Hydrocarbon Derivatives Halocarbons Alcohols Ethers Hydrocarbons Contain only

Hydrocarbon Derivatives: Halocarbons, Alcohols, & Ethers

Hydrocarbons • Contain only carbon & hydrogen • But carbon can form strong covalent bonds to other elements, incl. O, N, F, Cl, Br, I, S, & P

Functional Group • One or more H’s in a hydrocarbon can be replaced by an atom or group of atoms. • An atom or group of atoms in an organic molecule that always behaves in the same way is called a functional group. • Adding a functional group changes the chemical & physical properties in specific ways, depending on the functional group.

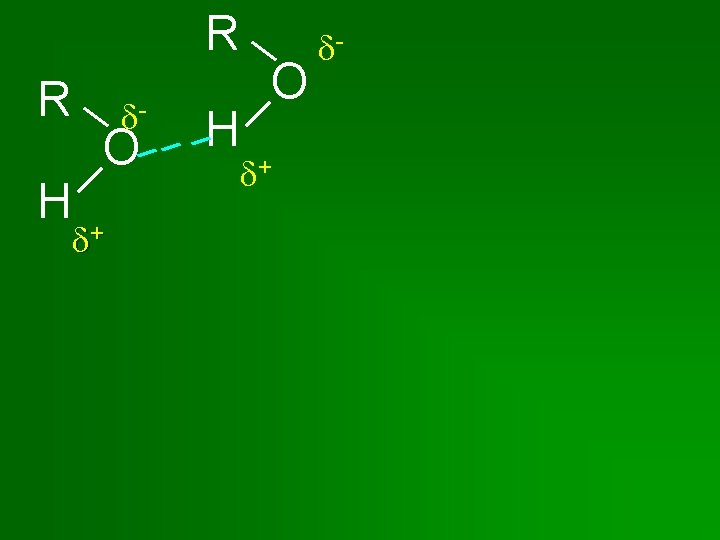
Intermolecular Forces • Determine Boiling Point & Solubility • Van der Waals or dispersion – nonpolar – weakest. • Dipole-dipole – intermediate. Molecule must have atoms with different electronegativities & not arranged symmetrically. • Hydrogen bonding – strongest. Molecules must contain H bonded to F, O, or N.

Functional Groups • • Halocarbons Alcohols Ethers Amines Aldehydes Ketones Carboxylic Acid Ester • Amide • Amino Acid


Organic Halides • One or more of the hydrogen atoms in an alkane is replaced with a halogen (F, Cl, Br, or I). • Not hydrocarbons! Often called halocarbons or alkyl halides.

Naming Halides • Figure out the base name. • Use prefixes to specify substituent: fluoro, chloro, bromo, iodo • If more than one, use di, tri, etc. to specify # of substituents. • If necessary, give locations of halogens by numbering C-atoms in backbone.
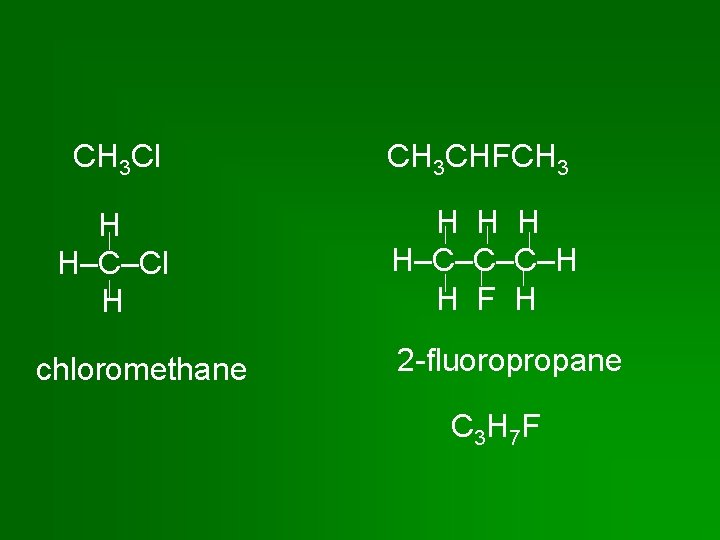
CH 3 Cl CH 3 CHFCH 3 H H–C–Cl H H H–C–C–C–H H F H chloromethane 2 -fluoropropane C 3 H 7 F
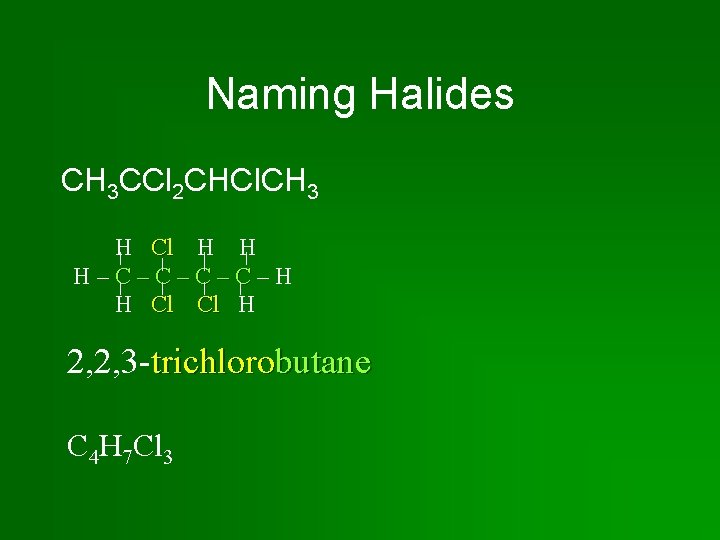
Naming Halides CH 3 CCl 2 CHCl. CH 3 H Cl H H H–C–C–H H Cl Cl H 2, 2, 3 -trichlorobutane C 4 H 7 Cl 3

