Hybridization How is the bond formed in H
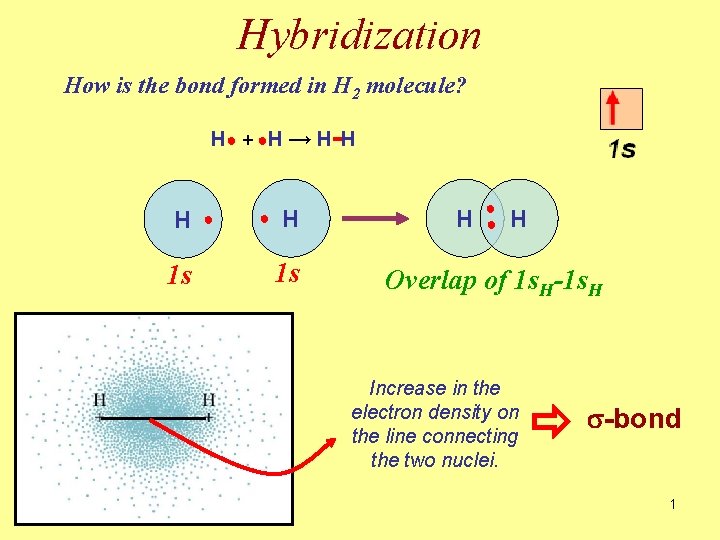
Hybridization How is the bond formed in H 2 molecule? H ● + ●H → H - H H 1 s ● ● H 1 s H ● ● H Overlap of 1 s. H-1 s. H Increase in the electron density on the line connecting the two nuclei. s-bond 1
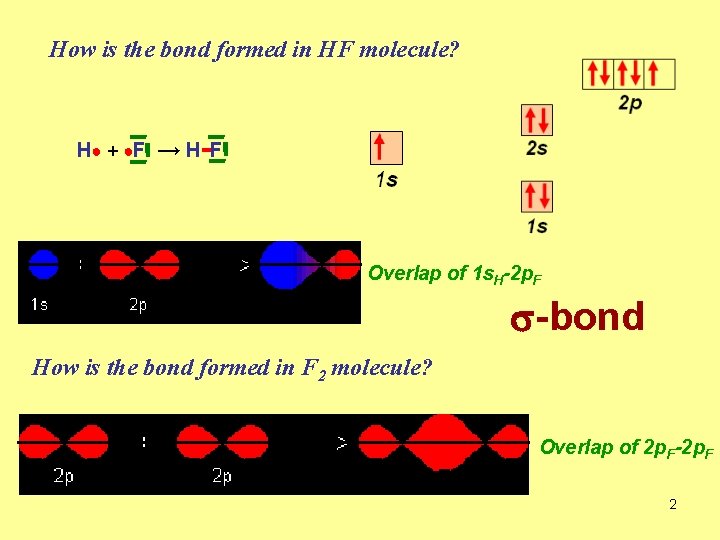
How is the bond formed in HF molecule? H ● + ●F → H -F Overlap of 1 s. H-2 p. F s-bond How is the bond formed in F 2 molecule? Overlap of 2 p. F-2 p. F 2
