Hybridization Polarity Intermolecular Forces SCH 4 UO Theoretical

Hybridization Polarity Intermolecular Forces SCH 4 UO

• Theoretical process involving the mixing of atomic orbitals to create a new set of orbitals that take part in covalent bonding • Mainly useful for describing the shapes of organic compounds
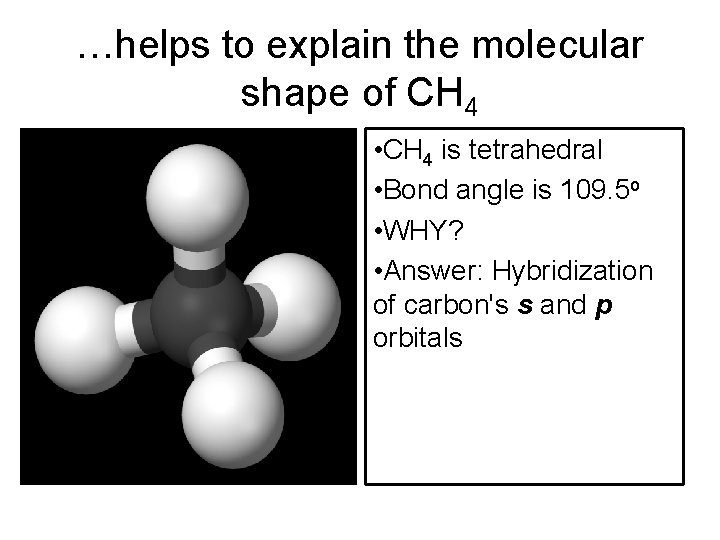
…helps to explain the molecular shape of CH 4 • CH 4 is tetrahedral • Bond angle is 109. 5 o • WHY? • Answer: Hybridization of carbon's s and p orbitals
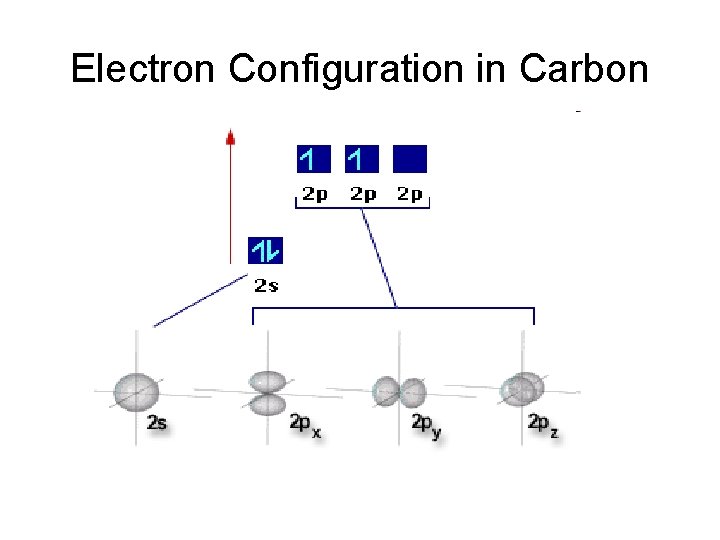
Electron Configuration in Carbon
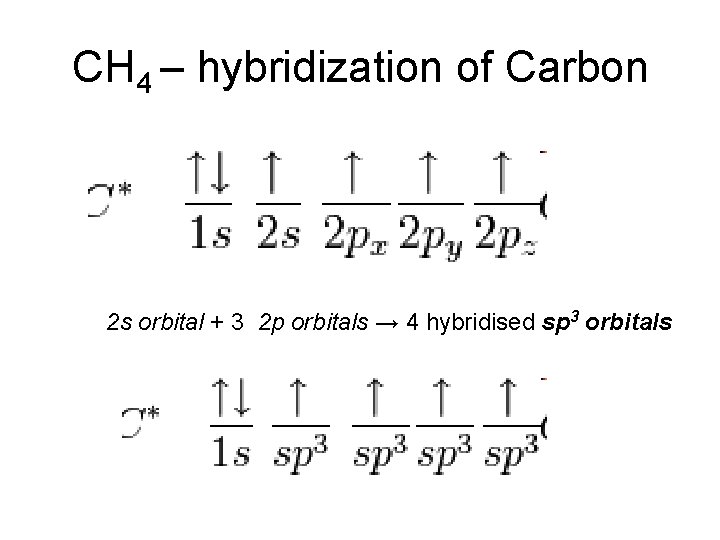
CH 4 – hybridization of Carbon 2 s orbital + 3 2 p orbitals → 4 hybridised sp 3 orbitals
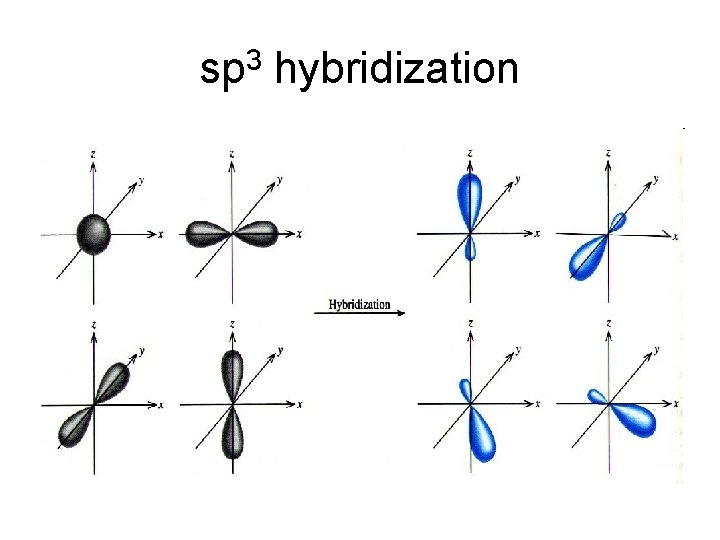
sp 3 hybridization

