Hydrocarbon Derivatives Amines Amides Amino Acids Amines Contain

Hydrocarbon Derivatives Amines, Amides, & Amino Acids


Amines • Contain N • General Format: R NH 2 – NH 2 = amino group – N: bonding capacity of 3 • Molecules: H bonded to N = H bonding – weaker than H-bonding in alcohols & carboxylic acids • Amines smell really, really bad

Table R R’ • Shows format R N R” (this is a 3 amine) H • 1 amines: R N H H • 2 amines: R N R’

Naming Amines • Find name of base hydrocarbon • Drop final -e & add -amine • # in front of name gives location of amino group
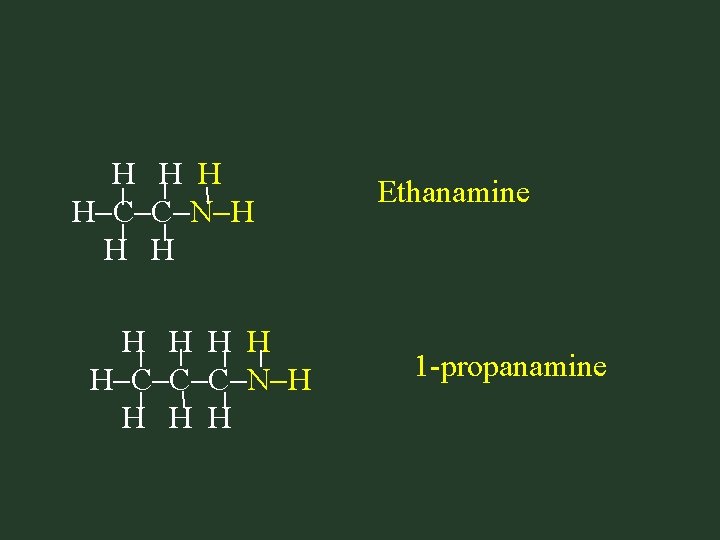
H H C C N H H H C C C N H H Ethanamine 1 -propanamine
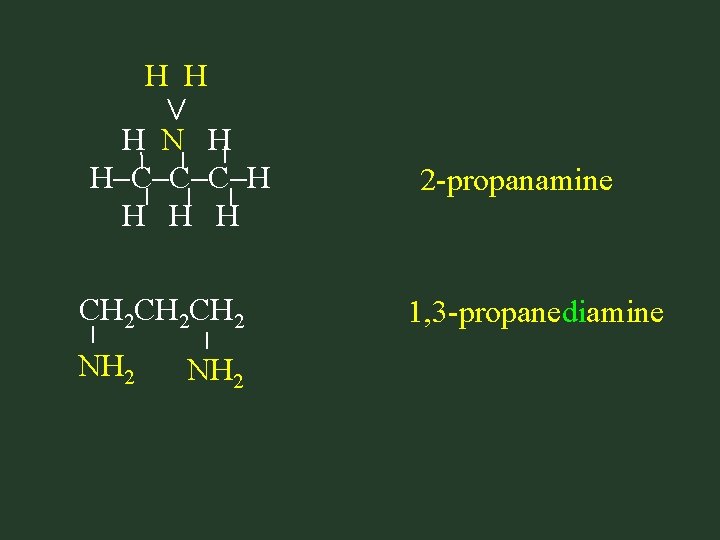
< H H H N H H C C C H H NH 2 CH 2 CH 2 NH 2 2 -propanamine 1, 3 -propanediamine
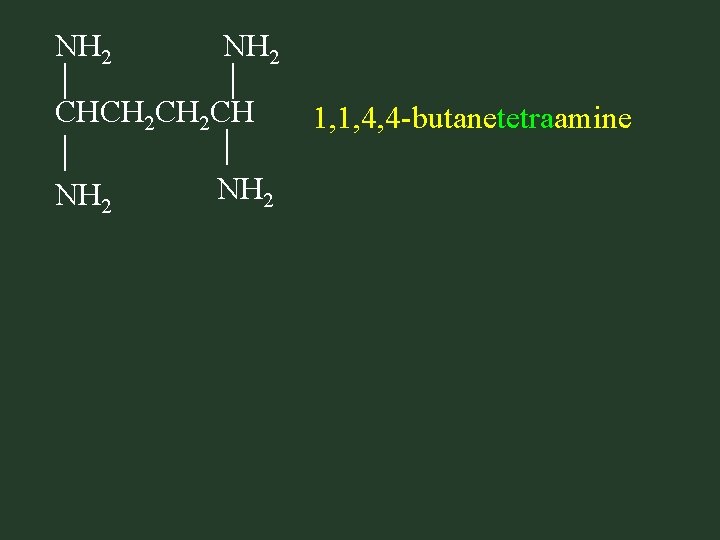
NH 2 CHCH 2 CH NH 2 1, 1, 4, 4 -butanetetraamine