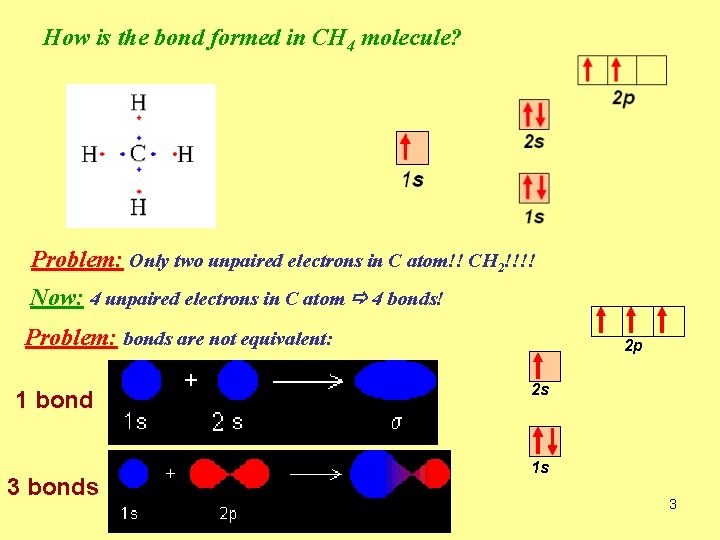
How is the bond formed in CH 4 molecule? Problem: Only two unpaired electrons in C atom!! CH 2!!!! Now: 4 unpaired electrons in C atom 4 bonds! Problem: bonds are not equivalent: 1 bond 3 bonds 2 p 2 s 1 s 3
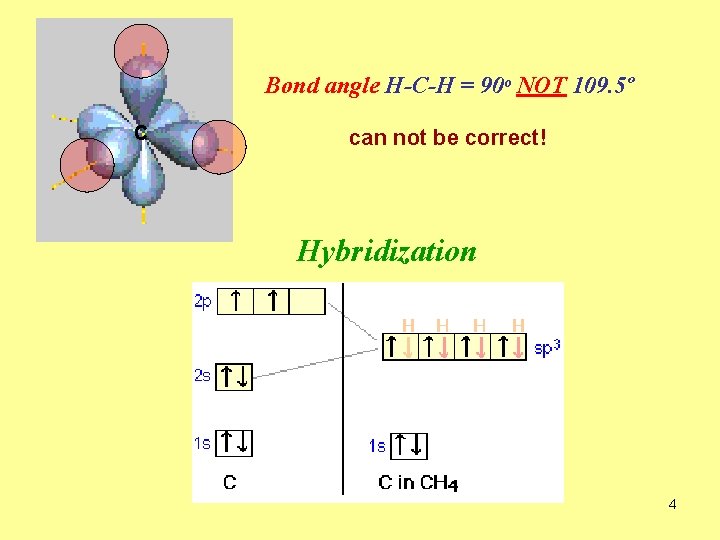
Bond angle H-C-H = 90º NOT 109. 5º C can not be correct! Hybridization 4
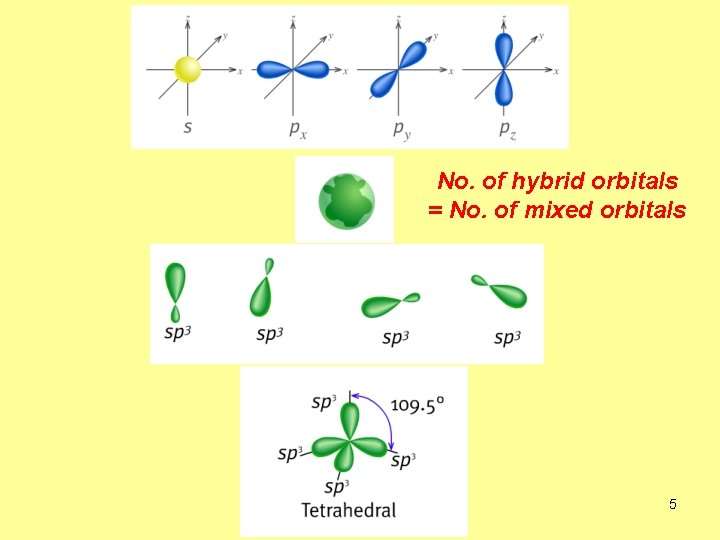
