Hydrocarbon Derivatives Aldehydes Ketones Carboxylic Acids Esters Carbonyl

Hydrocarbon Derivatives Aldehydes, Ketones, Carboxylic Acids, & Esters


Carbonyl Group • >C O • Oxygen attached to a carbon by a double covalent bond. • Strong dipole

Aldehydes Al O • General formula: R C H or RCHO • Carbonyl group is always at the end of the aldehyde (C on one side, H on the other). • Find the name of the alkane with the same # of carbons. Change the final e to -al. al • Never need #’s for aldehydes – always at end.
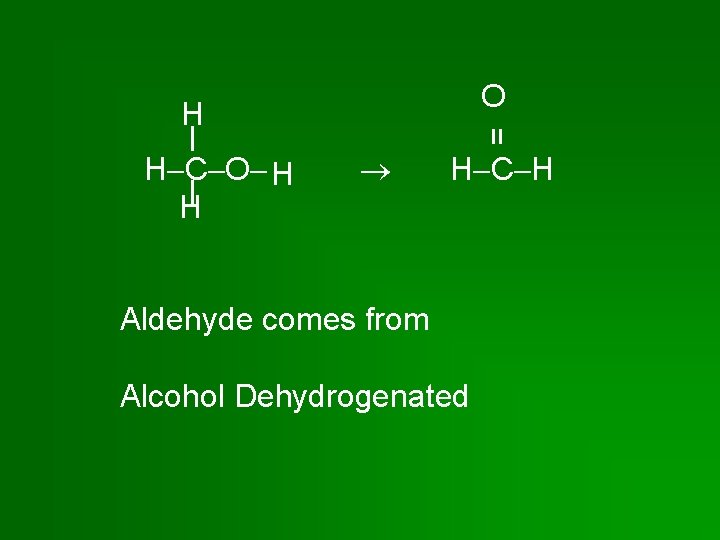
O H C O H H = H H C H Aldehyde comes from Alcohol Dehydrogenated
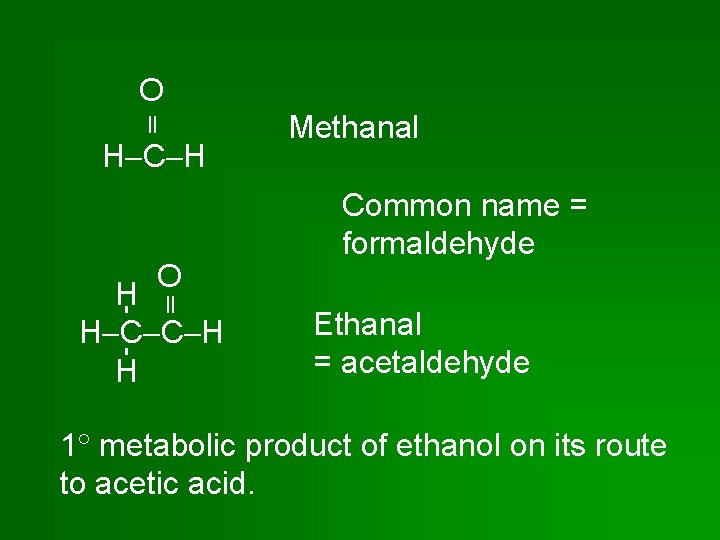
O H C H O H H C C H H Methanal Common name = formaldehyde Ethanal = acetaldehyde 1 metabolic product of ethanol on its route to acetic acid.

Acetaldehyde • Evidence that it is carcinogenic. • Natural component of many ripe fruits. • Contributes to odors of rosemary, daffodil, bitter orange, camphor, angelica, fennel, mustard, & peppermint. • A component of cigarette smoke.

CH 3 CH 2 CHO Ends in CHO so it’s an aldehyde. 4 Carbons so base name is butane. Drop final -e and add –al butanal.
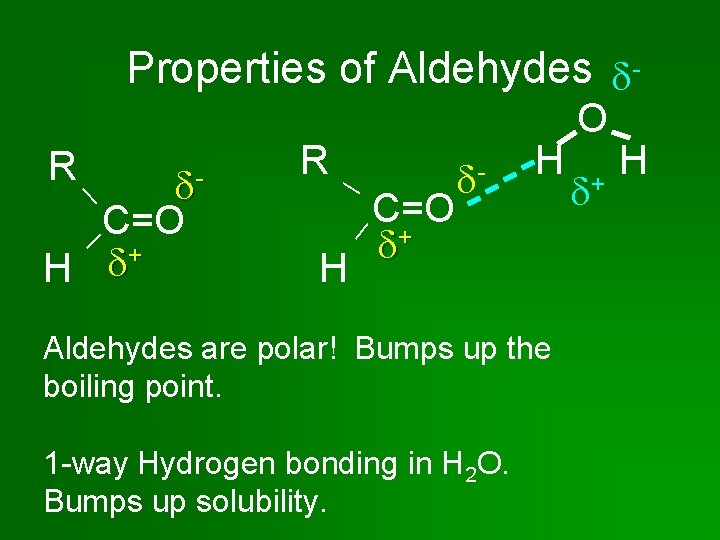
Properties of Aldehydes - C=O + H R R H C=O + - H Aldehydes are polar! Bumps up the boiling point. 1 -way Hydrogen bonding in H 2 O. Bumps up solubility. O + H


