HYBRIDIZATION IN SQUARE PLANER COMPLEXS TOPIC HYBRIDIZATION IN

HYBRIDIZATION IN SQUARE PLANER COMPLEXS • TOPIC: -HYBRIDIZATION IN SQUARE PLANER COMPLEX. • COURES : -M. Sc(Hons)CHEMISTR

ABSTRACT • HYBERDIZATION: -The phenomenon of mixing of orbitals of the same atom with slight difference in energies so as to redistribute their energies and give new orbitals of equivalent energy and shape. The new orbitals which get formed are known as hybrid. • TYPES OF HYBERDIZATION: -These are: • SP HYBERDIZATION • SP 2 HYBERDIZATION • SP 3 HYBERDIZATION (Regular tetrahedron geometry) • SP 3 D HYBERDIZATION (Trigonal bipyramidal geometry ) • SP 3 D 2 HYBERDIZATION (Octahedral geometry ) • SP 3 D 3 HYBERDIZATION (Pentagonal bipyramedal geometry • DSP 2 HYBERDIZATION (Square planer geometry )

INTRODUCTION Hybridization and the LE Model of Bonding — Lewis structures of molecules — prediction of geometry of molecules — hybrid orbitals (sp 3, sp 2, sp, dsp 3, d 2 sp 3) — interpretation of structure and bonding Molecular Orbital Model of Bonding in Molecules — molecular orbital diagrams — bond order — magnetism Molecular Spectroscopy — Vibrational/Rotational Spectroscopies — Electronic — Nuclear Magnetic Resonance (NMR) Spectroscopy

Hybridization and the LE Model of Bonding — Assume bonding involves only valence orbitals — Methane, CH 4: Isolated atoms Valence orbitals H 1 s 1 C 2 s 22 p 2 (2 p: 2 px, 2 py, 2 pz) H atoms in CH 4 will use 1 s orbitals Of the two types of orbitals (2 s and 2 p) Which will C atoms use for bonding in CH 4? — If both are used: 2 different types of C-H bonds (Contrary to experimental facts) — Neither of the “native” atomic orbitals of C atoms are used; instead, new hybrid orbitals are used.

Hybridization of atomic orbitals The mixing of the “native” atomic orbitals to form special orbitals for bonding is called hybridization. The 4 new equivalent orbitals formed by mixing the one 2 s and three 2 p orbitals are called sp 3 orbitals. The carbon atom is said to undergo sp 3 hybridization, i. e. is sp 3 hybridized. Energy-level diagram showing the sp 3 hybridization 2 p hybridization Energy 2 s Orbitals in isolated C atom sp 3 Orbitals in CH 4 molecule
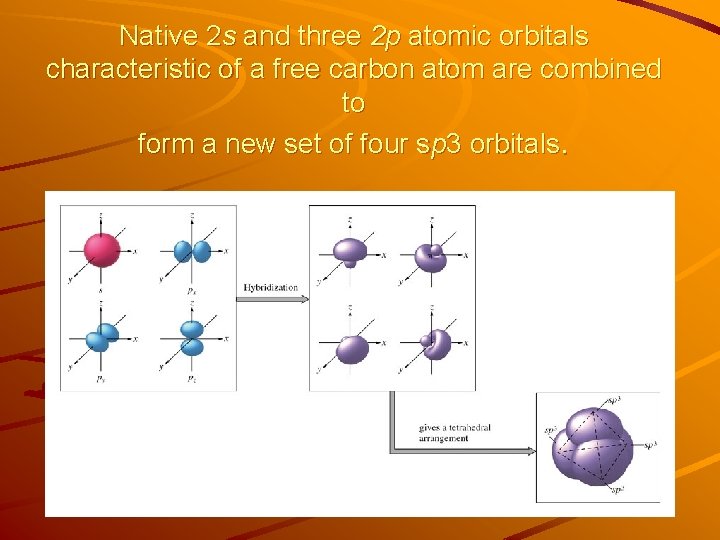
Native 2 s and three 2 p atomic orbitals characteristic of a free carbon atom are combined to form a new set of four sp 3 orbitals.
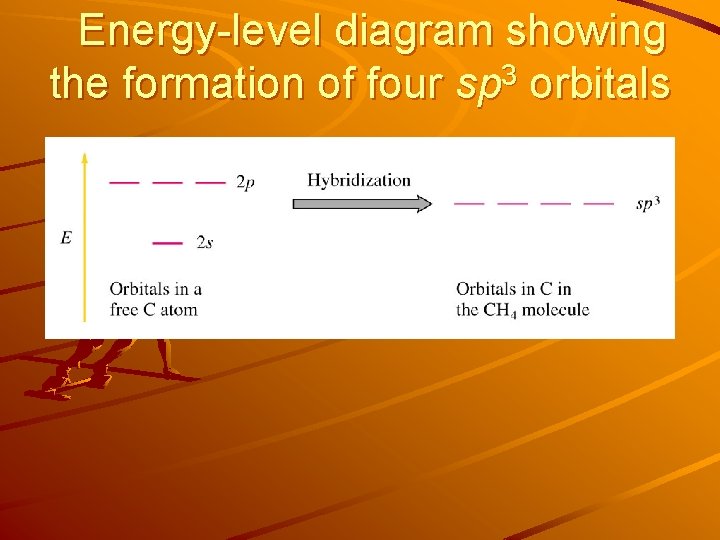
Energy-level diagram showing the formation of four sp 3 orbitals

Sp 2 Hybridization Consider ethylene C 2 H 4 molecule Lewis structure — 12 valence e-s in the molecule — What orbitals do the carbon atoms use to bond in ethylene? — 3 effective electron pairs around each carbon • VSEPR model predicts a trigonal planar geometry 120 o angles • sp 3 orbitals with tetrahedral geometry and 109. 5 o angles will not work here.

