Hydrocarbons Compounds containing hydrogen and carbon only We

Hydrocarbons Compounds containing hydrogen and carbon only We get them from crude oil They are made up of molecules A molecule is made up of 2 or more atoms joined by covalent bonds. Hydrogen is in group 1 and has 1 outer electron Hydrogen can form one bond Carbon is in group 4 and has 4 outer electrons Carbon can form 4 bonds
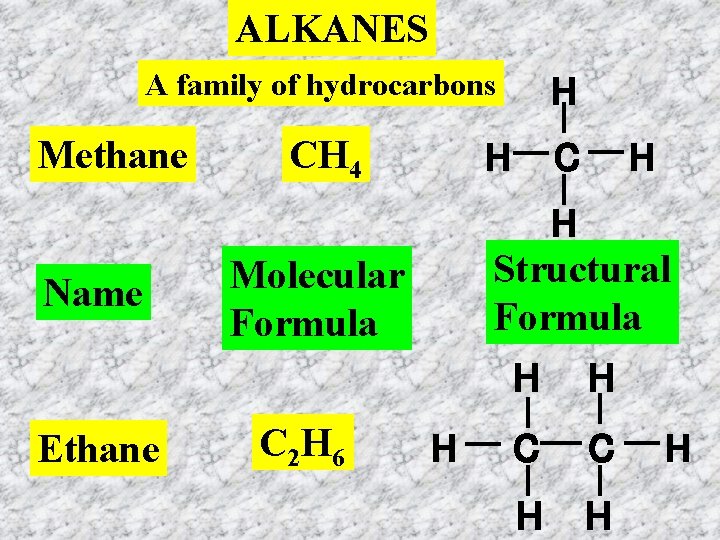
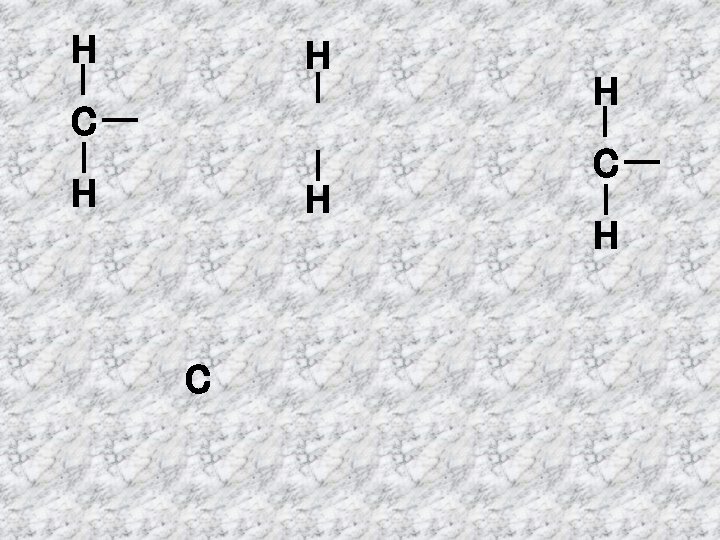
ALKANES A family of hydrocarbons Methane Name Ethane CH 4 H C H H Structural Formula Molecular Formula C 2 H 6 H H C C H H H
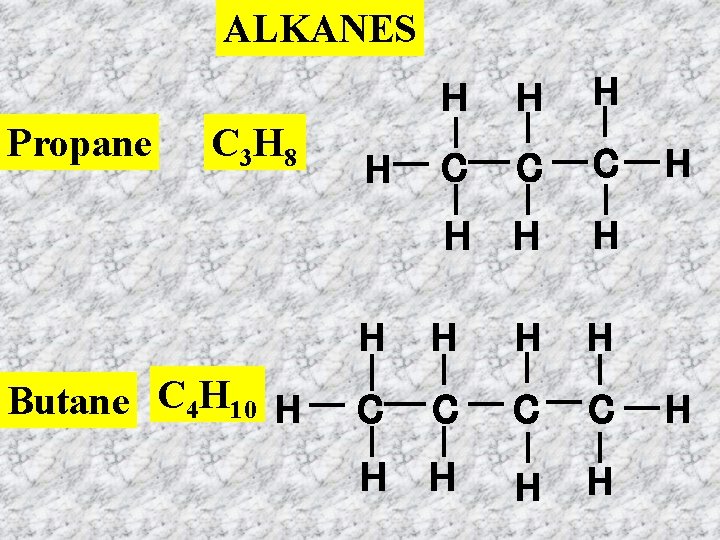
ALKANES Propane C 3 H 8 Butane C 4 H 10 H H H C C C H H H H C C H H H
