of Alkenes A flow scheme for the reaction looks like this:

Manufacturing Other Alcohols • If you start from an unsymmetrical alkene like propene, you have to be careful to think about which way around the water adds across the carbon-carbon double bond. • Markovnikov's Rule says that when you add a molecule HX across a carbon-carbon double bond, the hydrogen joins to the carbon atom which already has the more hydrogen atoms attached to it. • Thinking of water as H-OH, the hydrogen will add to the carbon with the more hydrogens already attached. That means that in the propene case, you will get propan-2 -ol rather than propan-1 -ol. • The conditions used during manufacture vary from alcohol to alcohol.

The Polymerisation Of Alkenes The polymerisation of alkenes to produce polymers like poly(ethene) (usually known as polythene, and sometimes as polyethylene), poly(propene) (old name: polypropylene), PVC and PTFE. It also looks briefly at how the structure of the polymers affects their properties and uses.

Polymers are • large, long-chain molecules • found in nature, including cellulose in plants, starches in food, proteins, and DNA in the body • also synthetic, such as polyethylene and polystyrene, Teflon, and nylon • composed of small repeating units called monomers • made from reaction of small alkenes
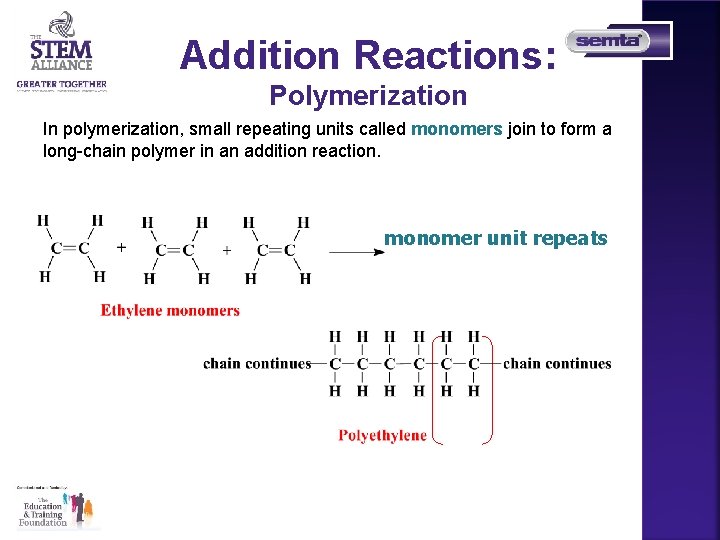
Addition Reactions: Polymerization In polymerization, small repeating units called monomers join to form a long-chain polymer in an addition reaction. monomer unit repeats

Common Synthetic Polymers Synthetic polymers provide a wide variety of items that we use every day.

Common Synthetic Polymers: Polyethylene is made using an addition reaction with the monomer unit ethylene. Common uses for polyethylene include plastic bottles, film, and insulation materials.
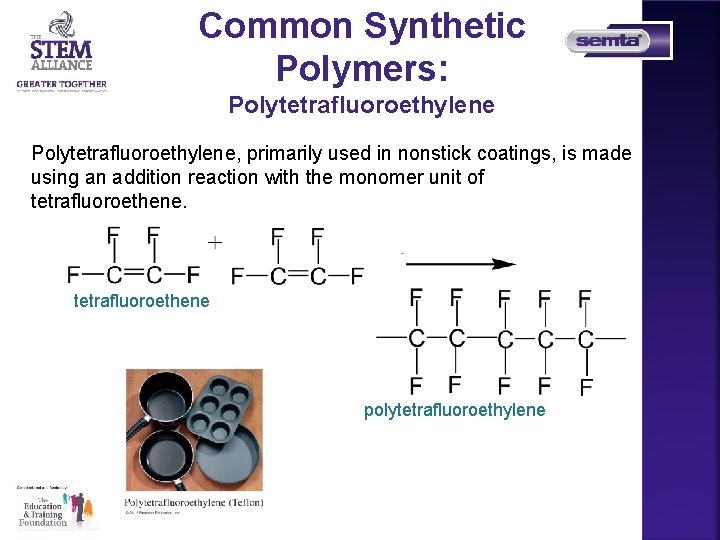
Common Synthetic Polymers: Polytetrafluoroethylene, primarily used in nonstick coatings, is made using an addition reaction with the monomer unit of tetrafluoroethene polytetrafluoroethylene

Common Synthetic Polymers: Polypropylene is used for ski and hiking clothing, carpets, and artificial joints.