No. of hybrid orbitals = No. of mixed orbitals 5
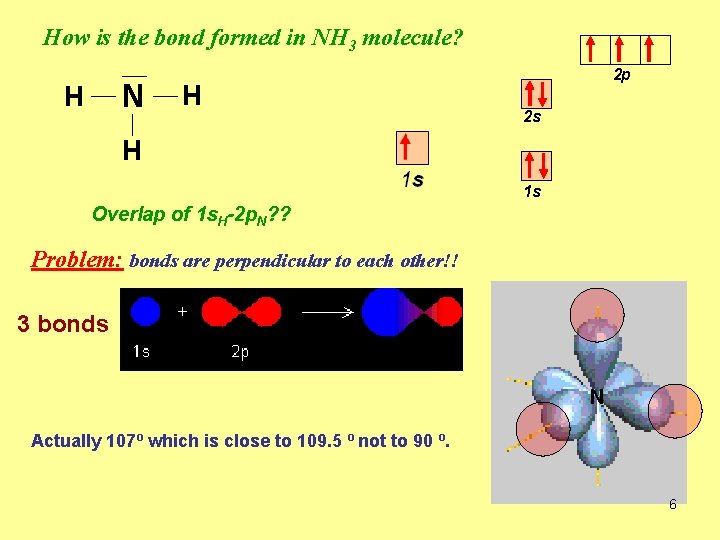
How is the bond formed in NH 3 molecule? N H H 2 p 2 s H 1 s Overlap of 1 s. H-2 p. N? ? Problem: bonds are perpendicular to each other!! 3 bonds N Actually 107º which is close to 109. 5 º not to 90 º. 6
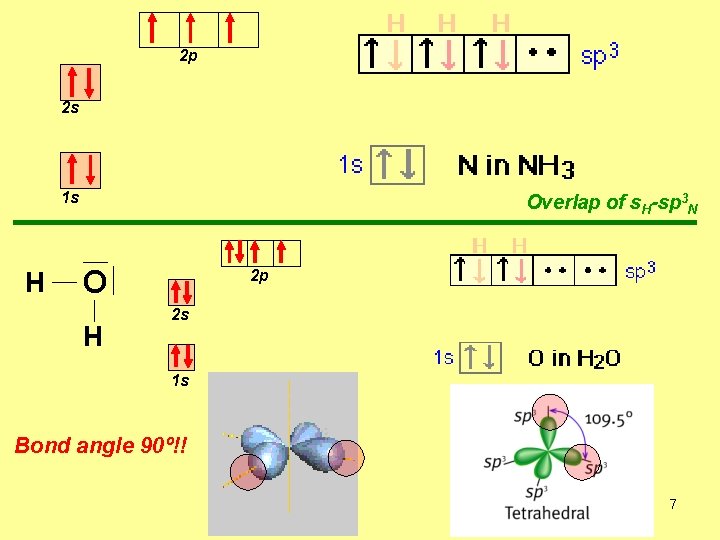
2 p 2 s 1 s H Overlap of s. H-sp 3 N O H 2 p 2 s 1 s Bond angle 90º!! 7