The plastics shown here were manufactured with ethylene.
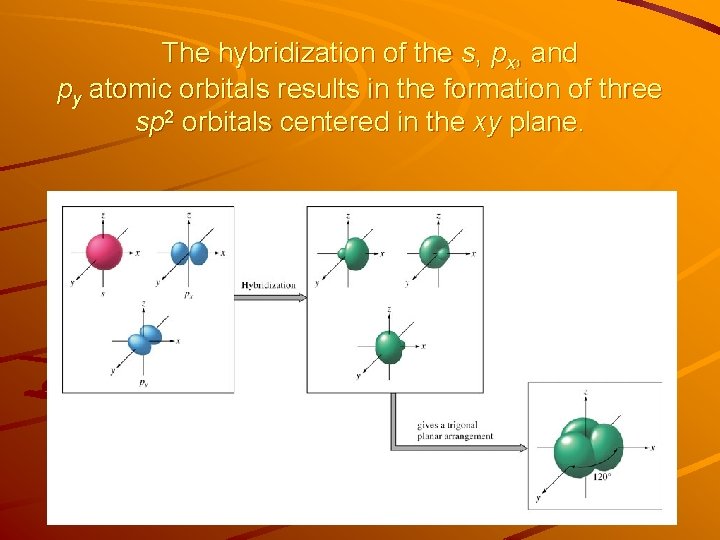
The hybridization of the s, px, and py atomic orbitals results in the formation of three sp 2 orbitals centered in the xy plane.
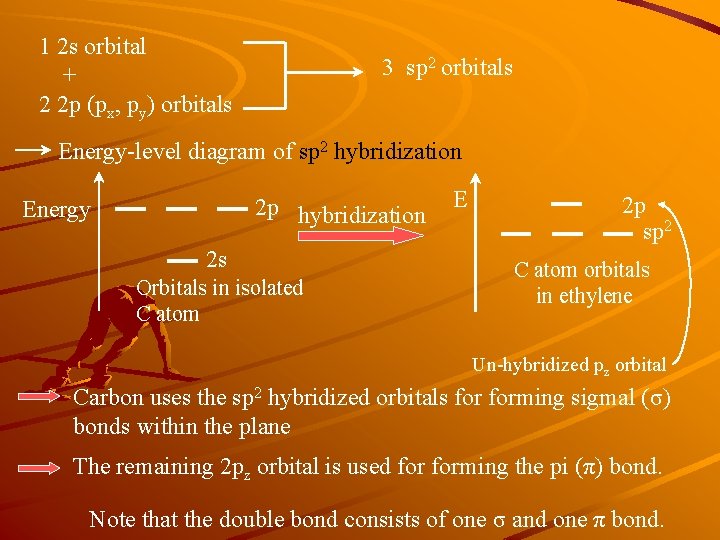
1 2 s orbital + 2 2 p (px, py) orbitals 3 sp 2 orbitals Energy-level diagram of sp 2 hybridization 2 p hybridization Energy 2 s Orbitals in isolated C atom E 2 p sp 2 C atom orbitals in ethylene Un-hybridized pz orbital Carbon uses the sp 2 hybridized orbitals forming sigmal (σ) bonds within the plane The remaining 2 pz orbital is used forming the pi (π) bond. Note that the double bond consists of one σ and one π bond.
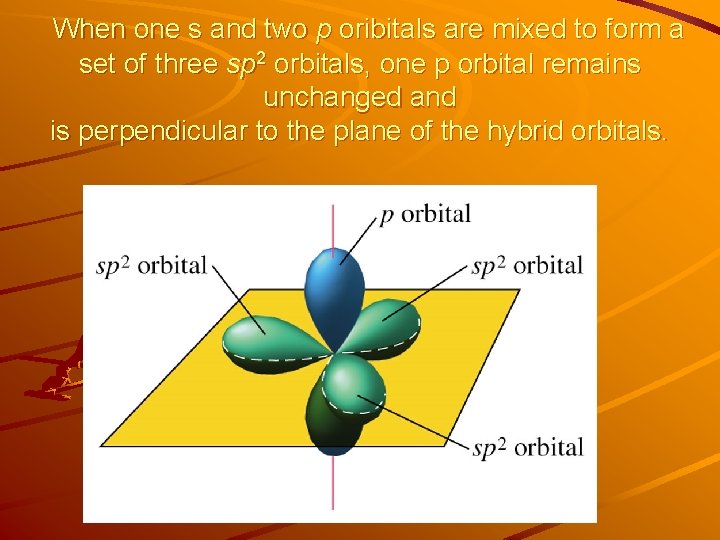
When one s and two p oribitals are mixed to form a set of three sp 2 orbitals, one p orbital remains unchanged and is perpendicular to the plane of the hybrid orbitals.
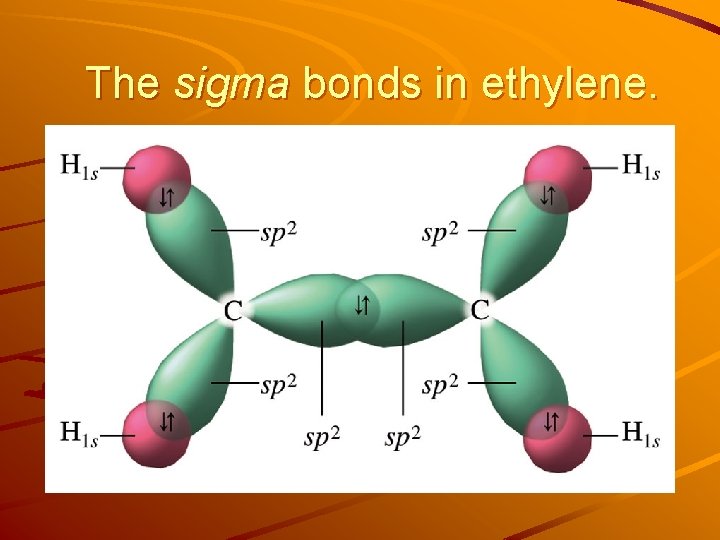
The sigma bonds in ethylene.
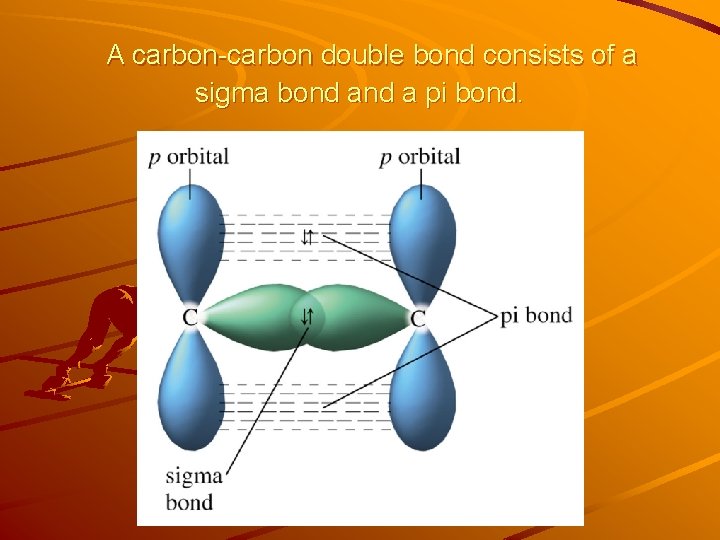
A carbon-carbon double bond consists of a sigma bond a pi bond.
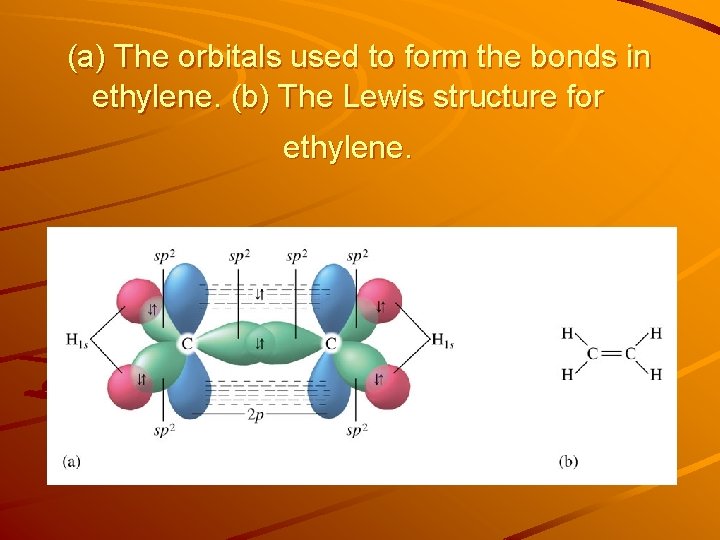
(a) The orbitals used to form the bonds in ethylene. (b) The Lewis structure for ethylene.
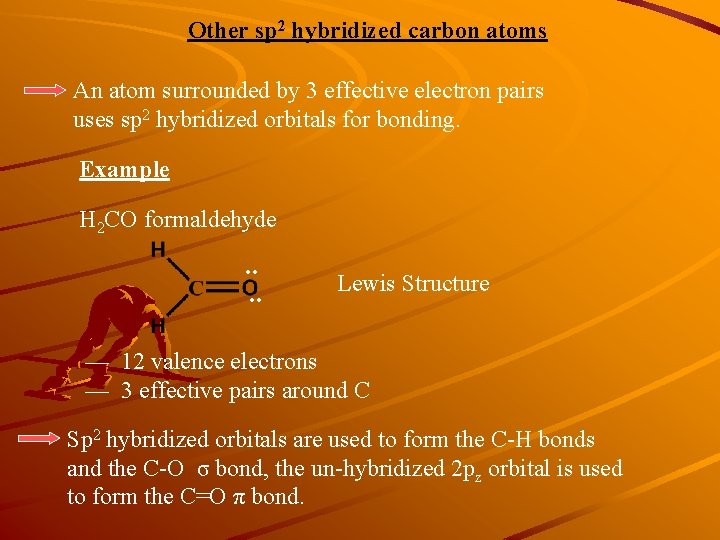
Other sp 2 hybridized carbon atoms An atom surrounded by 3 effective electron pairs uses sp 2 hybridized orbitals for bonding. Example H 2 CO formaldehyde . . Lewis Structure — 12 valence electrons — 3 effective pairs around C Sp 2 hybridized orbitals are used to form the C-H bonds and the C-O σ bond, the un-hybridized 2 pz orbital is used to form the C=O π bond.