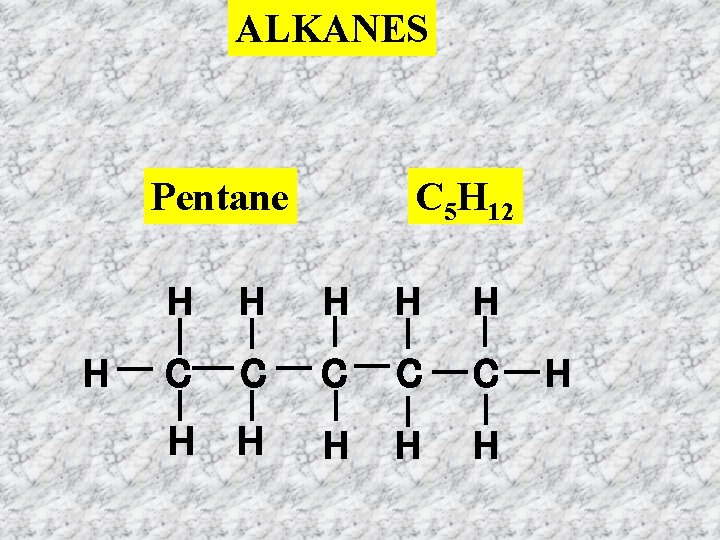
ALKANES Pentane H C 5 H 12 H H H C C C H H H
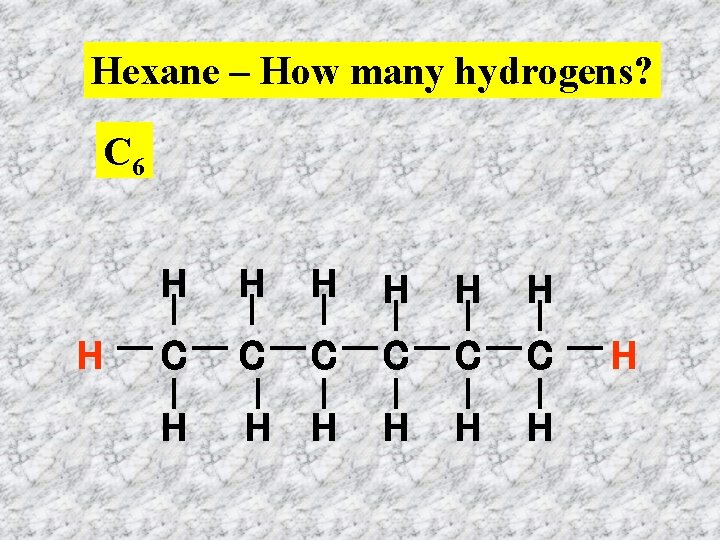
Hexane – How many hydrogens? C 6 H H H H C C C H H H H
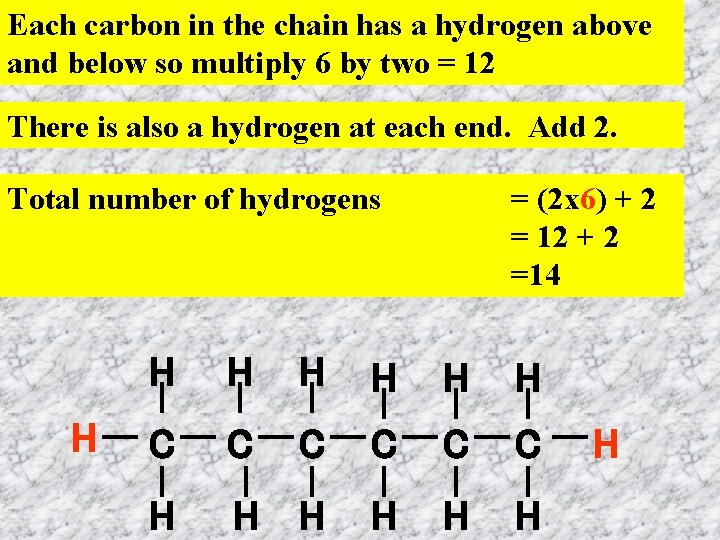
Each carbon in the chain has a hydrogen above and below so multiply 6 by two = 12 There is also a hydrogen at each end. Add 2. Total number of hydrogens H = (2 x 6) + 2 = 12 + 2 =14 H H H C C C H H H H

Give the complete formula for each of these hydrocarbons. You are given the number of carbons. C 8 H 18 C 10 (2 x 8) + 2 = 16 + 2 =18 H 22 (2 x 10) + 2 = 20 + 2 = 22 C 15 H 32 (2 x 15) + 2 = 30 + 2 = 32 C 31 H 64 (2 x 31) + 2 = 62 + 2 = 64 C 100 H 202 (2 x 100) + 2 = 200 + 2 = 202 Cn H 2 n + 2 (2 x n) + 2