Ketones • Carbonyl group , >C=O, located within a C chain instead of at the end. • General Format: = O R C R' R and R' may or may not be the same.

Naming Ketones one • • • Nearly always have a number. Take the corresponding alkane name. Drop final –e. Add –one. Number in front gives location of >C=O. (Lowest possible #. )
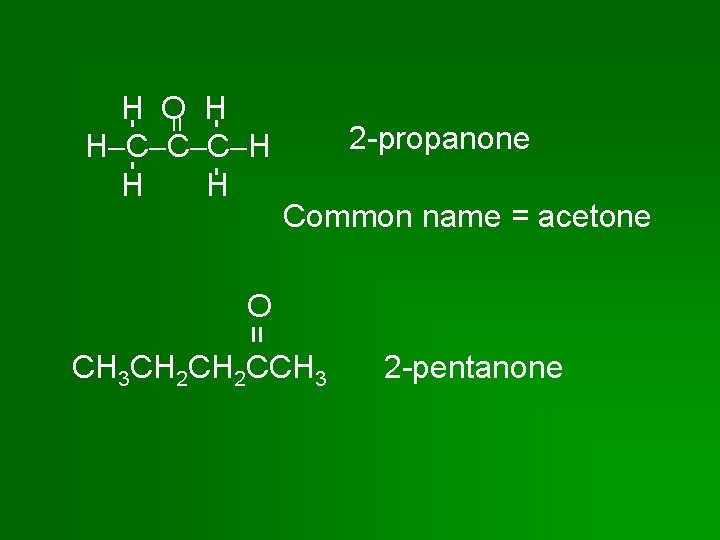
= H O H H C C C H H H 2 -propanone Common name = acetone = O CH 3 CH 2 CCH 3 2 -pentanone

Aldehydes & Ketones • • Known for appealing tastes & smells. Used as flavorings in food & candy. Used as fragrances in perfumes. Vanilla & cinnamon = aldehydes

Properties of Aldehydes & Ketones • Aldehydes & ketones contain C=O group, which makes the molecules polar. • Boiling point: Higher than alkanes, lower than alcohols. • In pure aldehydes & ketones, hydrogen bonding does NOT occur. No H bonded to O. • Can have 1 -way Hydrogen bonding in water, so they are pretty soluble in H 2 O.