Common Synthetic Polymers: Polystyrene is used for plastic coffee cups, cartons, and insulation.

Common Synthetic Polymers: Polyvinylchloride, also known as PVC, is used for plastic pipes and tubing, garden hoses, and garbage bags.

Common Synthetic Polymers: Polydichloroethylene is used in plastic film and wraps.

Recycling Plastics Recycling is simplified by using codes on plastic items. 1 PETE Polyethylene terephtalate 2 HDPE High-density polyethylene 3 PV Polyvinyl chloride 4 LDPE Low-density polyethylene 5 PP Polypropylene 6 PS Polystyrene 7 OTHER Other plastic: fiberglass, polycarbonate, etc.
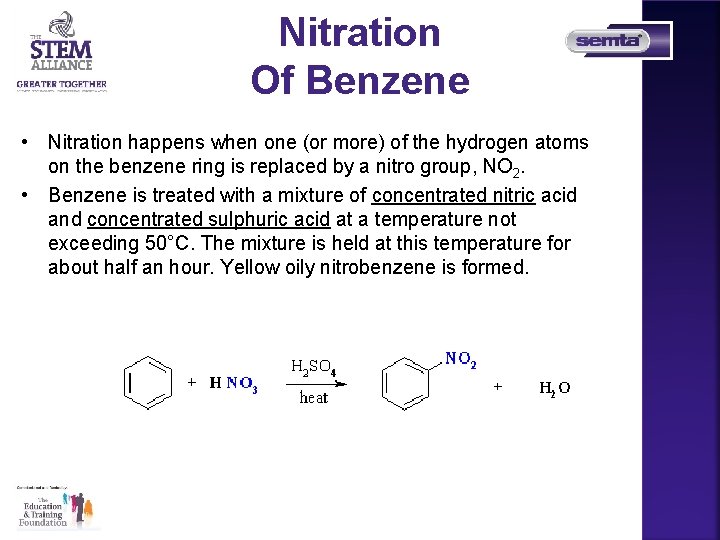
Nitration Of Benzene • Nitration happens when one (or more) of the hydrogen atoms on the benzene ring is replaced by a nitro group, NO 2. • Benzene is treated with a mixture of concentrated nitric acid and concentrated sulphuric acid at a temperature not exceeding 50°C. The mixture is held at this temperature for about half an hour. Yellow oily nitrobenzene is formed.
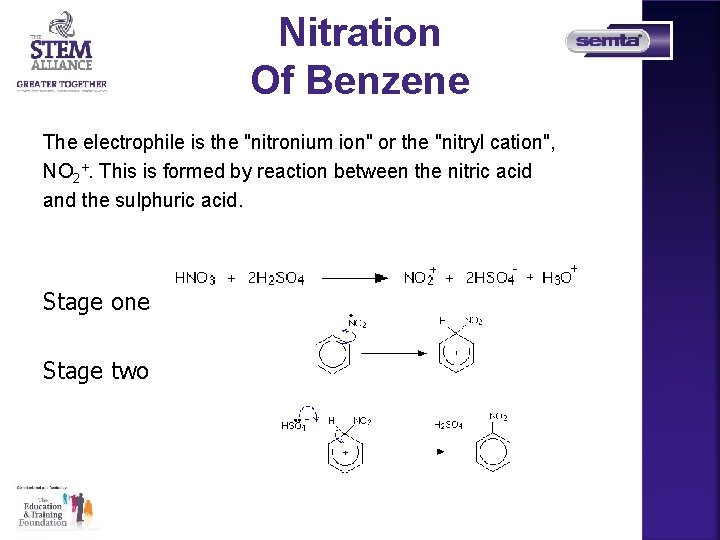
Nitration Of Benzene The electrophile is the "nitronium ion" or the "nitryl cation", NO 2+. This is formed by reaction between the nitric acid and the sulphuric acid. Stage one Stage two