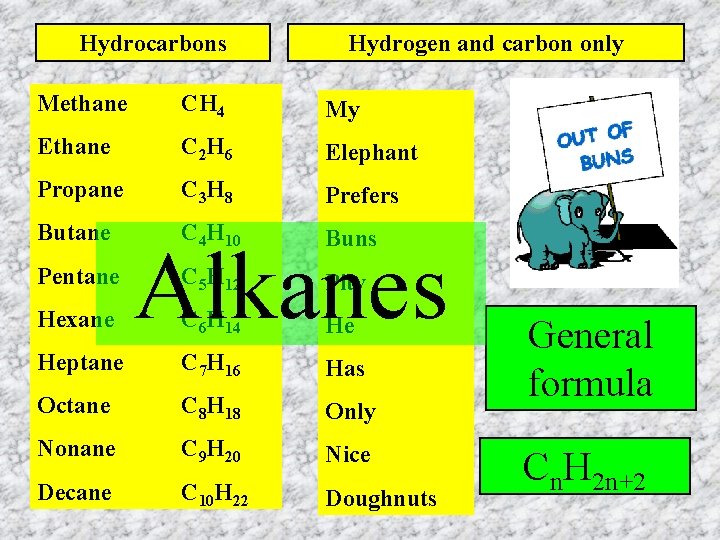
Hydrocarbons Hydrogen and carbon only Methane CH 4 My Ethane C 2 H 6 Elephant Propane C 3 H 8 Prefers Butane C 4 H 10 Buns C 5 H 12 Pity C 6 H 14 He Heptane C 7 H 16 Has Octane C 8 H 18 Only Nonane C 9 H 20 Nice Decane C 10 H 22 Doughnuts Pentane Hexane Alkanes General formula Cn. H 2 n+2
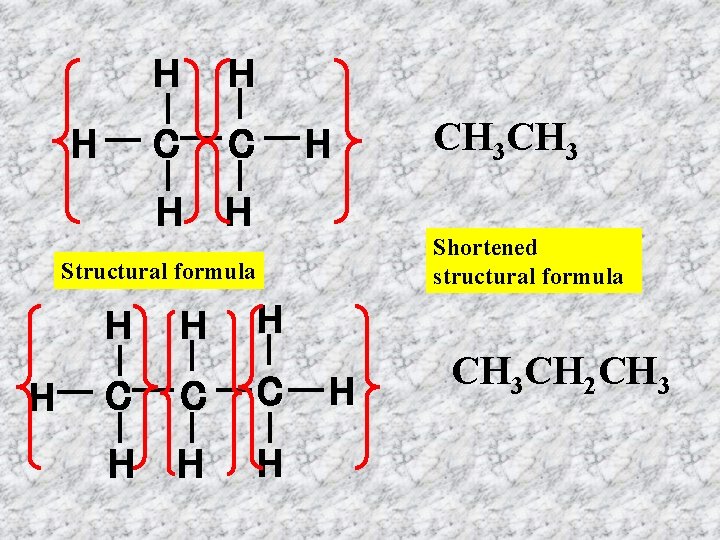
H H H C C H H H Shortened structural formula Structural formula H H H CH 3 H C C C H H CH 3 CH 2 CH 3
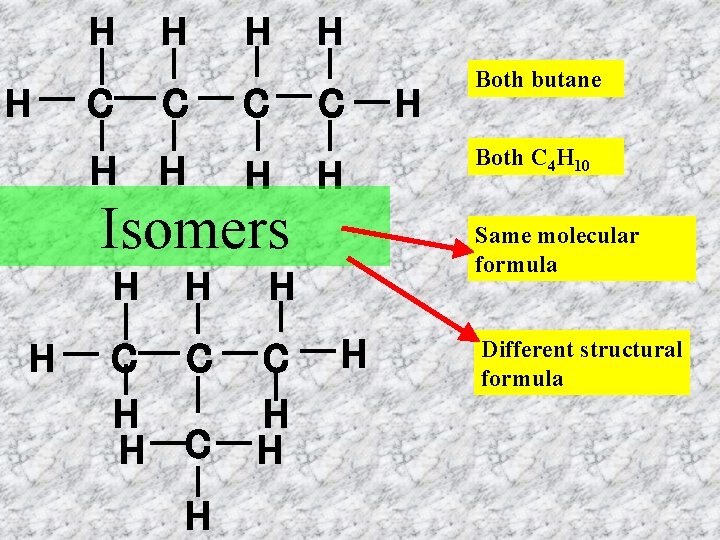
H H C H Isomers H H C C H H H H C H H Both butane Both C 4 H 10 Same molecular formula H Different structural formula