Ranking Halogens • If more than 1 kind of halogen atom, list them alphabetically. • Chain is numbered to give lowest number to halogen that comes first in alphabet.
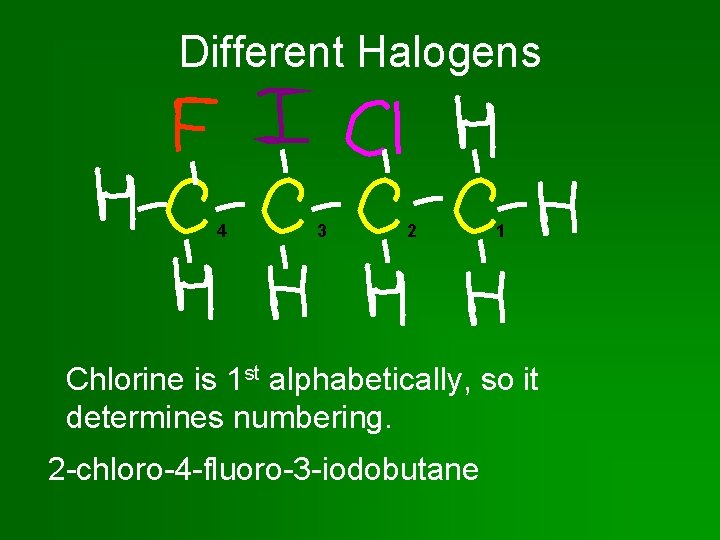
Different Halogens 4 3 2 1 Chlorine is 1 st alphabetically, so it determines numbering. 2 -chloro-4 -fluoro-3 -iodobutane
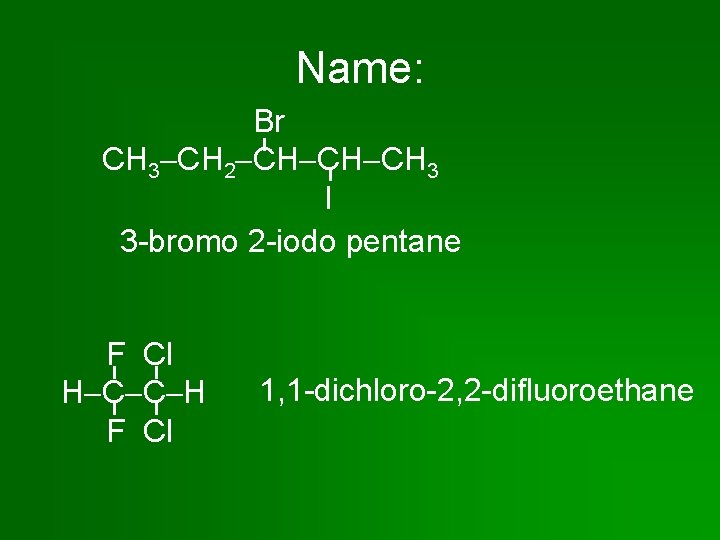
Name: Br CH 3 CH 2 CH CH CH 3 I 3 -bromo 2 -iodo pentane F Cl H C C H F Cl 1, 1 -dichloro-2, 2 -difluoroethane