sp Hybridization Carbon in carbon dioxide, CO 2 uses another type of hybridization (rather than sp 2 or sp 3) ‧‧ ‧‧ ‧‧ O=C=O ‧‧ 2 hybrid orbitals required to meet the 180° (linear) geometry requirement are sp orbitals. 2 effective pairs around C atom sp hybrid orbitals 3 effective pairs around O atom sp 2 orbitals Energy 2 p 1 s Orbitals in a free C atom Hybridization 2 p sp Orbitals in sp hybridized orbitals in CO 2
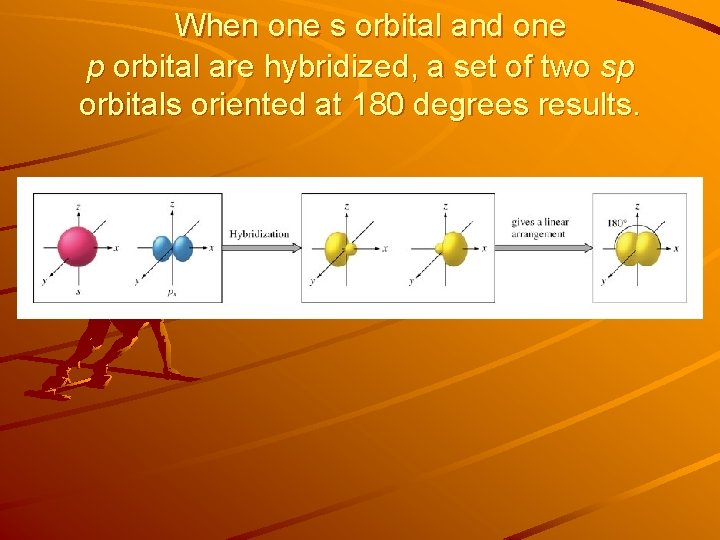
When one s orbital and one p orbital are hybridized, a set of two sp orbitals oriented at 180 degrees results.
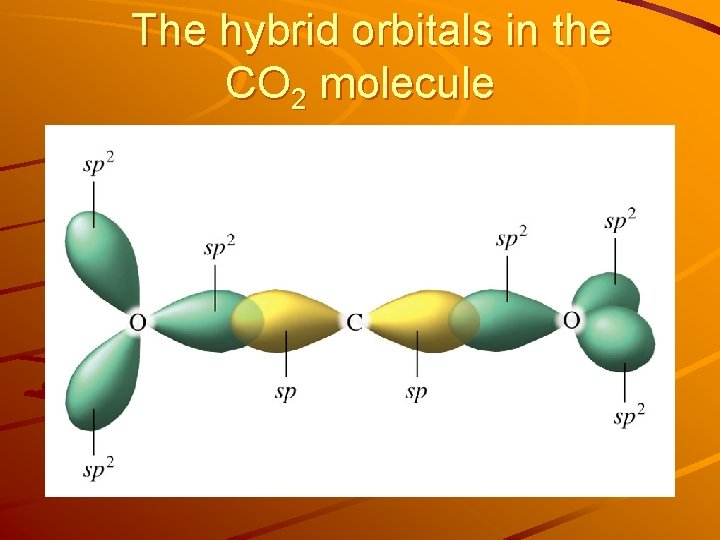
The hybrid orbitals in the CO 2 molecule
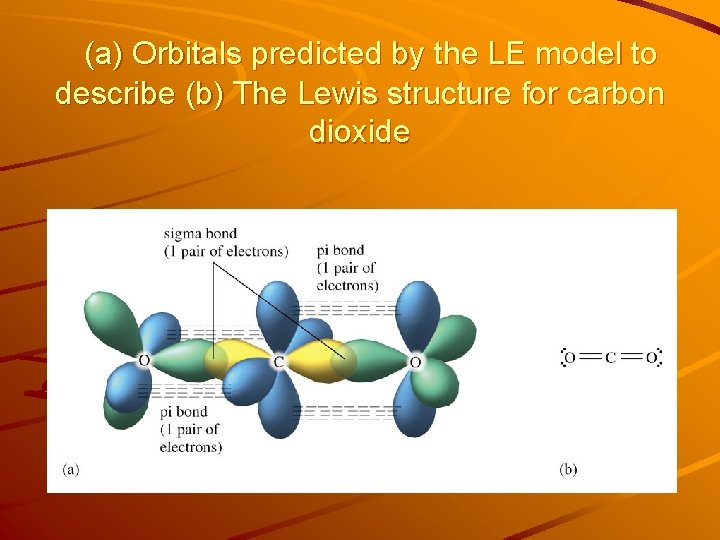
(a) Orbitals predicted by the LE model to describe (b) The Lewis structure for carbon dioxide