Carboxylic Acids Acidic H+ = O • General Formula = R C OH • Contains a carbonyl group & a hydroxyl group bonded to same C. • The H is acidic. It ionizes in water! • Carboxylic Acids are electrolytes!
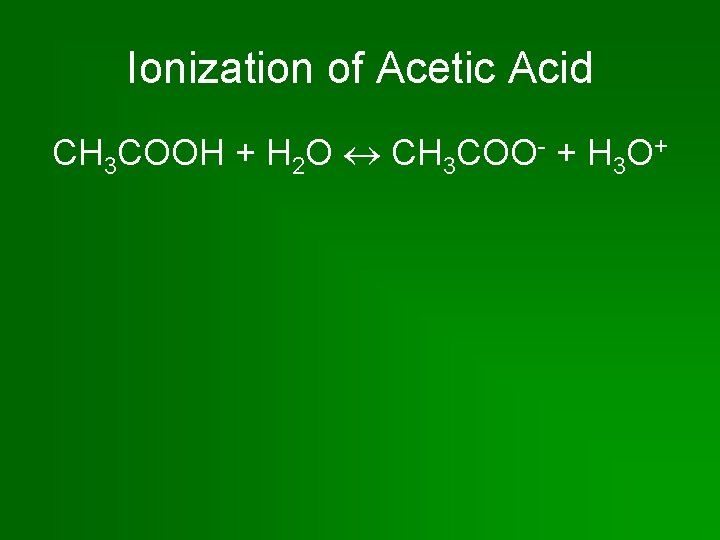
Ionization of Acetic Acid CH 3 COOH + H 2 O CH 3 COO- + H 3 O+
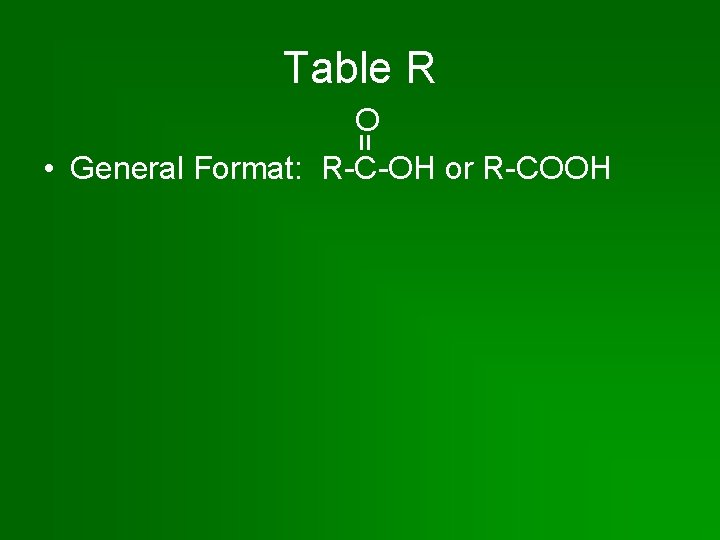
Table R = O • General Format: R-C-OH or R-COOH

Which of the following is an electrolyte? A. B. C. D. CH 3 OH alcohol CH 3 COOH aldehyde CH 2 O C 3 H 6 O ketone Correct answer is B, the carboxylic acid.

Which of the following is a nonelectrolyte? A. B. C. D. HCl CH 3 COOH Na. OH CH 3 OH Correct answer is D, an alcohol.

Naming Carboxylic Acids • Never need a number – functional group always at end. • Find name of corresponding hydrocarbon. • Drop final –e. • Add the ending –oic + the word acid.
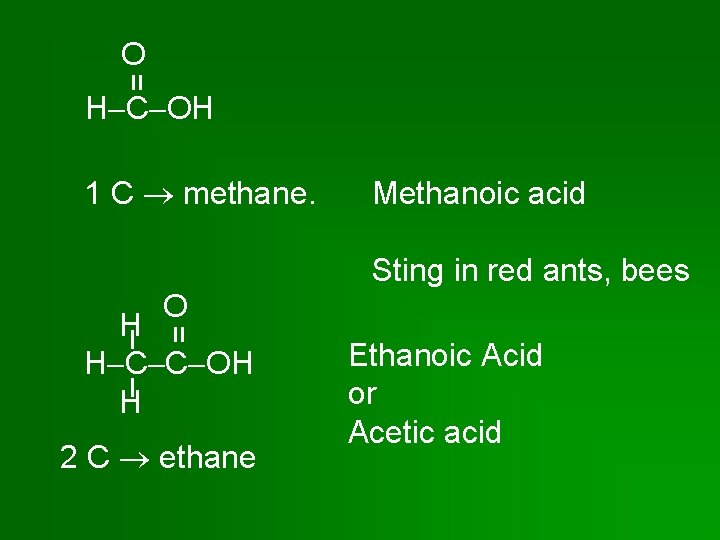
= O H C OH 1 C methane. Methanoic acid Sting in red ants, bees O = H H C C OH H 2 C ethane Ethanoic Acid or Acetic acid

CH 3 CH 2 CH 2 COOH 5 C’s pentane so the name is pentanoic acid.

Common carboxylic acids • Acetic acid – vinegar. Also produced in doughs leavened with a specific yeast sourdough bread • Citric acid • Tannic acid • Ascorbic acid • Lactic acid – Poly(lactic acid) – biodegradable polymers used as sutures in internal surgery.

Properties of Carboxylic Acids • Contain –COOH group. • H is bonded to O. Hydrogen bonding occurs. Leads to increases in boiling point over corresponding alkane. • Also can form hydrogen bonds with water so the smaller acids are pretty soluble.