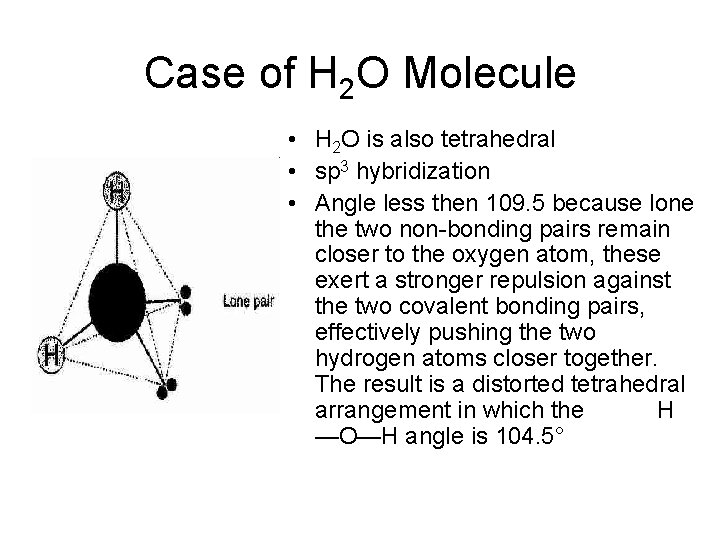
Case of H 2 O Molecule • H 2 O is also tetrahedral • sp 3 hybridization • Angle less then 109. 5 because lone the two non-bonding pairs remain closer to the oxygen atom, these exert a stronger repulsion against the two covalent bonding pairs, effectively pushing the two hydrogen atoms closer together. The result is a distorted tetrahedral arrangement in which the H —O—H angle is 104. 