Other Examples of sp Hybridization Example N 2 molecule :N N: Lewis Structure N atom: 2 s 22 p 3 2 effective pairs around each N atom in the Lewis structure – Linear (180°) geometry – 2 sp orbitals for each N atom: .1 sp orbital forming the σ bond .1 sp orbital for holding the lone pair The remaining un-changed 2 p orbitals are used to form the 2 π bonds. Each triple bond consists of one σ and two π bonds.
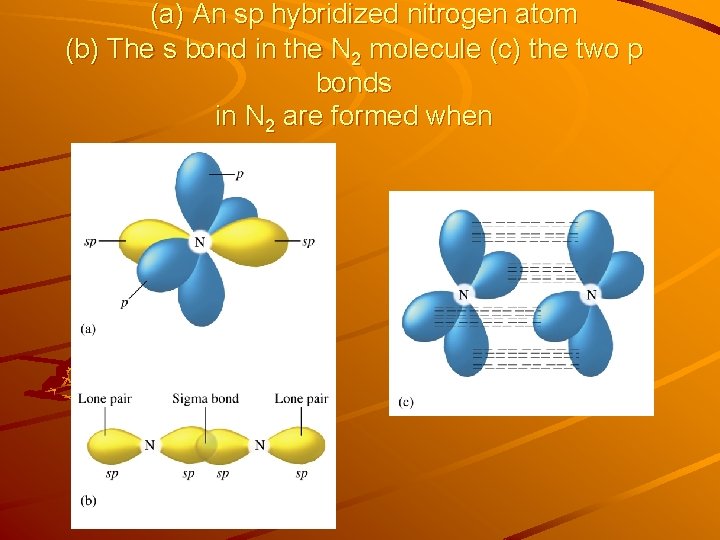
(a) An sp hybridized nitrogen atom (b) The s bond in the N 2 molecule (c) the two p bonds in N 2 are formed when
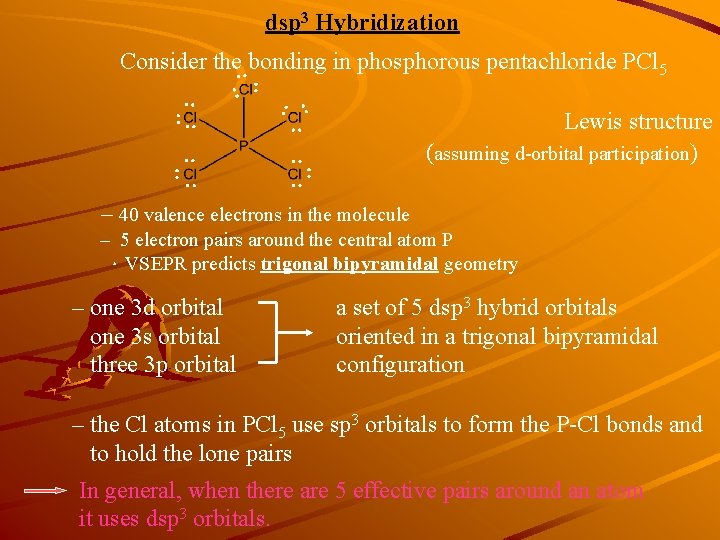
dsp 3 Hybridization : Consider the bonding in phosphorous pentachloride PCl 5 :: : : : : Lewis structure (assuming d-orbital participation) : :: : – 40 valence electrons in the molecule – 5 electron pairs around the central atom P .VSEPR predicts trigonal bipyramidal geometry – one 3 d orbital one 3 s orbital three 3 p orbital a set of 5 dsp 3 hybrid orbitals oriented in a trigonal bipyramidal configuration – the Cl atoms in PCl 5 use sp 3 orbitals to form the P-Cl bonds and to hold the lone pairs In general, when there are 5 effective pairs around an atom it uses dsp 3 orbitals.
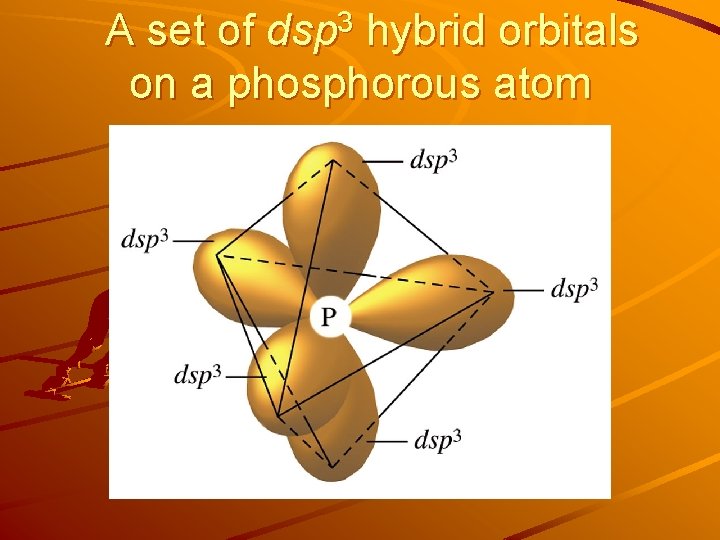
A set of dsp 3 hybrid orbitals on a phosphorous atom
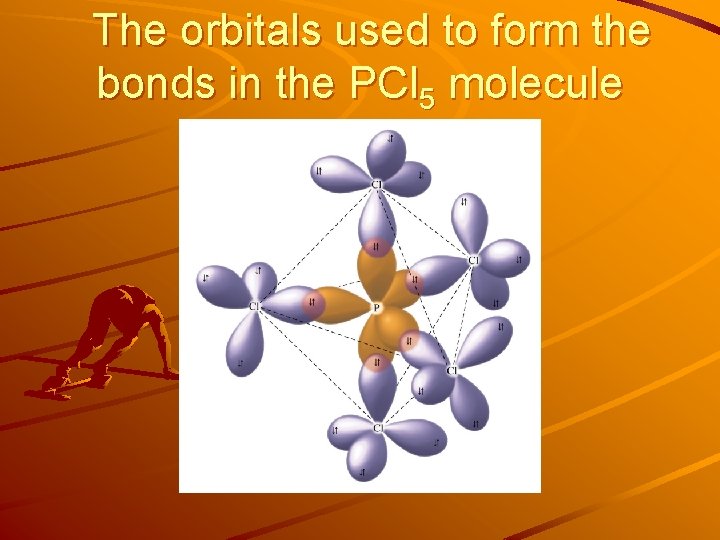
The orbitals used to form the bonds in the PCl 5 molecule
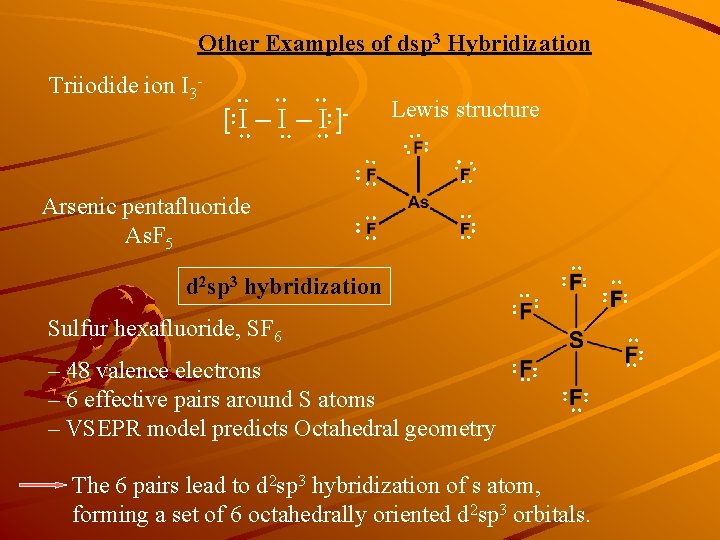
Other Examples of dsp 3 Hybridization : : : Triiodide ion I 3 - Lewis structure : : :: : : : :: Arsenic pentafluoride As. F 5 : [:I – I:]- : :: :: : – 48 valence electrons – 6 effective pairs around S atoms – VSEPR model predicts Octahedral geometry : Sulfur hexafluoride, SF 6 : d 2 sp 3 hybridization The 6 pairs lead to d 2 sp 3 hybridization of s atom, forming a set of 6 octahedrally oriented d 2 sp 3 orbitals.