The Halogenation Of Benzene A. Substitution reactions Benzene reacts with chlorine or bromine in an electrophilic substitution reaction, but only in the presence of a catalyst. The catalyst is either aluminium chloride (or aluminium bromide if you are reacting benzene with bromine) or iron. The reactions happen at room temperature
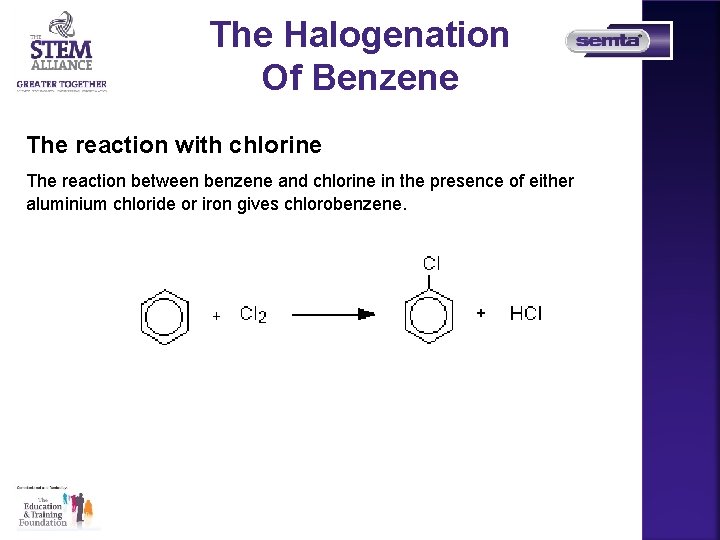
The Halogenation Of Benzene The reaction with chlorine The reaction between benzene and chlorine in the presence of either aluminium chloride or iron gives chlorobenzene.
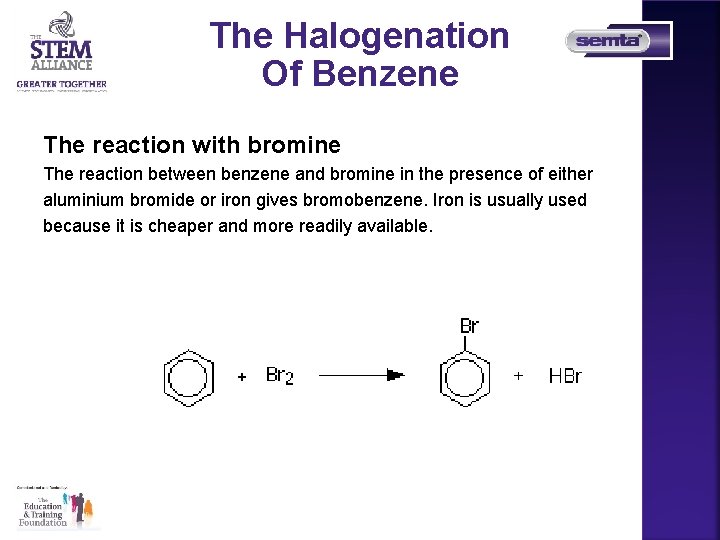
The Halogenation Of Benzene The reaction with bromine The reaction between benzene and bromine in the presence of either aluminium bromide or iron gives bromobenzene. Iron is usually used because it is cheaper and more readily available.
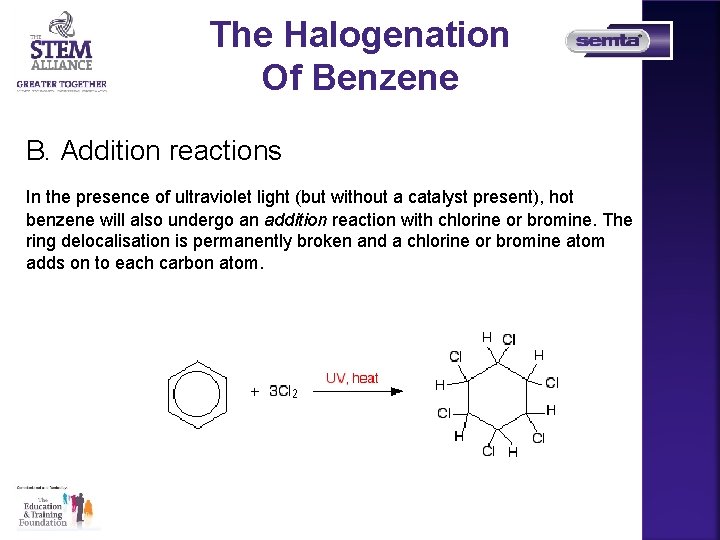
The Halogenation Of Benzene B. Addition reactions In the presence of ultraviolet light (but without a catalyst present), hot benzene will also undergo an addition reaction with chlorine or bromine. The ring delocalisation is permanently broken and a chlorine or bromine atom adds on to each carbon atom.