Esters = O • General format: R C O R‘ • R and R‘ = hydrocarbon branches. Can be the same or different. • Esters contain a carbonyl group and a bridge O, both in the middle of the chain. • Esters are POLAR, but NO H bonded to O however.
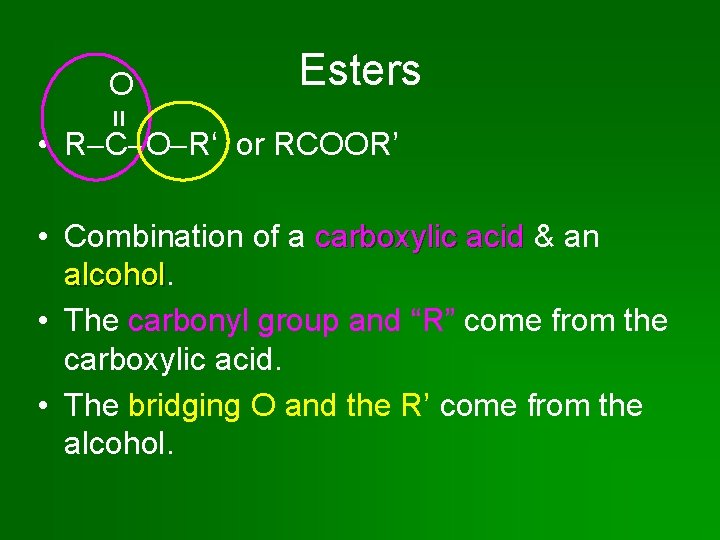
Esters = O • R C O R‘ or RCOOR’ • Combination of a carboxylic acid & an alcohol • The carbonyl group and “R” come from the carboxylic acid. • The bridging O and the R’ come from the alcohol.

Esters • Responsible for many distinctive odors • • • Pineapple Banana Orange Apple Wintergreen