An octahedral set of d 2 sp 3 orbitals on a sulfur atom

The relationship among the number of effective pairs, their spatial arrangement, and the hybrid orbital set required
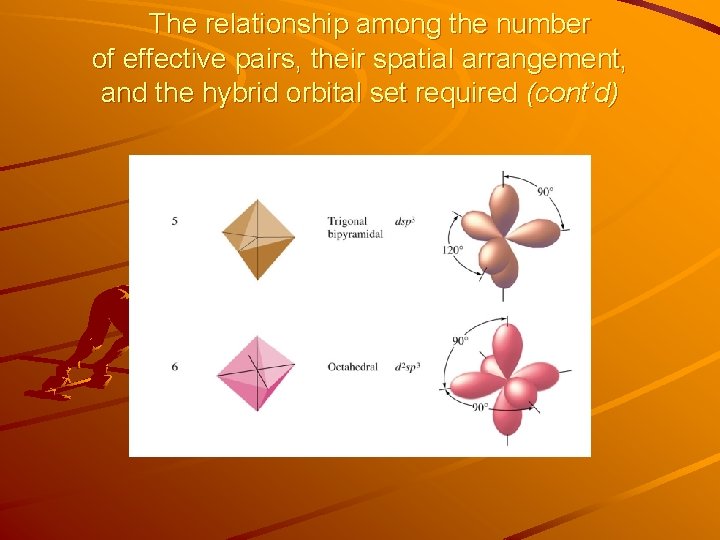
The relationship among the number of effective pairs, their spatial arrangement, and the hybrid orbital set required (cont’d)

Turning to Square Planar Complexes Most convenient to use a local coordinate system on each ligand with y pointing in towards the metal. py to be used for s bonding. z being perpendicular to the molecular plane. pz to be used for p bonding perpendicular to the plane, p^. x lying in the molecular plane. px to be used for p bonding in the molecular plane, p|.
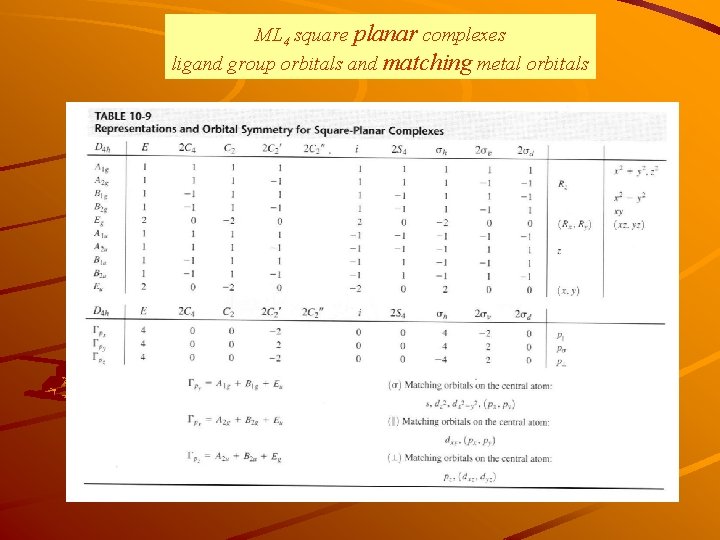
ML 4 square planar complexes ligand group orbitals and matching metal orbitals
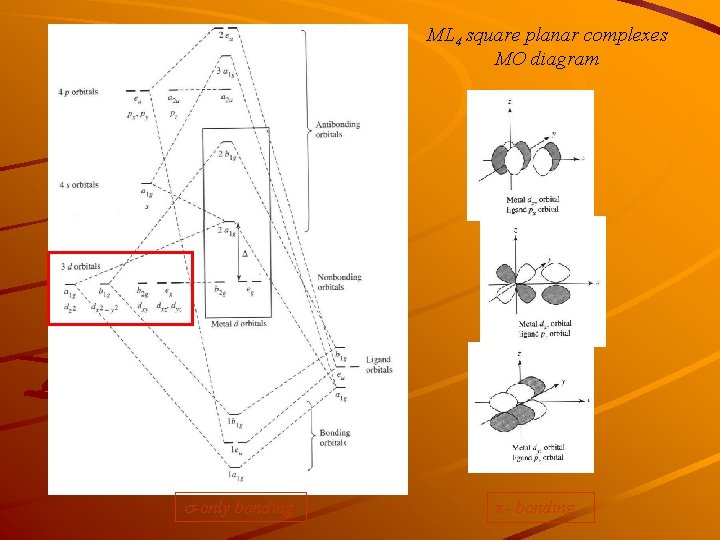
ML 4 square planar complexes MO diagram s-only bonding π - bonding

SQUARE PLANER MOLECULE GEOMETRY • Idealized structure of a compound with square planar coordination geometry. • The square planar molecular geometry in chemistry describes the stereochemistry (spatial arrangement of atoms) that is adopted by certain chemical compounds. As the name suggests, molecules of this geometry have their atoms positioned at the corners of a square on the same plane about a central atom.
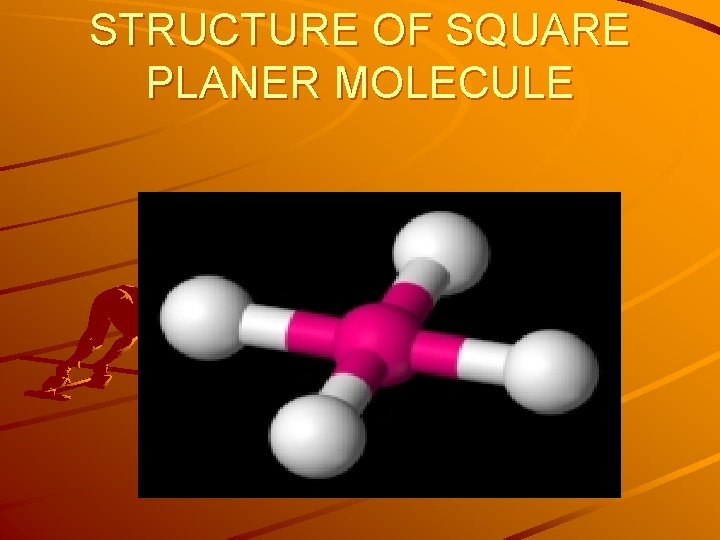
STRUCTURE OF SQUARE PLANER MOLECULE