No. of mixed orbitals = No. of s bonds + No. of lone EP • CH 4: 4+0 = 4 (1 s + 3 p) = 4 sp 3 • NH 3: 3+1 = 4 (1 s + 3 p) = 4 sp 3 • H 2 O: 2+2 = 4 (1 s + 3 p) = 4 sp 3 8
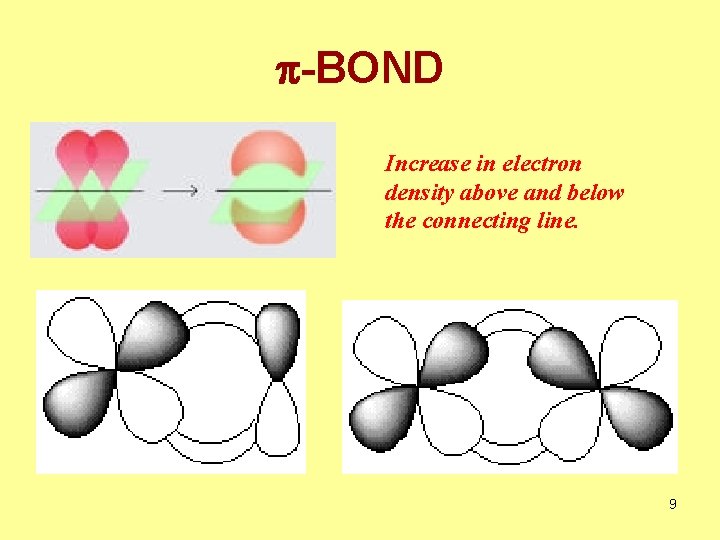
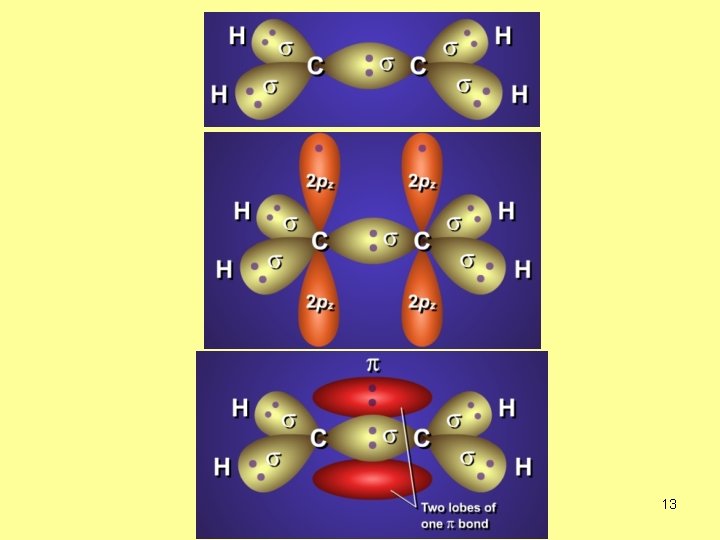
p-BOND Increase in electron density above and below the connecting line. 9
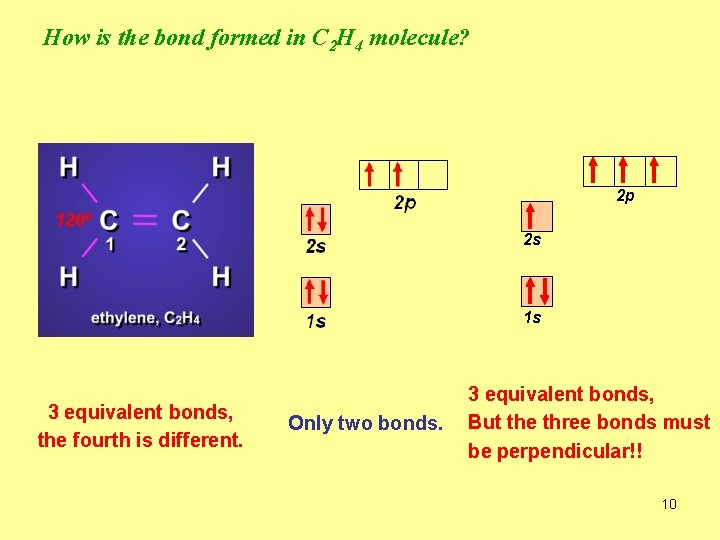
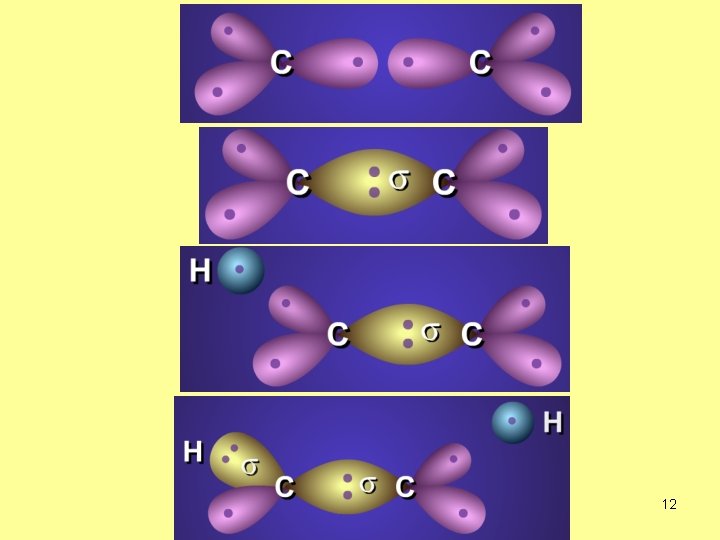
How is the bond formed in C 2 H 4 molecule? 2 p 120º 2 s 1 s 3 equivalent bonds, the fourth is different. Only two bonds. 