Properties of Halocarbons • For an alkane & an alkyl halide of similar size & shape, the alkyl halide has the higher boiling point & higher density. Why? Stronger intermolecular forces. • CH 4: bp = -162 C & density = 0. 423 g/ml • CH 3 Cl: bp = -24 C & density = 0. 911 g/ml What intermolecular forces for CCl 4?
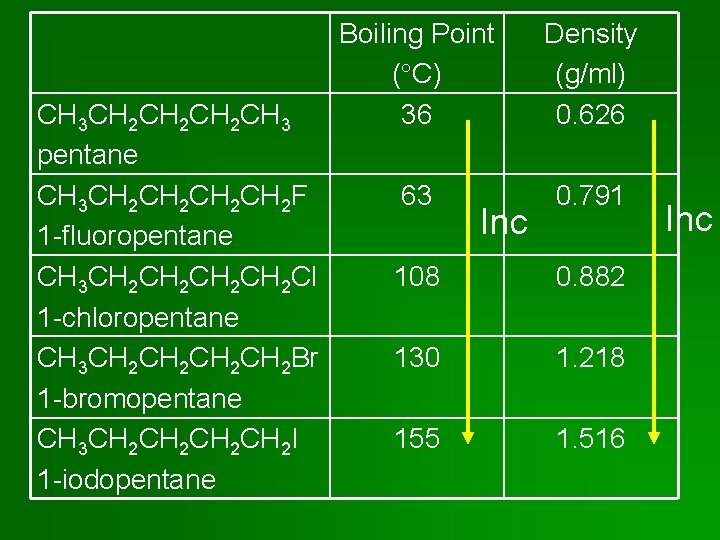
CH 3 CH 2 CH 2 CH 3 pentane CH 3 CH 2 CH 2 F 1 -fluoropentane CH 3 CH 2 CH 2 Cl 1 -chloropentane CH 3 CH 2 CH 2 Br 1 -bromopentane CH 3 CH 2 CH 2 I 1 -iodopentane Boiling Point ( C) 36 Density (g/ml) 0. 626 63 0. 791 Inc 108 0. 882 130 1. 218 155 1. 516 Inc

Uses of Alkyl Halides • Cleaners & solvents • Teflon & PVC’s are alkyl halides. • Refrigerants. (used to be chlorofluorocarbons. Now hydrofluorocarbons. ) • Starting materials in many reactions.

Halogen Derivatives • • • CH 3 Cl = local anesthetic CHCl 3 = solvent, general anesthetic CHI 3 = antiseptic CCl 4 = dry cleaning solvent CF 2 Cl 2 = refrigerant Fluorocarbons = teflon, lubricants, synthetic blood • Chlorofluorocarbons = aerosol propellants, refrigerants

Table R • General Formula for halocarbons: R-X • R represents the entire hydrocarbon part of the molecule. (The alkyl part. ) • X represents the halogen (F, Cl, Br, or I).

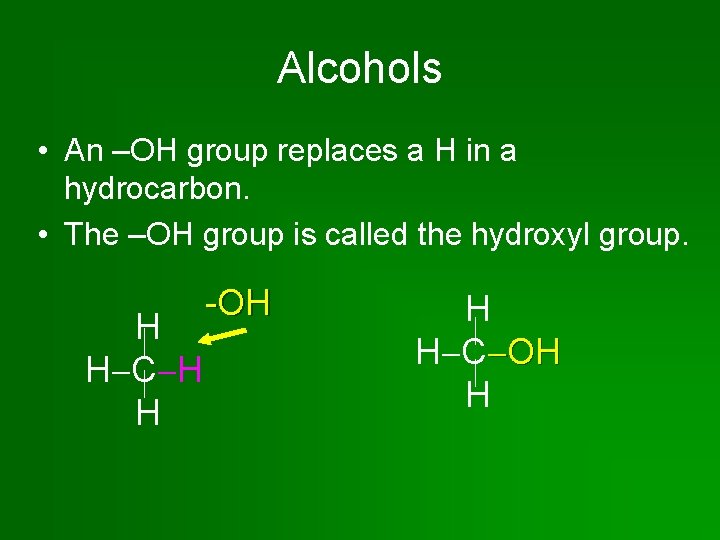
Alcohols • An –OH group replaces a H in a hydrocarbon. • The –OH group is called the hydroxyl group. H H C H H -OH H H C OH H

Alcohols are nonelectrolytes! non • The hydroxyl group resembles the hydroxide ion of inorganic bases, but it does not form ions in water! • The hydroxyl group is polar. So alcohols are soluble in water.