Molecular Geometry bond length, angle determined experimentally Lewis structures bonding geometry VSEPR Valence Shell Electron Pair Repulsion octahedron 90 o bond angles small groups big groups trigonal bipyramid equatorial 120 o axial 180 o
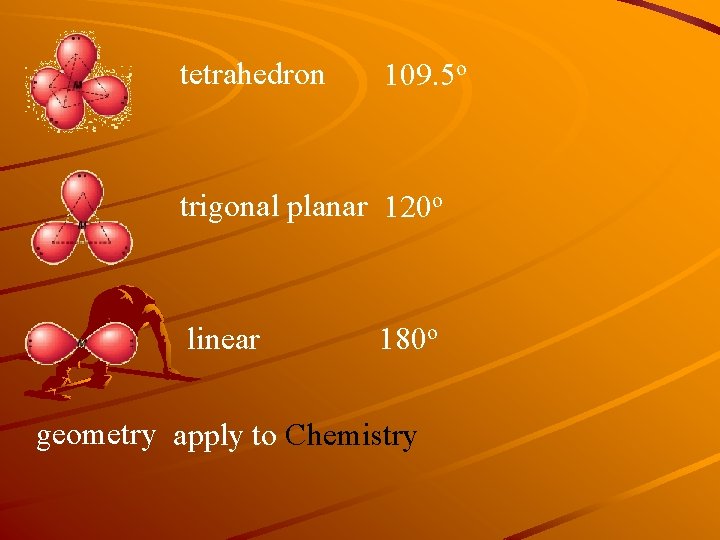
tetrahedron 109. 5 o trigonal planar 120 o linear 180 o geometry apply to Chemistry

