Hybridization Part of VBT n The 2 s

Hybridization (Part of VBT) n The 2 s and 2 p orbitals are similar in energy. n n n Recall that they are identical for hydrogen. Combining the wavefunctions for 2 s and 2 p gives a solution to the Schrödinger equation. The s orbital is combined with p orbitals – the number of p orbitals used depends on the molecule to be formed. n n The resulting (new) atomic orbitals are hybridized orbitals. A molecule is not simply atoms stuck together. 1
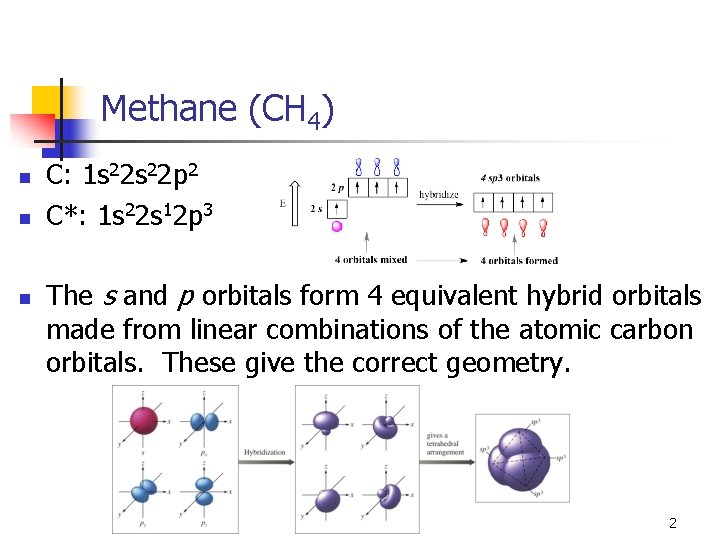
Methane (CH 4) n n n C: 1 s 22 p 2 C*: 1 s 22 s 12 p 3 The s and p orbitals form 4 equivalent hybrid orbitals made from linear combinations of the atomic carbon orbitals. These give the correct geometry. 2
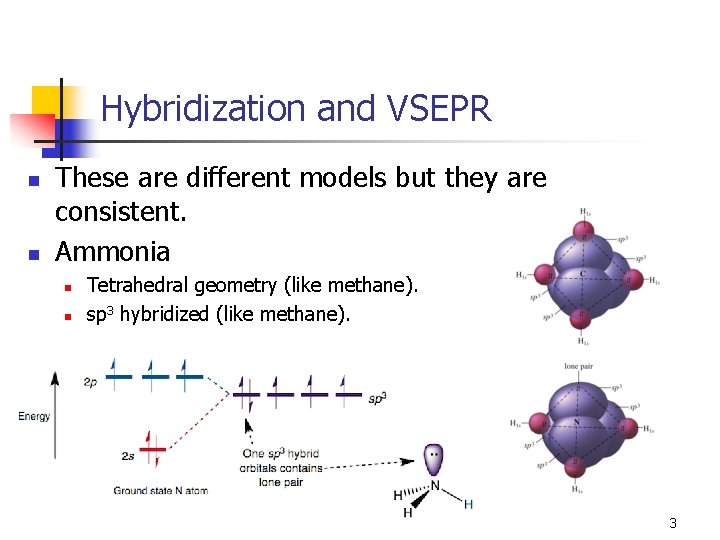
Hybridization and VSEPR n n These are different models but they are consistent. Ammonia n n Tetrahedral geometry (like methane). sp 3 hybridized (like methane). 3
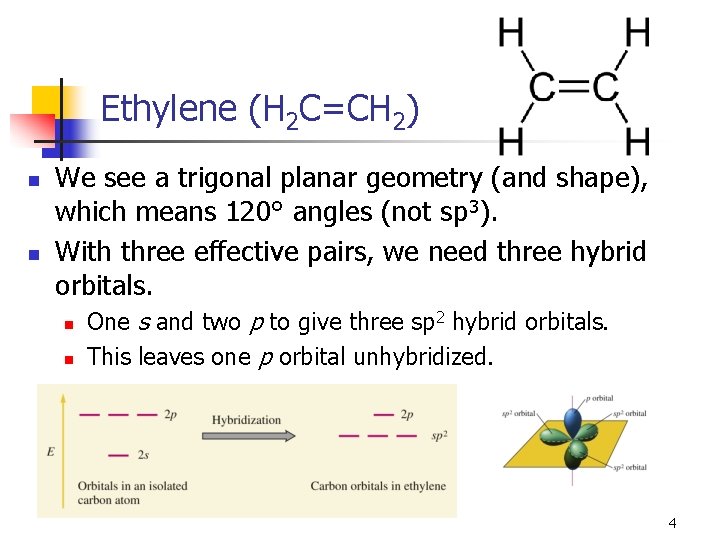
Ethylene (H 2 C=CH 2) n n We see a trigonal planar geometry (and shape), which means 120° angles (not sp 3). With three effective pairs, we need three hybrid orbitals. n n One s and two p to give three sp 2 hybrid orbitals. This leaves one p orbital unhybridized. 4
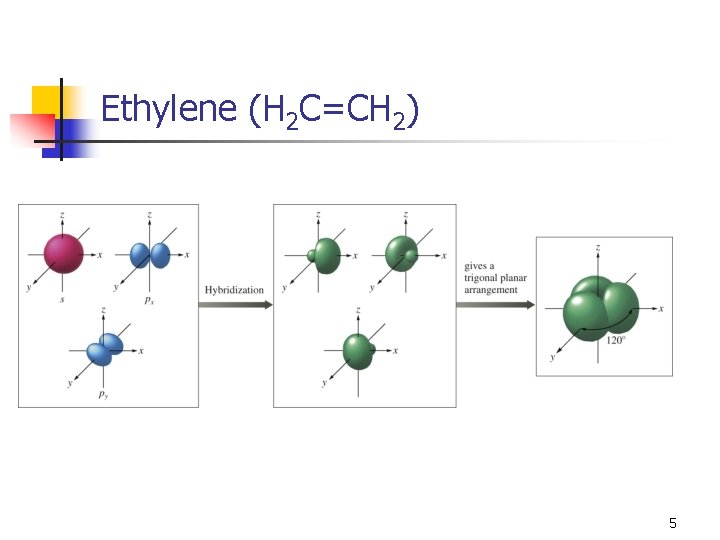
Ethylene (H 2 C=CH 2) 5
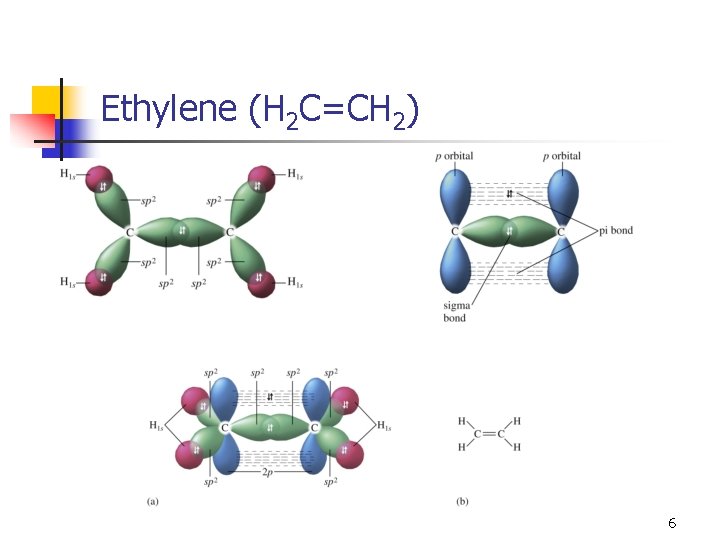
Ethylene (H 2 C=CH 2) 6