3 equivalent bonds, But the three bonds must be perpendicular!! 10
11
12
13
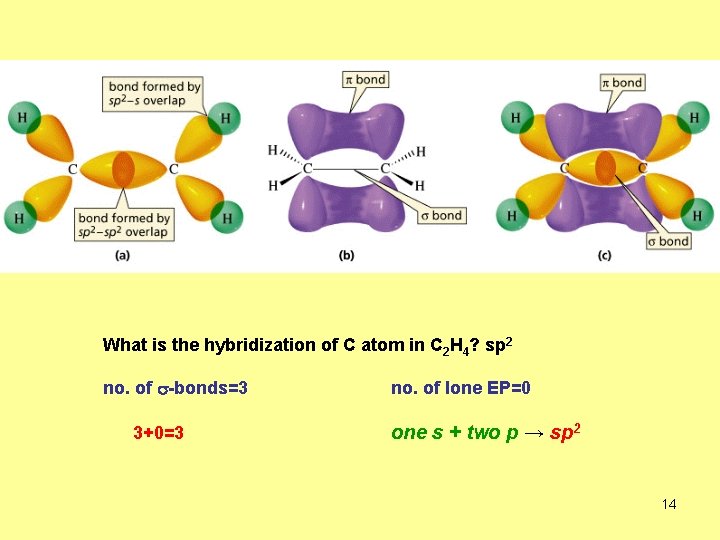
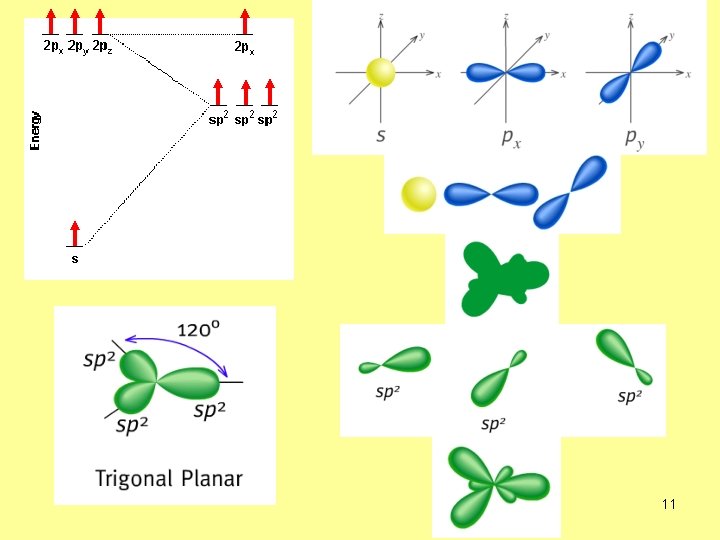
What is the hybridization of C atom in C 2 H 4? sp 2 no. of s-bonds=3 3+0=3 no. of lone EP=0 one s + two p → sp 2 14
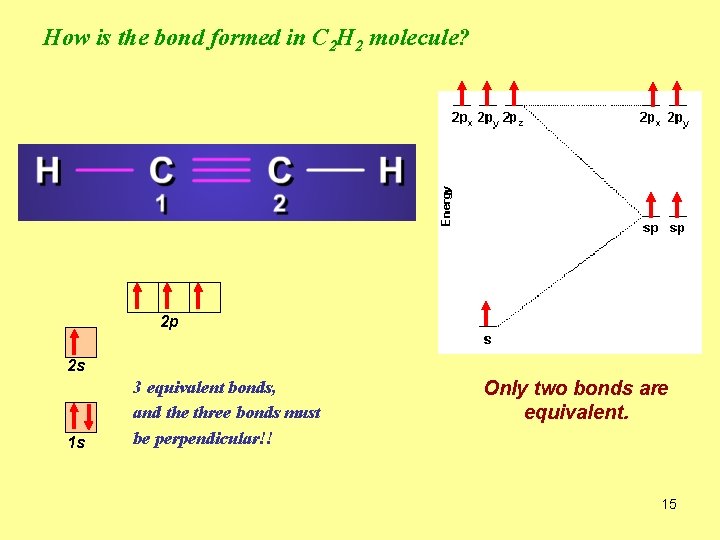
How is the bond formed in C 2 H 2 molecule? 2 p 2 s 1 s 3 equivalent bonds, and the three bonds must be perpendicular!! Only two bonds are equivalent. 15