linear 180 o Be. Cl 2 valence e- = 2 + (2 x 7) = 16 efewer than 8 e. Be . . Cl. . two valence pairs on Be bonding elinear molecule
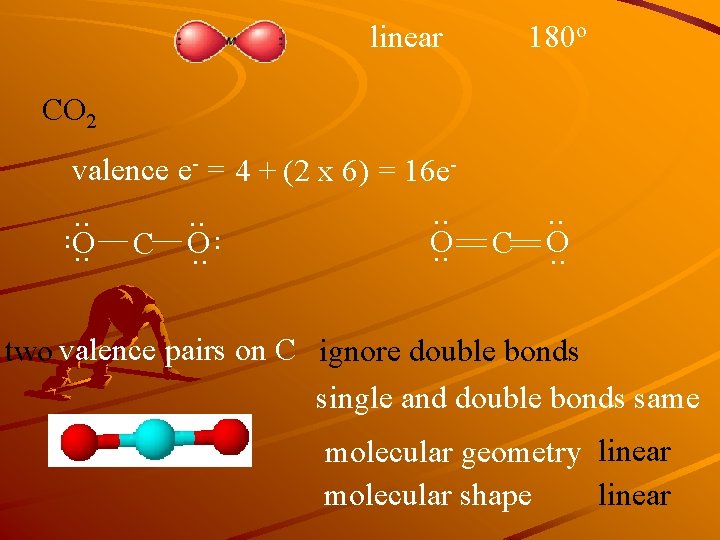
linear 180 o CO 2 valence e- = 4 + (2 x 6) = 16 e- . . C O. . two valence pairs on C ignore double bonds single and double bonds same molecular geometry linear molecular shape linear
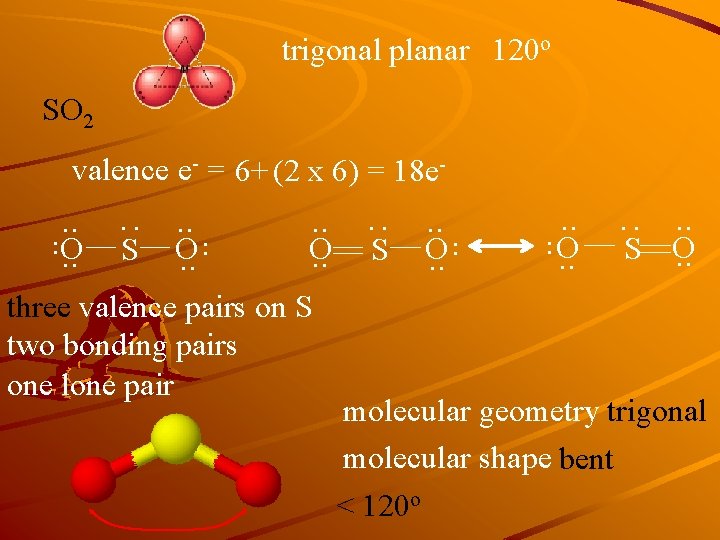
trigonal planar 120 o SO 2 three valence pairs on S two bonding pairs one lone pair . . O. . S O. . : S . . O. . : . . O. . S . . O. . : valence e- = 6+ (2 x 6) = 18 e- molecular geometry trigonal molecular shape bent < 120 o
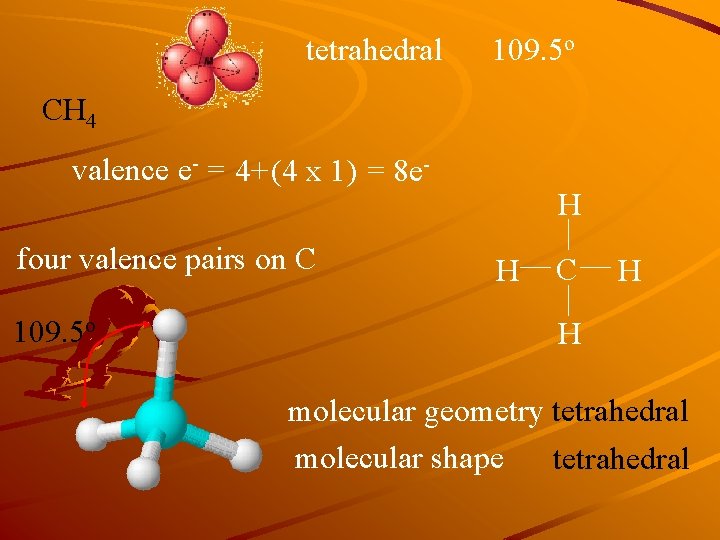
tetrahedral 109. 5 o CH 4 valence e- = 4+(4 x 1) = 8 efour valence pairs on C 109. 5 o H H C H H molecular geometry tetrahedral molecular shape tetrahedral
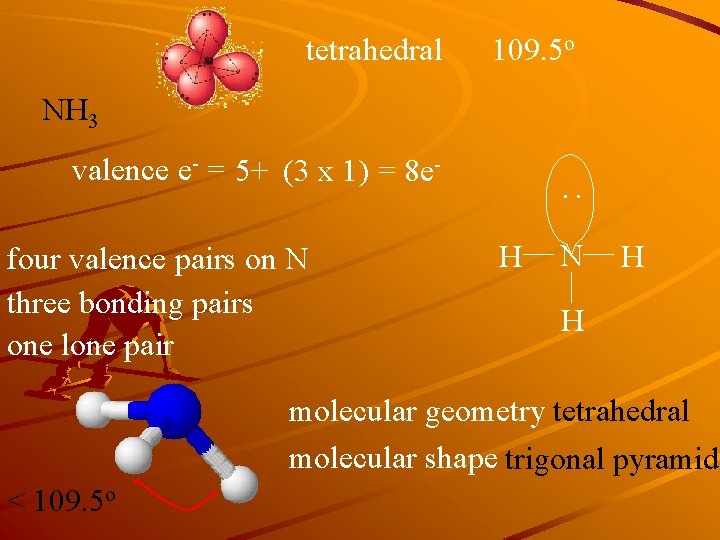
tetrahedral 109. 5 o NH 3 valence e- = 5+ (3 x 1) = 8 e- : four valence pairs on N three bonding pairs one lone pair H N H H molecular geometry tetrahedral molecular shape trigonal pyramid < 109. 5 o
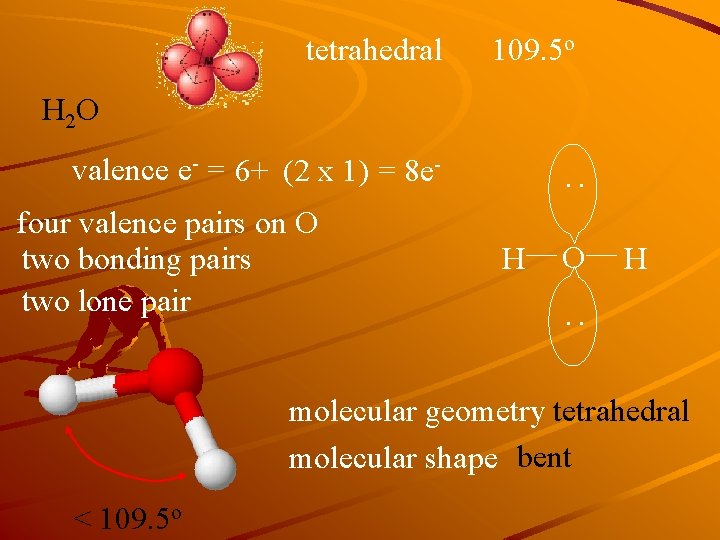
tetrahedral 109. 5 o H 2 O four valence pairs on O two bonding pairs two lone pair : valence e- = 6+ (2 x 1) = 8 e. H O H : molecular geometry tetrahedral molecular shape bent < 109. 5 o
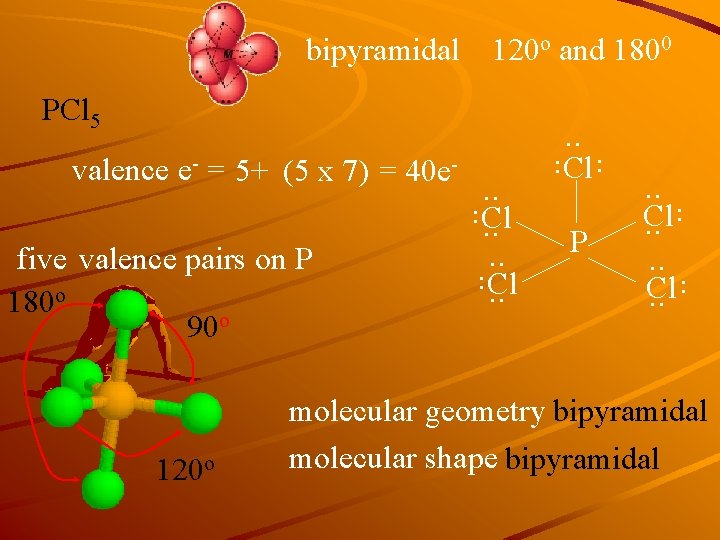
bipyramidal 120 o and 1800 PCl 5 . . 120 o . . five valence pairs on P 180 o 90 o P . . Cl. . Cl . . valence e- = 5+ (5 x 7) = 40 e- . . molecular geometry bipyramidal molecular shape bipyramidal
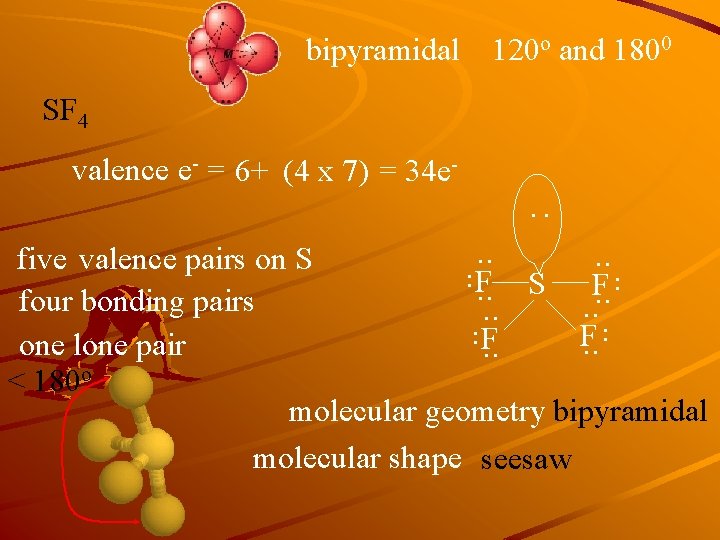
bipyramidal 120 o and 1800 SF 4 valence e- = 6+ (4 x 7) = 34 e- : . . . five valence pairs on S F. . four bonding pairs. . F F. . one lone pair. . < 180 o molecular geometry bipyramidal molecular shape seesaw
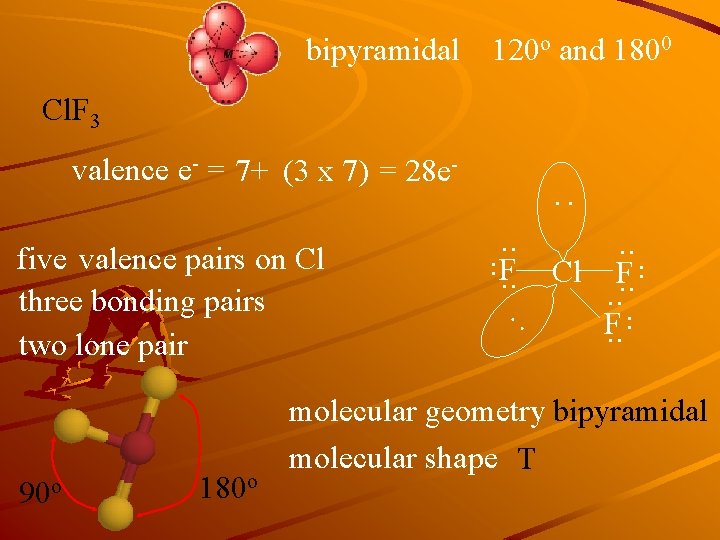
bipyramidal 120 o and 1800 Cl. F 3 valence e- = 7+ (3 x 7) = 28 e- : . . 180 o : 90 o . . five valence pairs on Cl three bonding pairs two lone pair . . F. . Cl. . F. . molecular geometry bipyramidal molecular shape T
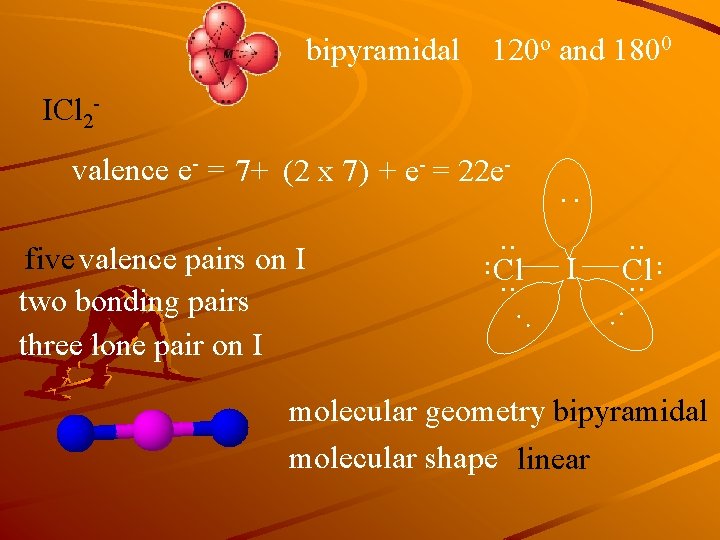
bipyramidal 120 o and 1800 ICl 2 valence e- = 7+ (2 x 7) + e- = 22 e- : : : I . . Cl. . . five valence pairs on I two bonding pairs three lone pair on I . . Cl. . molecular geometry bipyramidal molecular shape linear
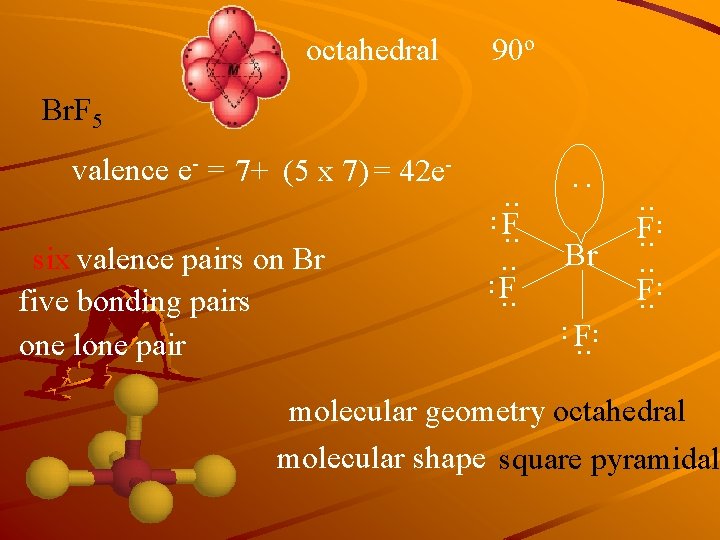
octahedral 90 o Br. F 5 Br . . . F. . six valence pairs on Br five bonding pairs one lone pair . . F. . : valence e- = 7+ (5 x 7) = 42 e- molecular geometry octahedral molecular shape square pyramidal
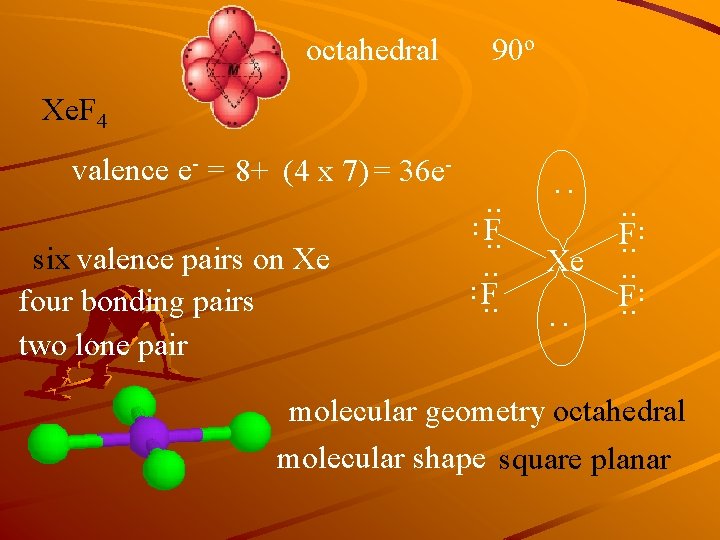
octahedral 90 o Xe. F 4 : . . six valence pairs on Xe four bonding pairs two lone pair . . F. . Xe. . F. . : valence e- = 8+ (4 x 7) = 36 e- molecular geometry octahedral molecular shape square planar