Naming Esters • Name the hydrocarbon branch bonded to the bridge O first. Branches end in –yl. • The base name is derived from the branch containing the carbonyl group. Count up all the C atoms in this branch, including the C in the carbonyl. • Find the hydrocarbon base name. Drop the final –e and add –oate.
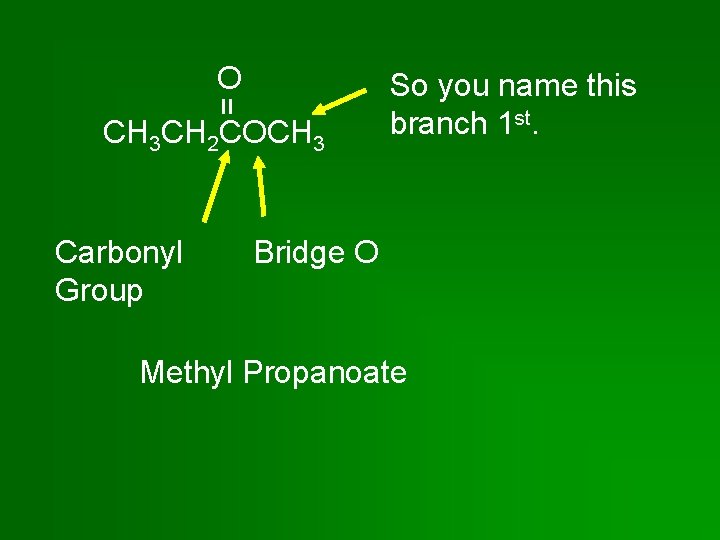
= O CH 3 CH 2 COCH 3 Carbonyl Group So you name this branch 1 st. Bridge O Methyl Propanoate
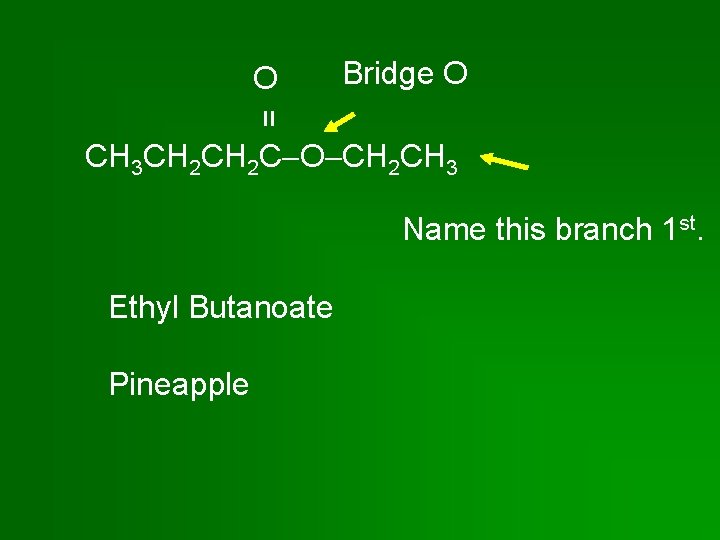
Bridge O = O CH 3 CH 2 C O CH 2 CH 3 Name this branch 1 st. Ethyl Butanoate Pineapple
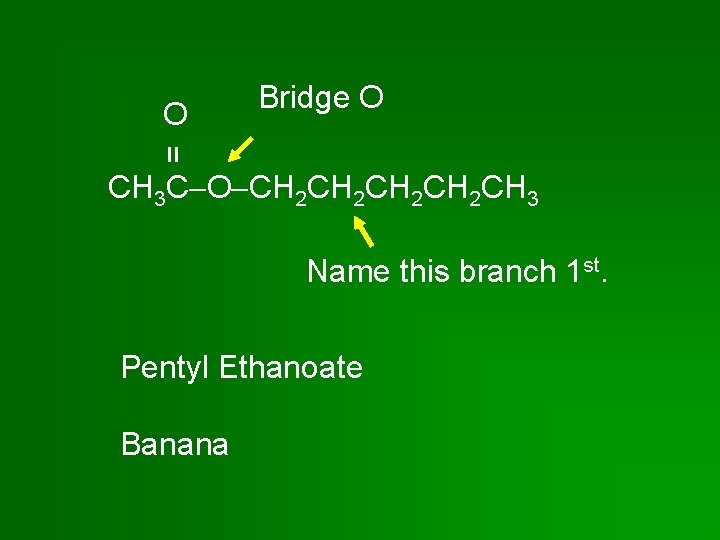
= O Bridge O CH 3 C O CH 2 CH 2 CH 3 Name this branch 1 st. Pentyl Ethanoate Banana
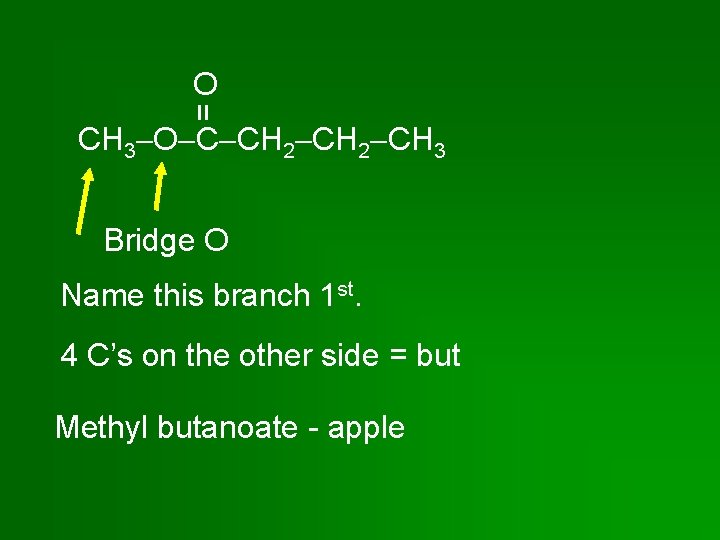
= O CH 3 O C CH 2 CH 3 Bridge O Name this branch 1 st. 4 C’s on the other side = but Methyl butanoate - apple
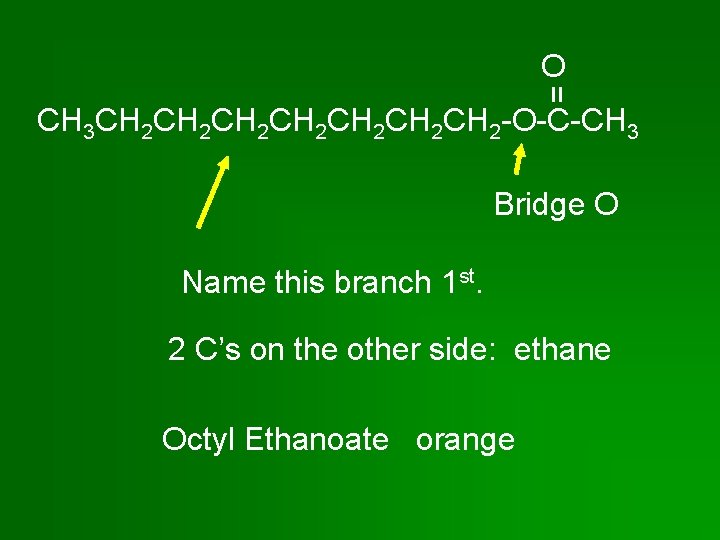
O = CH 3 CH 2 CH 2 CH 2 CH 2 -O-C-CH 3 Bridge O Name this branch 1 st. 2 C’s on the other side: ethane Octyl Ethanoate orange
- Slides: 36