In your jotter: Draw the 3 isomers of pentane At least 3 isomers of hexane
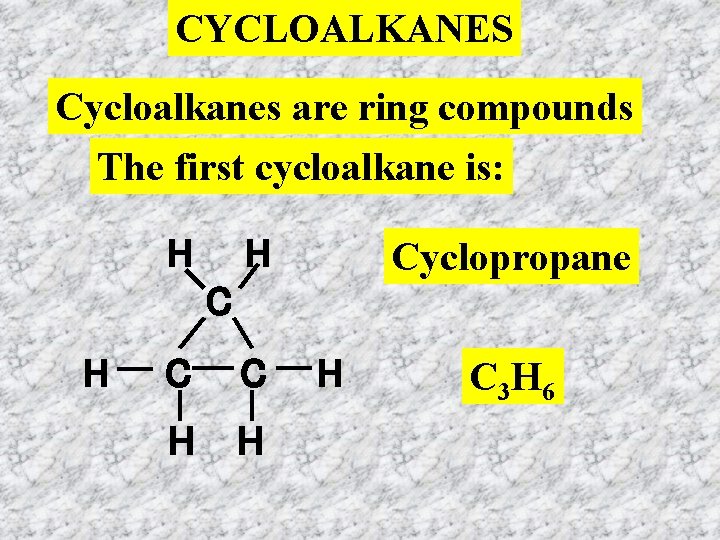
CYCLOALKANES Cycloalkanes are ring compounds The first cycloalkane is: H H Cyclopropane C H C C H H H C 3 H 6
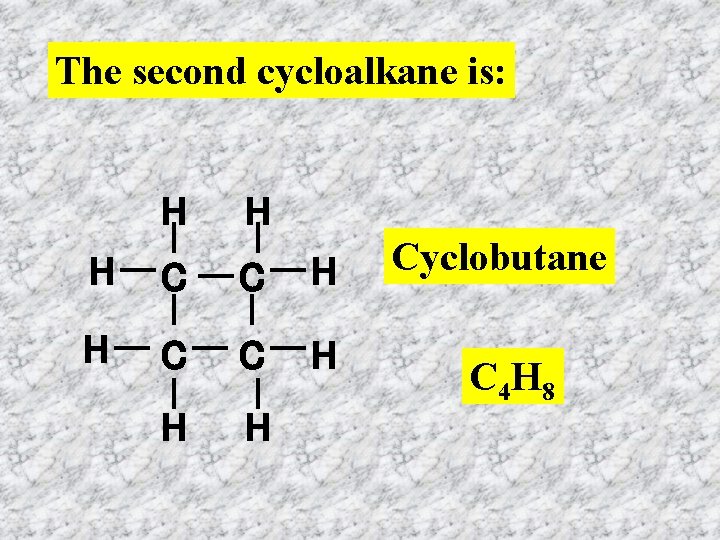
The second cycloalkane is: H H H C C H H H Cyclobutane C 4 H 8
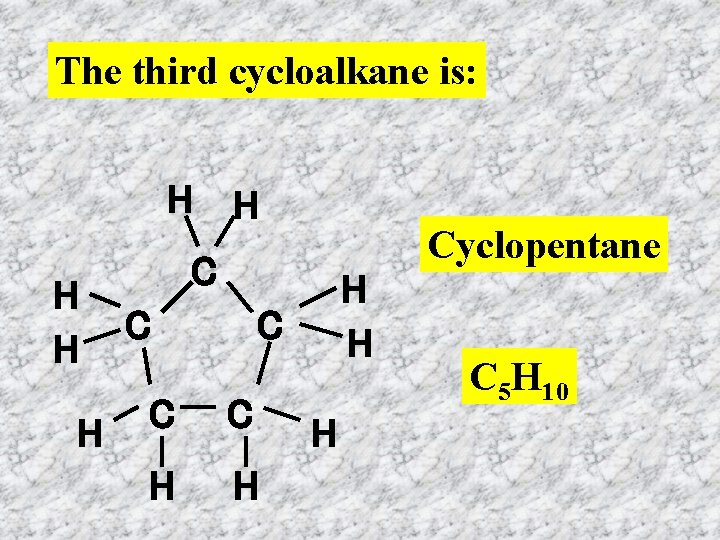
The third cycloalkane is: H H H Cyclopentane C C H H C C C H H H C 5 H 10
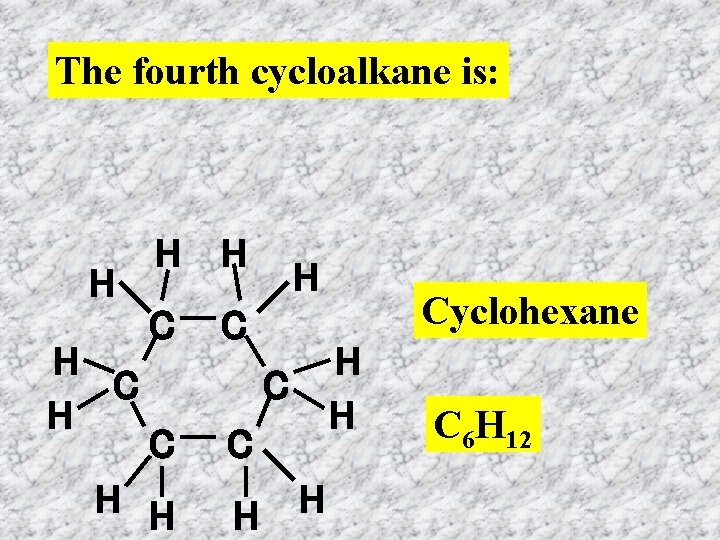
The fourth cycloalkane is: H H C H H H C C C H H H Cyclohexane C 6 H 12

Cycloalkane Molecular Formula Cyclopropane C 3 H 6 2 x 3 = 6 Cyclobutane C 4 H 8 2 x 4 = 8 Cyclopentane C 5 H 10 2 x 5 = 10 