Examples of amines • • • PABA Caffeine Novacain B-vitamins Preparation of dyes



Amides • Contain: – carbonyl group (>C=O) AND – amino group (NH 2) • amino group bonded to C of carbonyl group • Links amino acids together
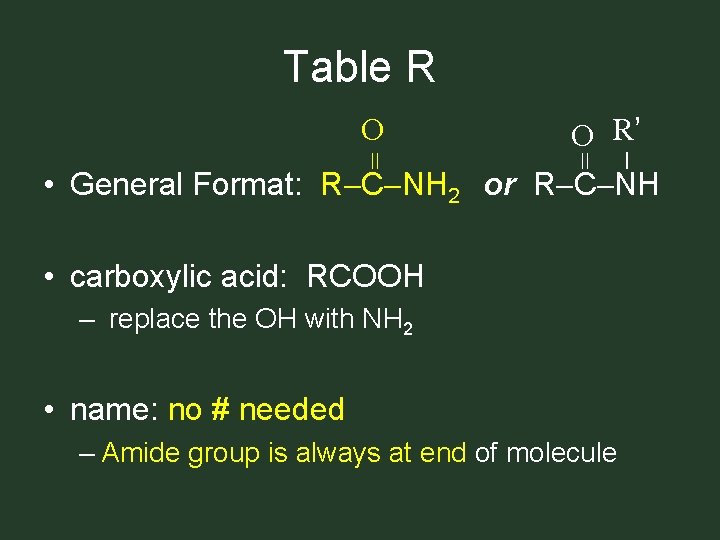
Table R O R’ = = O • General Format: R C NH 2 or R C NH • carboxylic acid: RCOOH – replace the OH with NH 2 • name: no # needed – Amide group is always at end of molecule

Naming Amides • Find name alkane with same # of C’s – drop the final -e & replace it with -amide
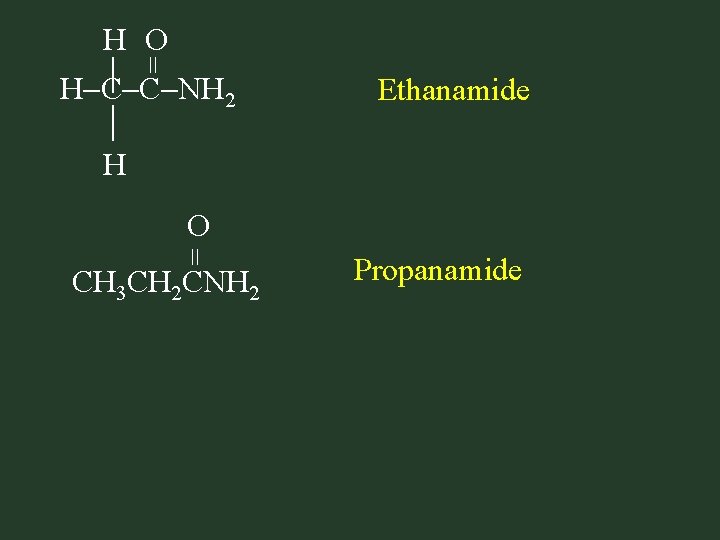
= H O H C C NH H 2 Ethanamide = O CH 3 CH 2 CNH 2 Propanamide

Properties of Amides • strong hydrogen bonding - so high bp • Simple amides are solids at room temp • Smaller amides are very soluble in water



Amino Acids • Only need to recognize them • Don’t need to name them – Contain carboxyl group (-COOH) AND – Contain amine group (-NH 2) – Amino group is on C next to carboxyl group
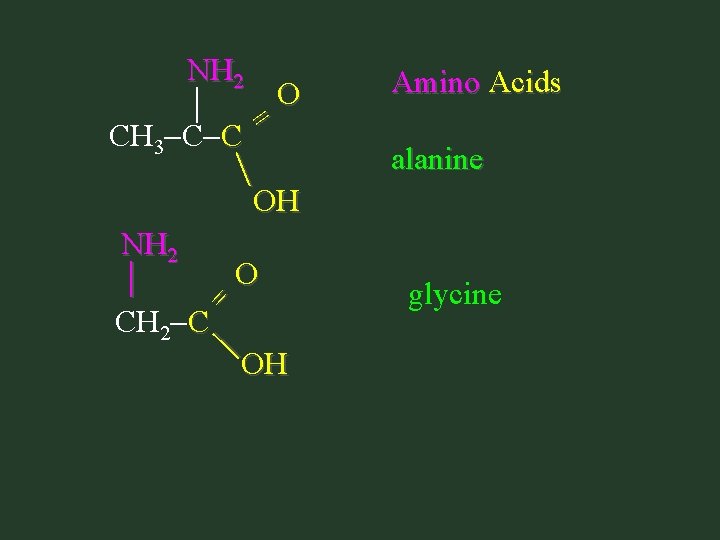
= NH 2 O CH 3 C C = OH NH 2 O CH 2 C OH Amino Acids alanine glycine
- Slides: 20