Carbon dioxide (O=C=O) n n Linear geometry and shape (180°). With two effective pairs, we need two hybrid orbitals. n n One s and one p to give two sp hybrid orbitals. This leaves two p orbitals unhybridized. 7
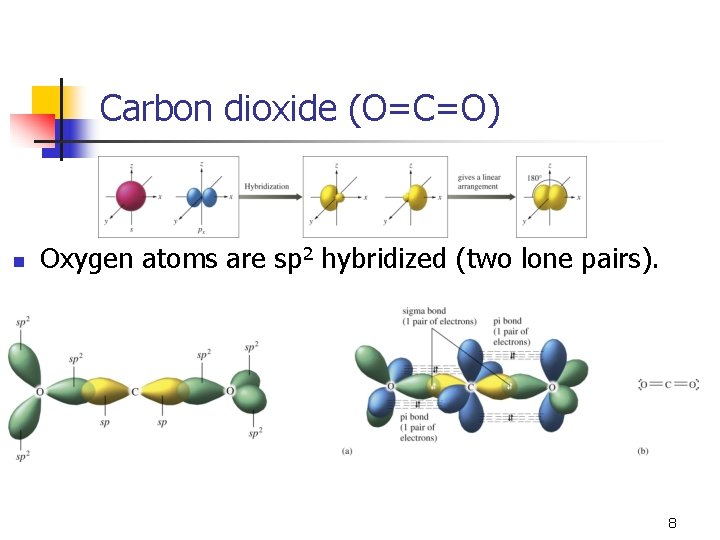
Carbon dioxide (O=C=O) n Oxygen atoms are sp 2 hybridized (two lone pairs). 8
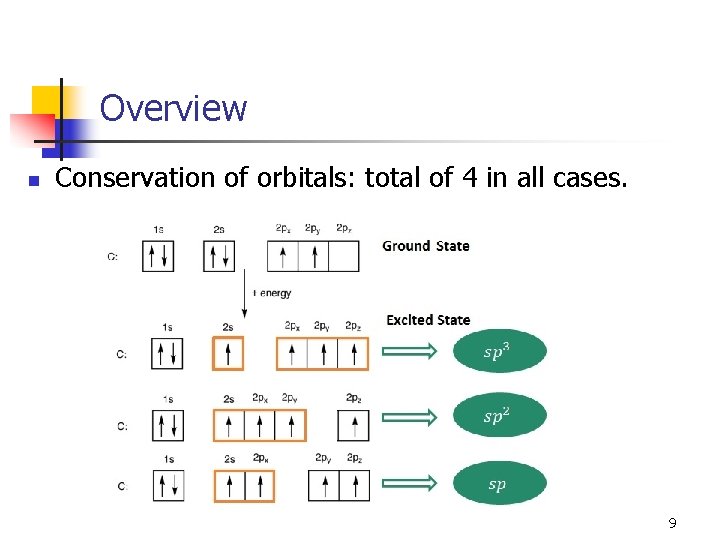
Overview n Conservation of orbitals: total of 4 in all cases. 9