Cyclohexane C 6 H 12 2 x 6 = 12 Cycloheptane C 7 H 14 2 x 7 = 14 Cyclooctane C 8 H 16 2 x 8 = 16 General formula Cn. H 2 n
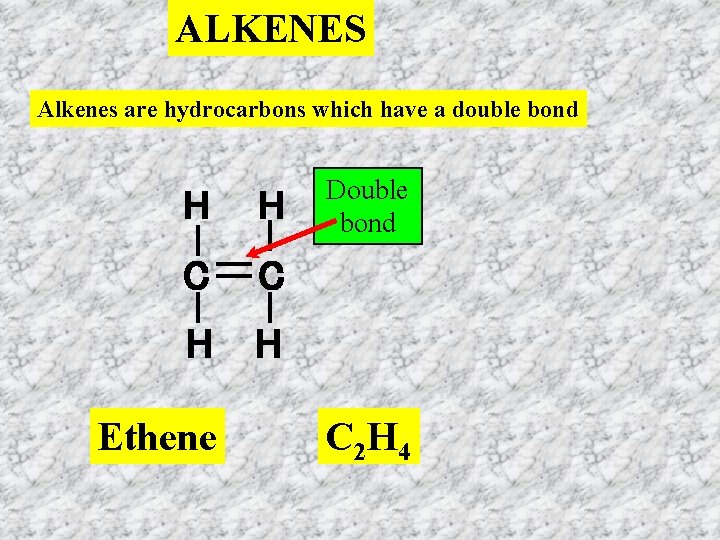
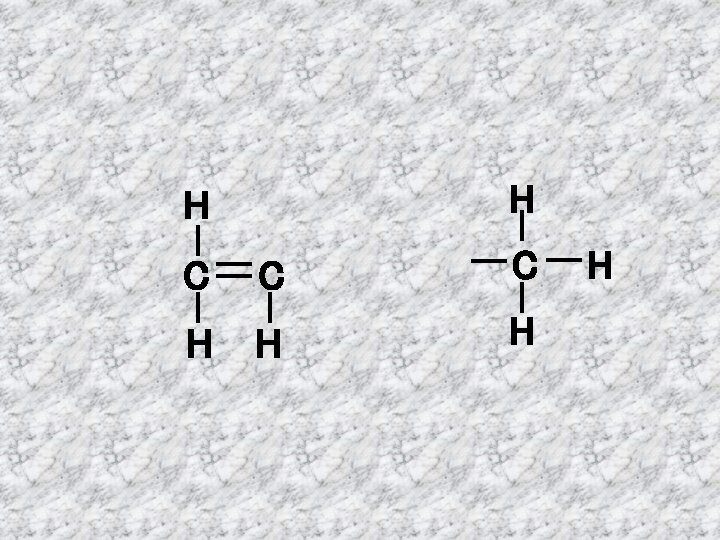
ALKENES Alkenes are hydrocarbons which have a double bond H H C C H H Ethene Double bond C 2 H 4
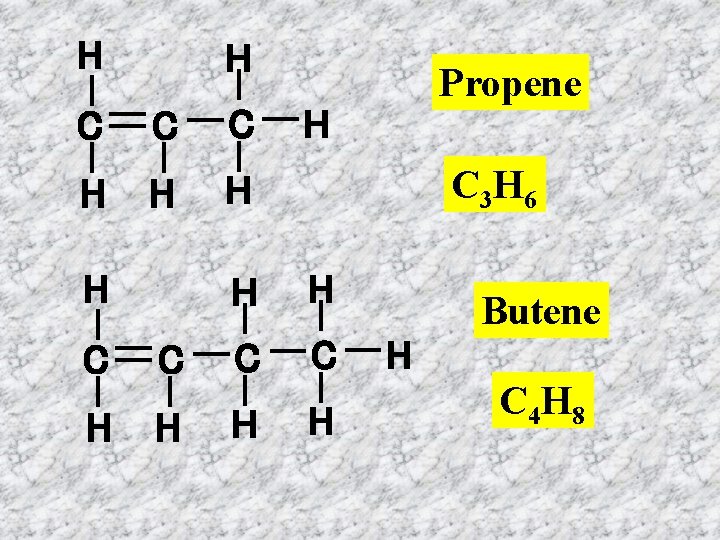
H H C C C H H H C 3 H 6 H H C C H H H Propene H Butene H C 4 H 8

Alkene Molecular Formula Ethene C 2 H 4 2 x 2 = 4 Propene C 3 H 6 2 x 3 = 6 Butene C 4 H 8 2 x 4 = 8 Pentene C 5 H 10 2 x 5 = 10 Hexene C 6 H 12 2 x 6 = 12 General formula Cn. H 2 n

1 Write the molecular formula of: a) the alkane with 6 carbons b) the alkane with 24 hydrogens c) the cycloalkane with 20 carbons d) the alkene with 100 hydrogens 2 Draw the structural formula of: a) the alkene with 5 carbons b) the cycloalkane with 7 carbons 3 Give the general formula of a) the alkanes b) the alkenes c) the cycloalkanes 4 Copy and complete: a) Isomers have the same ____ formula but different ___ formula b) Draw 2 isomers of heptane and 2 isomers of butene and 2 isomers of cyclopropane 5 What is meant by a hydrocarbon? 6 What is meant by a molecule? 7 What is produced when a hydrocarbon burns? 8 Which would have higher boiling point propane or pentane? 9 Which would be more flammable octene or butene? 10 Which would be more viscous cyclopropane or cyclooctane?