16
17
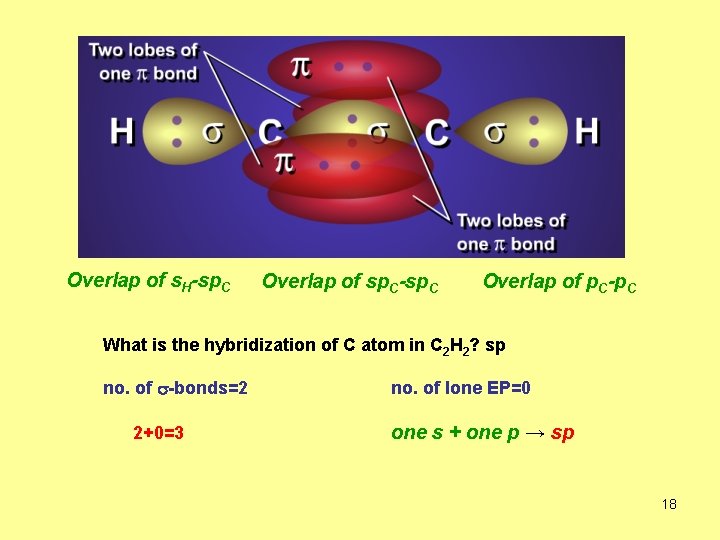
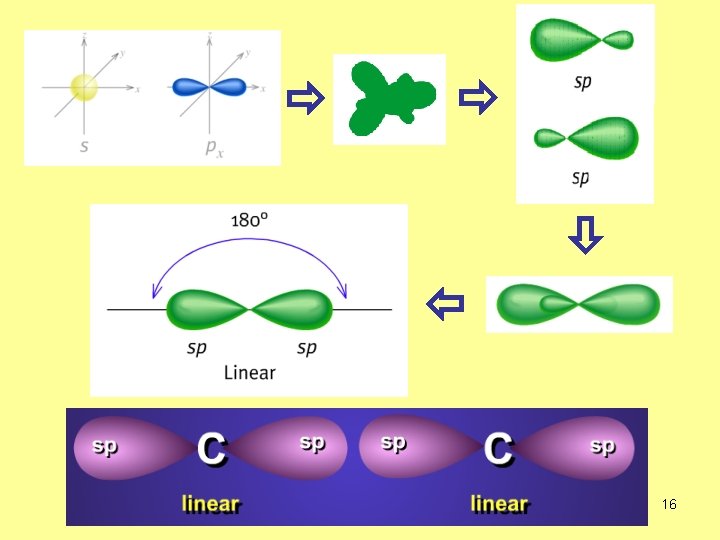
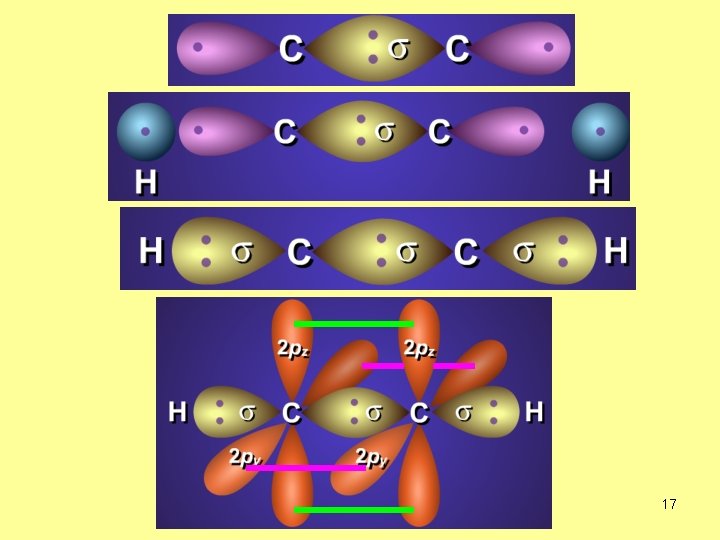
Overlap of s. H-sp. C Overlap of sp. C-sp. C Overlap of p. C-p. C What is the hybridization of C atom in C 2 H 2? sp no. of s-bonds=2 2+0=3 no. of lone EP=0 one s + one p → sp 18
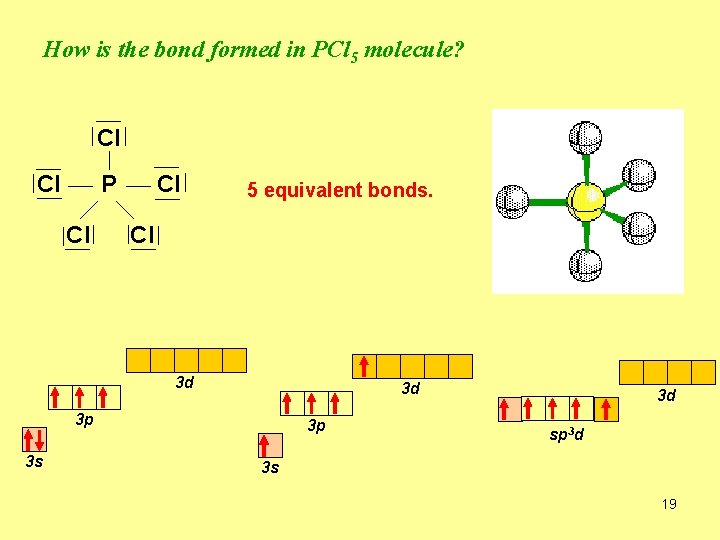