CO (: C≡O: ) is like N 2 (: N≡N: ) n We use the total number of electrons available in the molecule (as we did with Lewis structures). 10
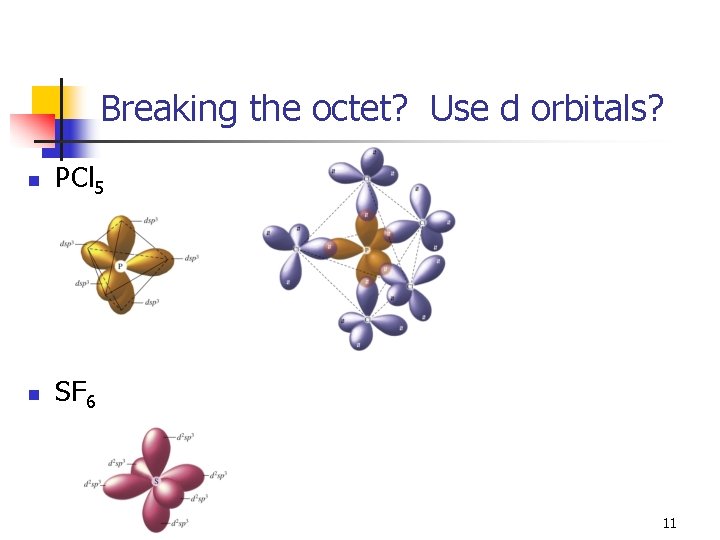
Breaking the octet? Use d orbitals? n PCl 5 n SF 6 11

Hybridization and VSEPR 12

Hybridization n Pros n n Molecules do not just consist of atoms stuck together (something “happens” to make a molecule different from the atoms). Relatively straightforward. n n Pictures! Wait until MO Theory… Easy to apply VSEPR. Cons n n Still cannot explain odd number of electrons. Bond energies not explained n n n Ions like O 2+. Magnetism not explained. Colors of solutions not explained. 13

Molecular Orbital Theory n Molecule: n n Simplest: overlap of unchanged atomic orbitals (atoms stuck together) Hybridization: overlap of hybridized atomic orbitals localized between nuclei MO Theory: collection of nuclei with delocalized molecular orbitals Linear combination of atomic orbitals in the molecule to form molecular orbitals. n n n Atomic orbitals must overlap to form molecular orbitals. Wave functions can constructively and destructively interfere. Conservation of the number of orbitals – the number of molecular orbitals equals the total number of molecular orbitals. 14
- Slides: 14