Friedel-crafts Reactions Of Benzene The Friedel–Crafts reactions are a set of reactions developed by Charles Friedel and James Crafts in 1877. There are two main types of Friedel–Crafts reactions: alkylation reactions and acylation reactions. This reaction type is a form of. The general reaction schemeelectrophilic aromatic substitution is shown below.

What is Acylation? • An acyl group is an alkyl group attached to a carbonoxygen double bond. If "R" represents any alkyl group, then an acyl group has the formula RCO-. Acylation means substituting an acyl group into something - in this case, into a benzene ring. • The most commonly used acyl group is CH 3 CO-. This is called the ethanoyl group, and in this case the reaction is sometimes called "ethanoylation".

What is Acylation? • The most reactive substance containing an acyl group is an acyl chloride (also known as an acid chloride). These have the general formula RCOCl. • Benzene is treated with a mixture of ethanoyl chloride, CH 3 COCl, and aluminium chloride as the catalyst. The mixture is heated to about 60°C for about 30 minutes. • A ketone called phenylethanone (old name: acetophenone) is formed.
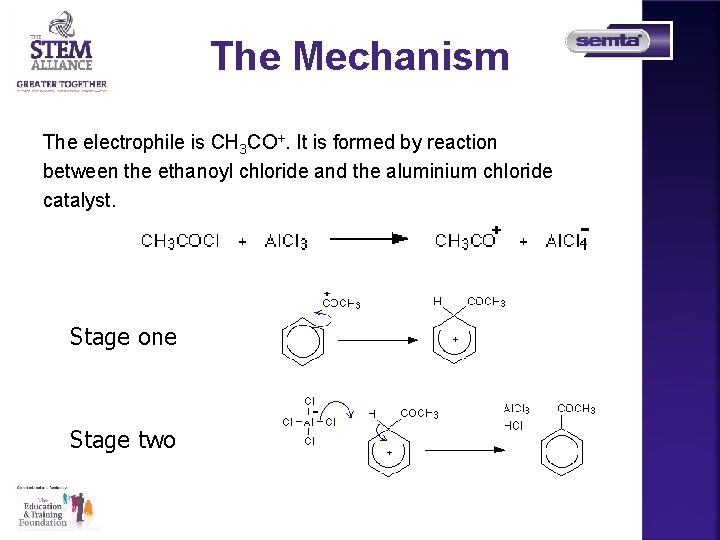
The Mechanism The electrophile is CH 3 CO+. It is formed by reaction between the ethanoyl chloride and the aluminium chloride catalyst. Stage one Stage two