How is the bond formed in PCl 5 molecule? Cl Cl P Cl Cl 5 equivalent bonds. Cl 3 d 3 d 3 p 3 s 3 p 3 d sp 3 d 3 s 19
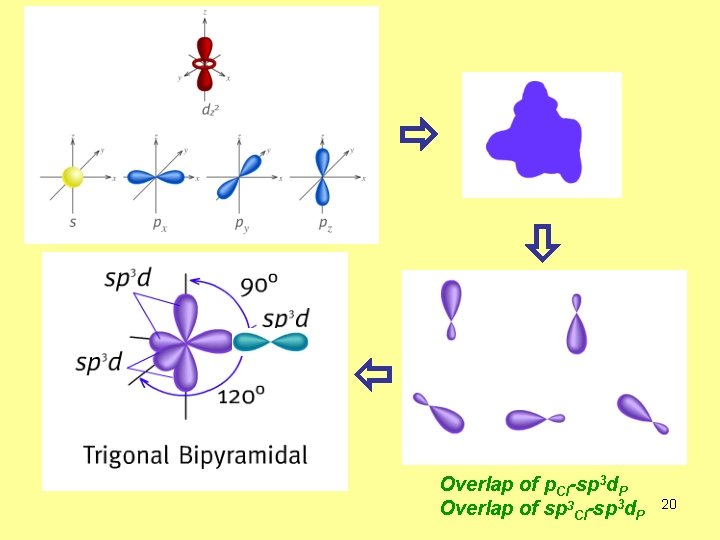
Overlap of p. Cl-sp 3 d. P Overlap of sp 3 Cl-sp 3 d. P 20
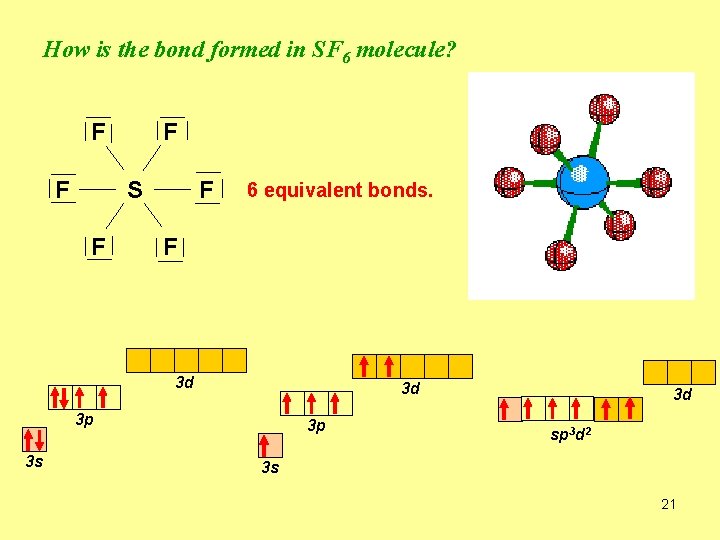
How is the bond formed in SF 6 molecule? F F F S F F 6 equivalent bonds. F 3 d 3 d 3 p 3 s 3 p 3 d sp 3 d 2 3 s 21
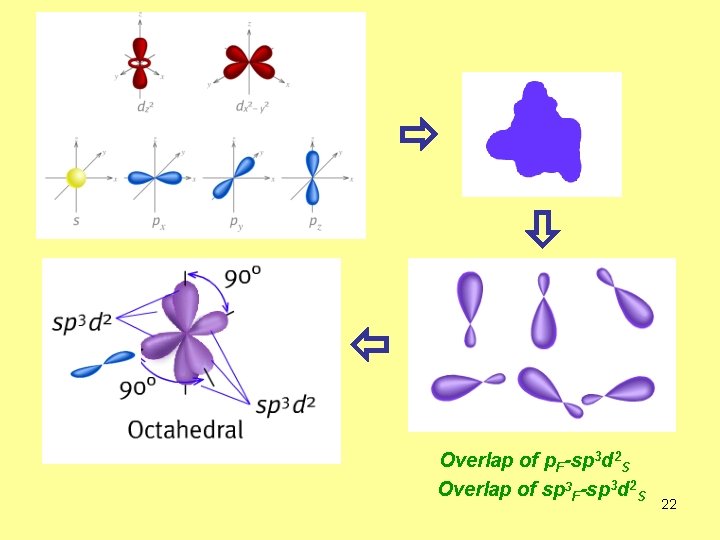
Overlap of p. F-sp 3 d 2 S Overlap of sp 3 F-sp 3 d 2 S 22