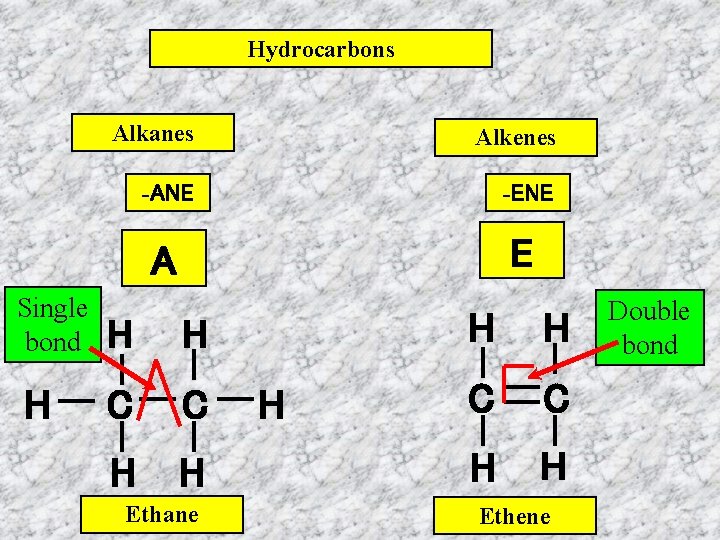
Hydrocarbons Alkanes Alkenes -ANE -ENE A E Single bond H H H C C H H Ethane H H H C C H H Ethene Double bond

The Bromine Water Test Place a small amount of each hydrocarbon in a test tube. To each add a little bromine water. Bromine water is a brownish colour and when dilute is yellowish. Set out your results by drawing up a table as shown below Hydrocarbon Hexane Hexene Heptane Octane Hexane Cyclohexane Formula C 6 H 14 C 6 H 12 C 7 H 16 C 8 H 18 C 6 H 14 C 6 H 12 Result of test
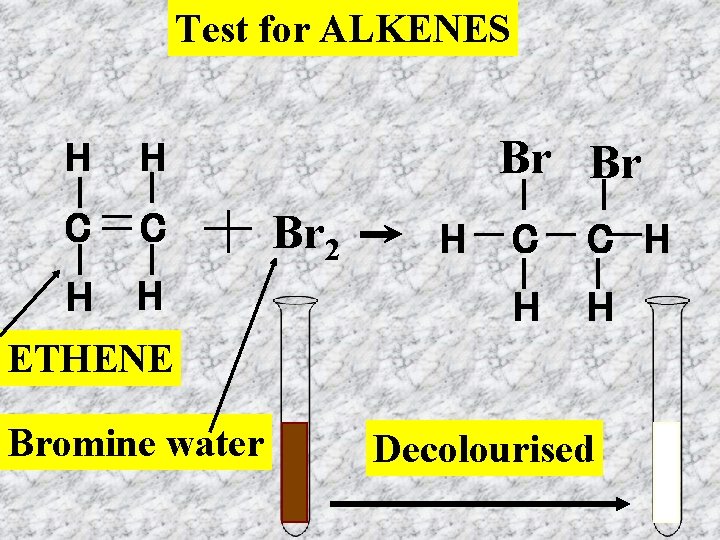
Test for ALKENES H H C C H H Br Br Br 2 H C C H H H ETHENE Bromine water Decolourised

1 Write out full structural formulae to show the reaction between: a) Propene and bromine water b) Butene and bromine water 2 Complete the following equations by putting in the missing compounds (first one is done for you). a) b) c) d) e) C 5 H 10 (pentene) + Br 2 C 6 H 12 (hexene) + Br 2 _____ (_______) + Br 2 C 4 H 8 (butene) + Cl 2 C 5 H 10 (pentene) + ____ C 5 H 10 Br 2 C 6 H 12 Br 2 C 8 H 16 Br 2 _____ C 5 H 12