5°
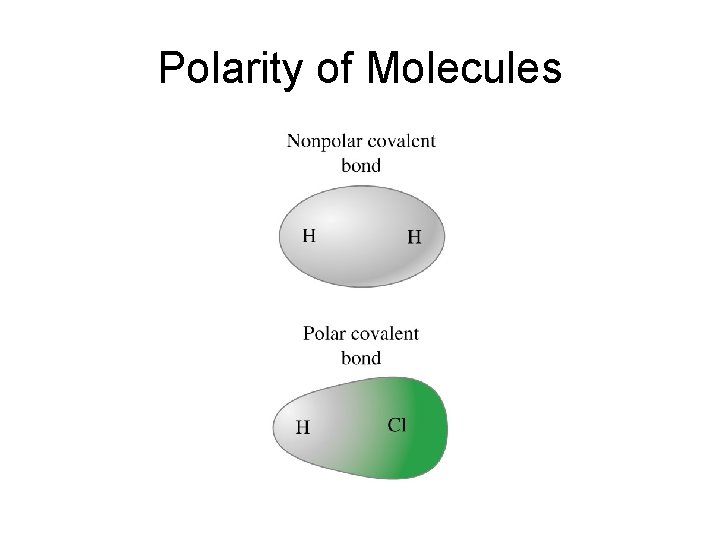
Polarity of Molecules
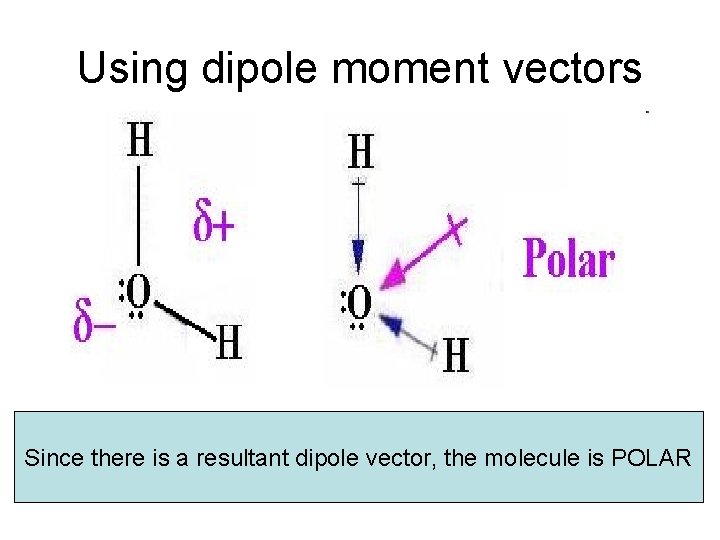
Using dipole moment vectors Since there is a resultant dipole vector, the molecule is POLAR
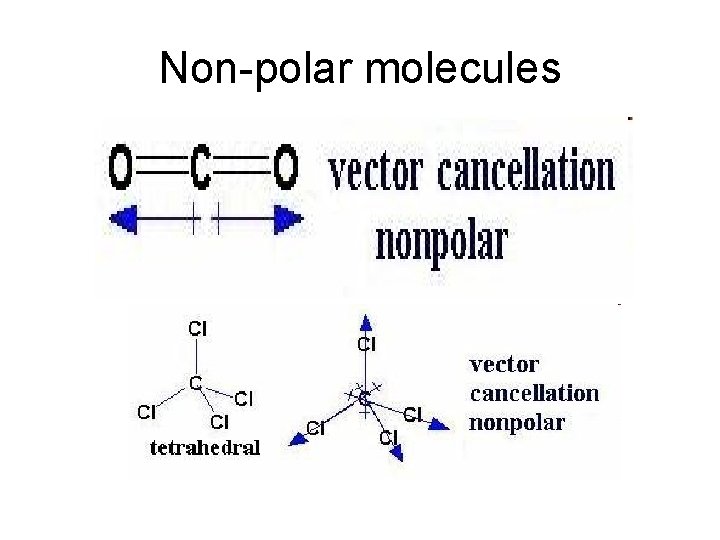
Non-polar molecules
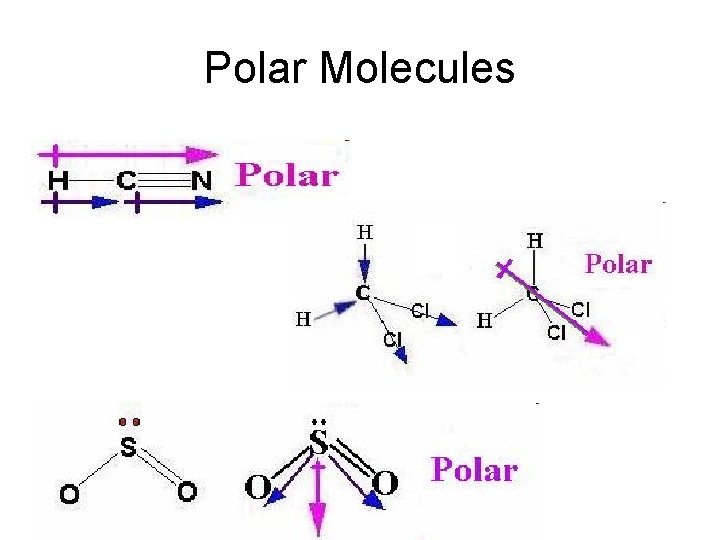
Polar Molecules