SUBSTITUTION IN SQUARE PLANER Substitution at Square Planar Metal Complexes Examples of Square Planar Transition Metal Complexes: Ni(II) (mainly d 8) Rh(I) Pd(II) Ir(I) Pt(II) Au(III) General Rate Law: Factors Which Affect The Rate Of Substitution 1. Role of the Entering Group 2. The Role of The Leaving Group 3. The Nature of the Other Ligands in the Complex 4. Effect of the Metal Centre
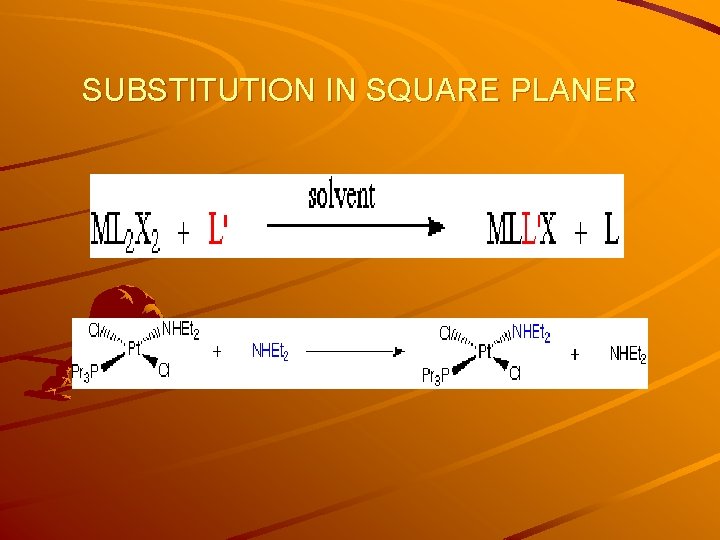
SUBSTITUTION IN SQUARE PLANER
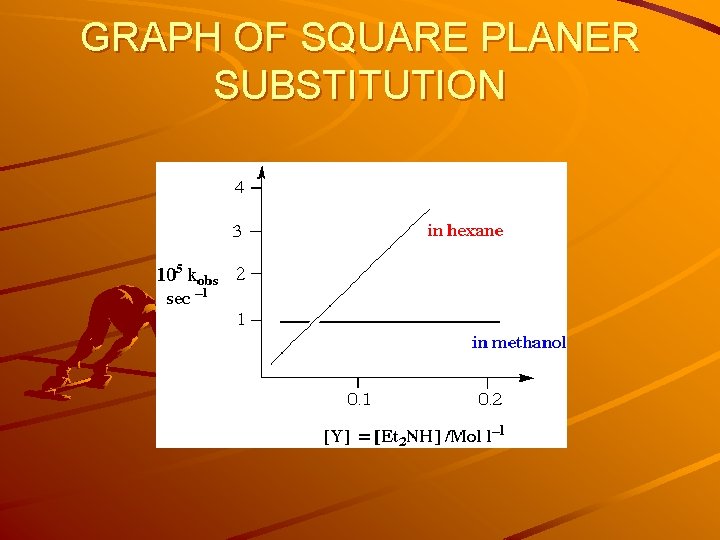
GRAPH OF SQUARE PLANER SUBSTITUTION

REFERENCE • ^ G. L. Miessler and D. A. Tarr. Inorganic Chemistry, 3 rd Ed. , • Pearson/Prentice Hall. . • Miessler and Tarr(inorgnic chemistry)

THE END • Submitted for – www. mycollegebag. in
- Slides: 53