Naming Alcoh. OLs OL • Based on alkane name. • Name the parent chain. – Drop the –e and add –OL. OL • If the parent chain has 3 or more C atoms, number the C’s & give the location of the – OH group.
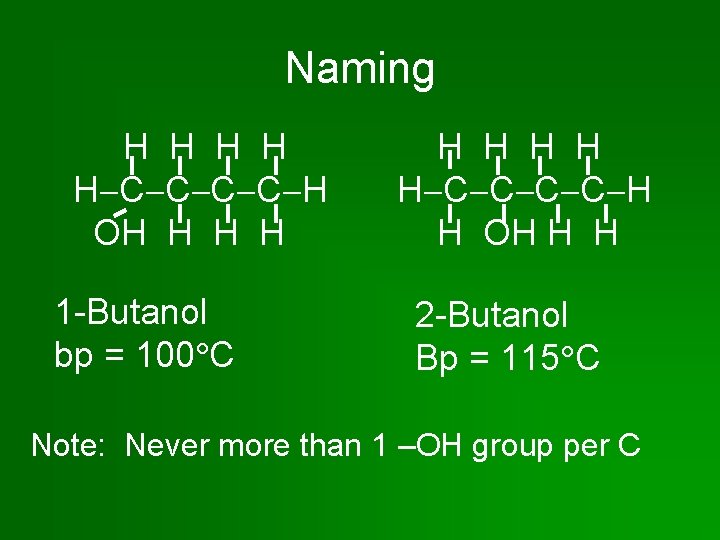
Naming H H H C C H OH H 1 -Butanol bp = 100 C H H H C C H H OH H H 2 -Butanol Bp = 115 C Note: Never more than 1 –OH group per C

More than 1 hydroxyl group • Prefixes di-, tri-, tetra- used before the –ol ending to tell the # of hydroxyl groups. • So don’t drop the -e from the alkane name. • These hydroxyls are on different C atoms!

Classifying Alcohols • By # of hydroxyl groups – Monohydroxy: 1 hydroxyl group – Dihydroxy: 2 “ “ – Trihydroxy: 3 “ “ • By position of each hydroxyl group on main carbon chain.

Monohydroxy Alcohols • Primary: Primary hydroxyl group attached to end C of chain or branch. • Secondary: Secondary hydroxyl group attached to C in a chain – C is bonded to 2 other C’s. • Tertiary: Tertiary hydroxyl group attached to C at a branch point – C is bonded to 3 other C’s.
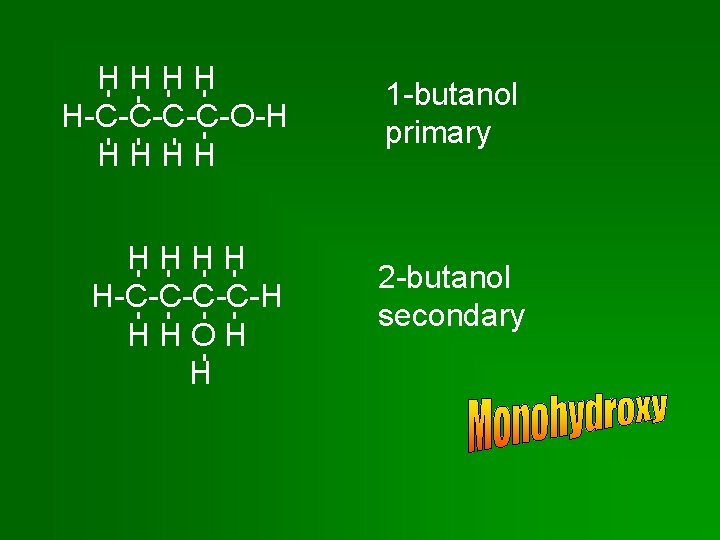
HHHH H-C-C-O-H HHHH H-C-C-H HHOH H 1 -butanol primary 2 -butanol secondary
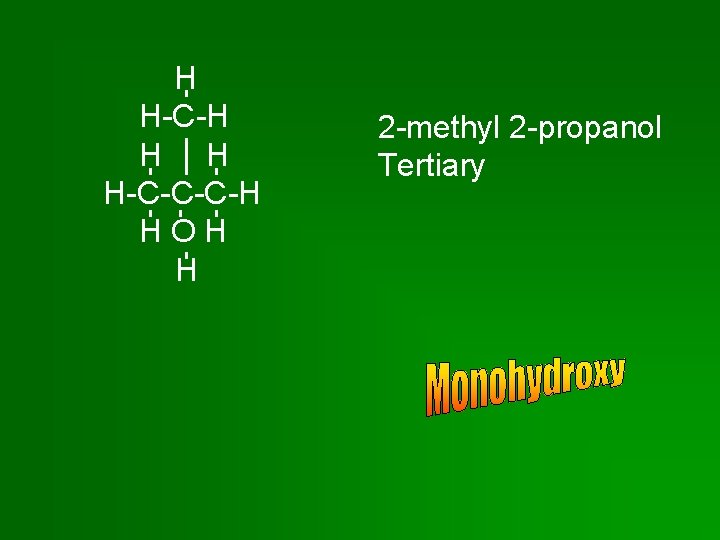
H H-C-H H H H-C-C-C-H HOH H 2 -methyl 2 -propanol Tertiary
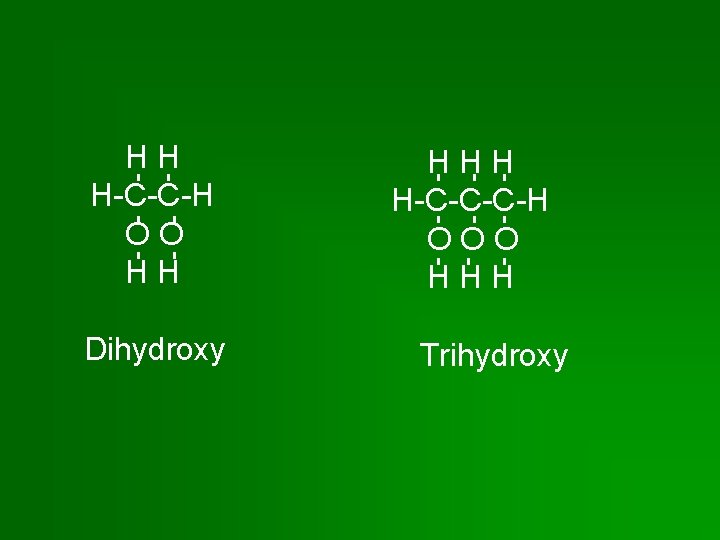
HH H-C-C-H OO HH Dihydroxy HHH H-C-C-C-H OOO HHH Trihydroxy