Test your skill… • Polar or non-polar? Draw the structure, determine shape and use dipole vectors to determine polarity: • CHCl 3 (chloromethane) • SO 3

Answers:
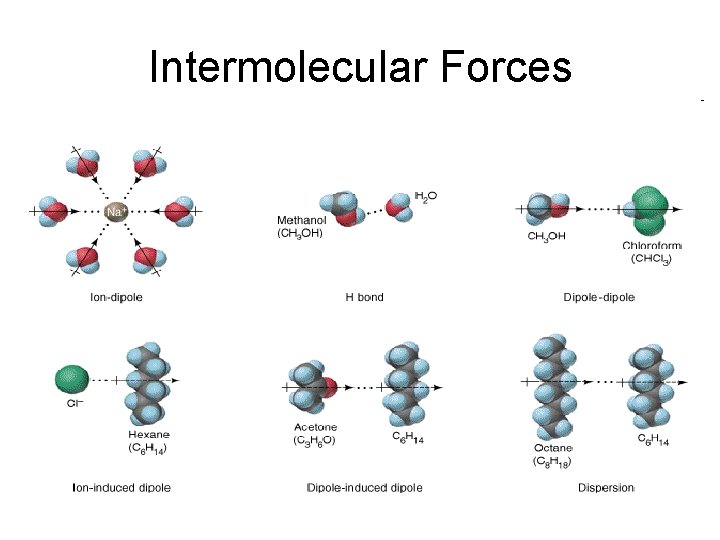
Intermolecular Forces



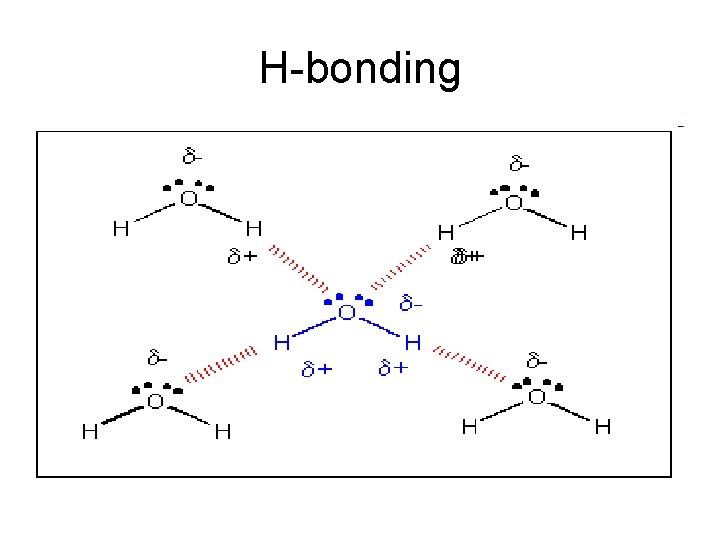
H-bonding

Practice Questions 1. Predict which compound in each pair will have the higher melting point. (a) HI or KI (b) HCl or H 2 O (c) CH 4 or NH 3 (d) CHCl 3 or CF 4
- Slides: 20