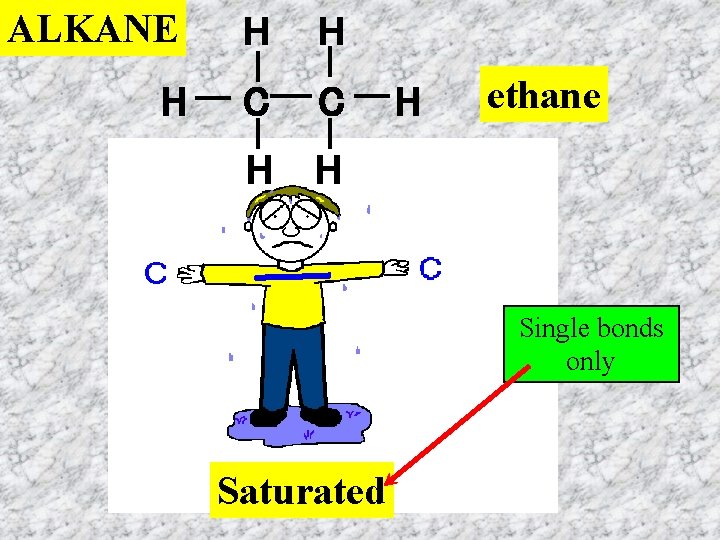
ALKANE H H H C C H H H ethane Single bonds only Saturated
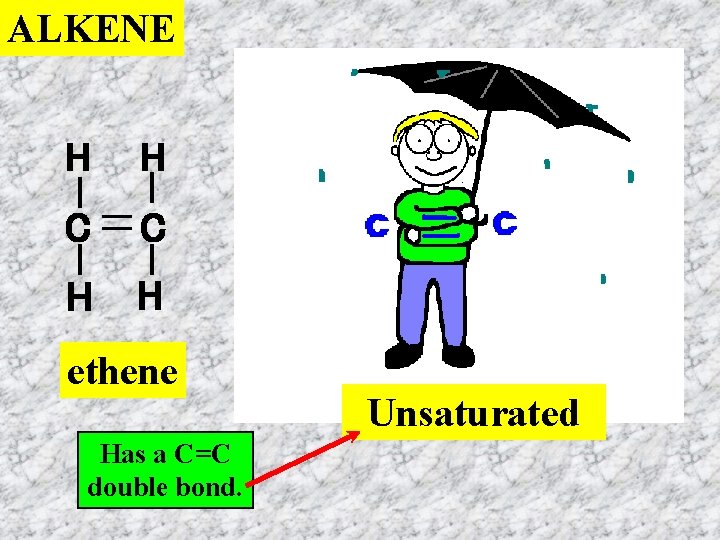
ALKENE H H C C H H ethene Has a C=C double bond. Unsaturated
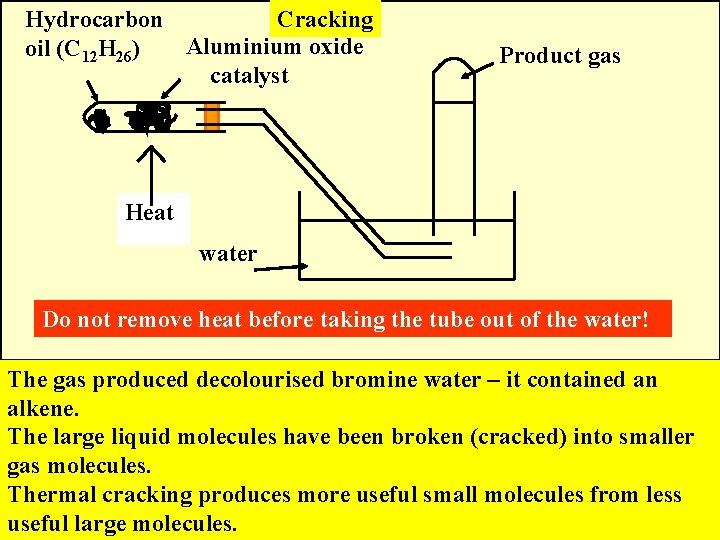
Hydrocarbon Cracking Aluminium oxide oil (C 12 H 26) catalyst Product gas Heat water Do not remove heat before taking the tube out of the water! The gas produced decolourised bromine water – it contained an alkene. The large liquid molecules have been broken (cracked) into smaller gas molecules. Thermal cracking produces more useful small molecules from less useful large molecules.
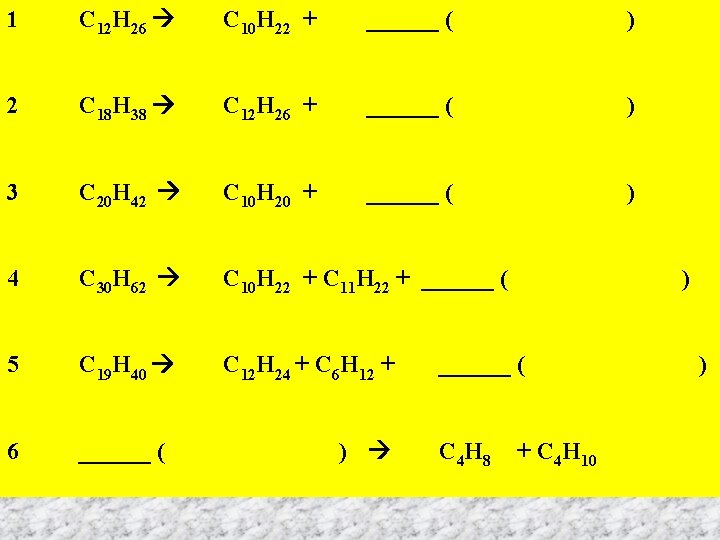
1 2 3 4 5 6 C 12 H 26 C 10 H 22 + ______ ( ) C 18 H 38 C 12 H 26 + ______ ( ) C 20 H 42 C 10 H 20 + ______ ( ) C 30 H 62 C 10 H 22 + C 11 H 22 + ______ ( ) C 19 H 40 C 12 H 24 + C 6 H 12 + ______ ( ) C 4 H 8 + C 4 H 10
H H C H C H
H H C C C H H
- Slides: 31