Properties of Alcohols • Contain a H bonded to an O atom. • Therefore Hydrogen Bonding occurs • Alcohols have a higher boiling point than the corresponding alkane. • Like dissolves Like Alcohols tend to be very soluble in water.

Which compound has the highest boiling point? A. B. C. D. CH 4 C 2 H 6 C 3 H 8 C 3 H 7 OH Correct response = D.

Table R • General Formula for Alcohols: R OH. • R represents the entire hydrocarbon part of the molecule. • OH is the hydroxyl group.


Ethers • General formula is R O R where R may or may not be the same as R. • R and R are hydrocarbon branches. • O is an oxygen bridge. • Ethers are not linear. They are bent, like water.

Properties of Ethers • In a pure ether, no hydrogen bonding – no H bonded to O. Do have weak dipole interactions – bent, like H 2 O. • Dipole-dipole interactions are between dispersion forces & hydrogen bonding. • Ethyl ether once used as anesthesia

Properties of Ethers • Compared to alkanes: – Higher boiling pts than similar alkanes. – More soluble in water than alkanes. • Compared to alcohols: – Lower boiling pts than similar alcohols. – Much less soluble in water than alcohols.

Naming Ethers • If the 2 hydrocarbon branches are identical, name the branch (once) & add the word ether. • If the 2 branches are different, list them in alphabetical order followed by the word ether.

H H H C O C H H H Methyl Ether H H H H C C C O C C C H H H H Propyl Ether
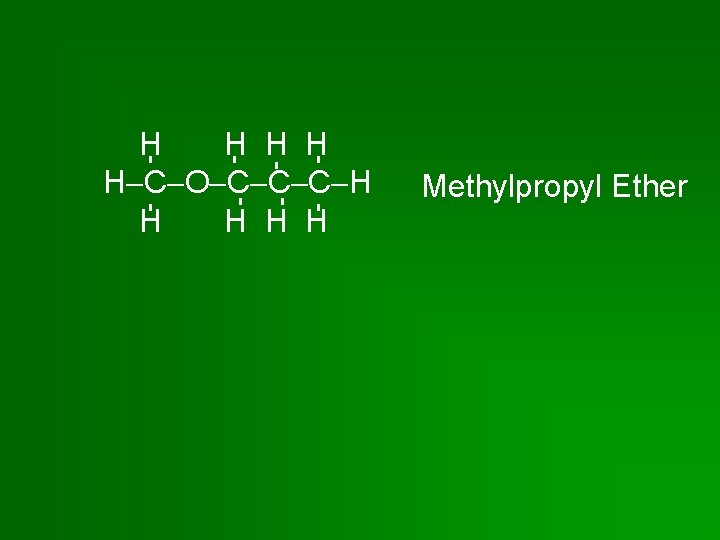
H H H C O C C C H H H Methylpropyl Ether
- Slides: 40