What is Alkylation? Alkylation means substituting an alkyl group into something - in this case into a benzene ring. A hydrogen on the ring is replaced by a group like methyl or ethyl and so on.
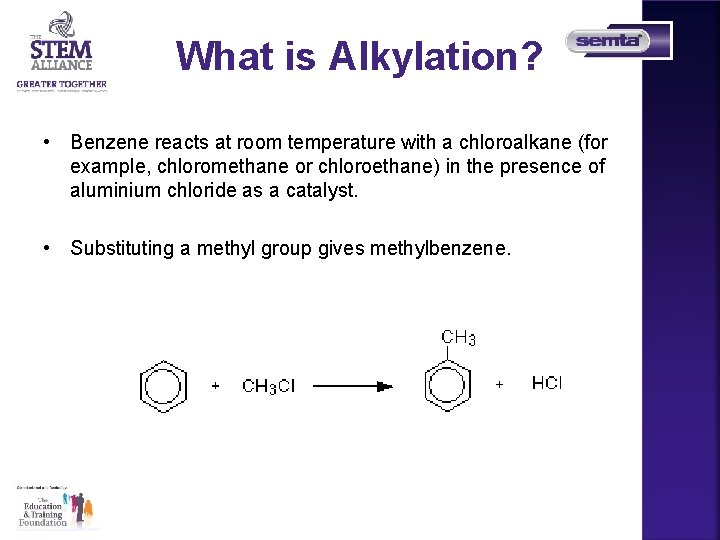
What is Alkylation? • Benzene reacts at room temperature with a chloroalkane (for example, chloromethane or chloroethane) in the presence of aluminium chloride as a catalyst. • Substituting a methyl group gives methylbenzene.
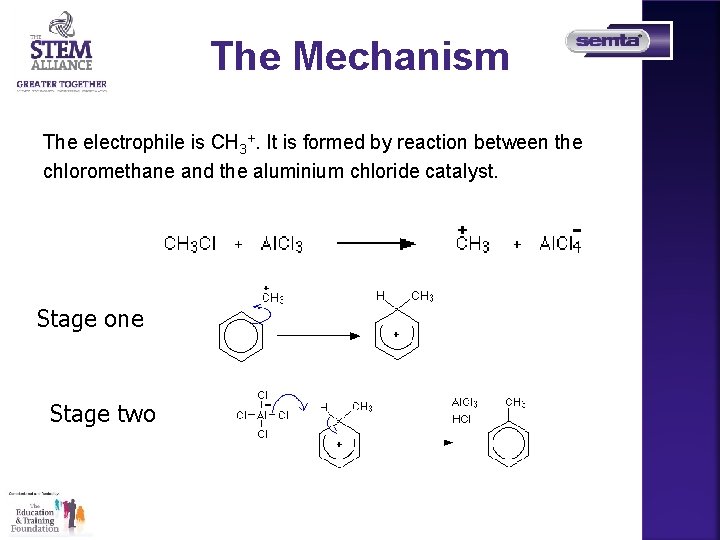
The Mechanism The electrophile is CH 3+. It is formed by reaction between the chloromethane and the aluminium chloride catalyst. Stage one Stage two
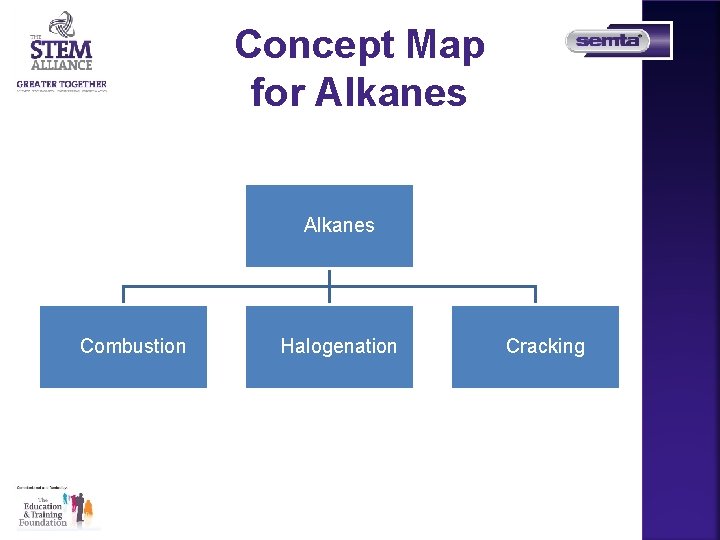
Concept Map for Alkanes FCombustion FHalogenation FCracking
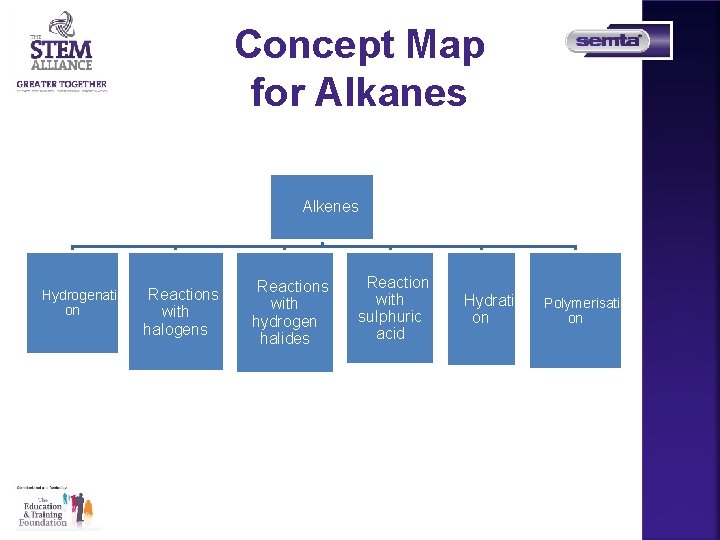
Concept Map for Alkanes FAlkenes FHydrogenati FReactions on with halogens FReactions with hydrogen halides FReaction with sulphuric acid FHydrati FPolymerisati on on

Concept map for Benzene FNitration FHalogenation FFriedel-Crafts reactions

For further information please contact The STEM Alliance enquiries@STEMalliance. uk or visit www. STEMalliance. uk
- Slides: 124