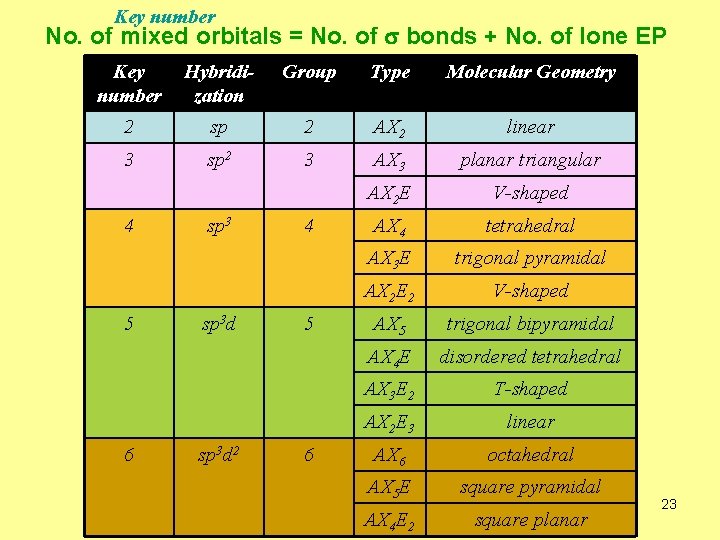
Key number No. of mixed orbitals = No. of s bonds + No. of lone EP Key number Hybridization Group Type Molecular Geometry 2 sp 2 AX 2 linear 3 sp 2 3 AX 3 planar triangular AX 2 E V-shaped AX 4 tetrahedral AX 3 E trigonal pyramidal AX 2 E 2 V-shaped AX 5 trigonal bipyramidal AX 4 E disordered tetrahedral AX 3 E 2 T-shaped AX 2 E 3 linear AX 6 octahedral AX 5 E square pyramidal AX 4 E 2 square planar 4 5 6 sp 3 d 2 4 5 6 23
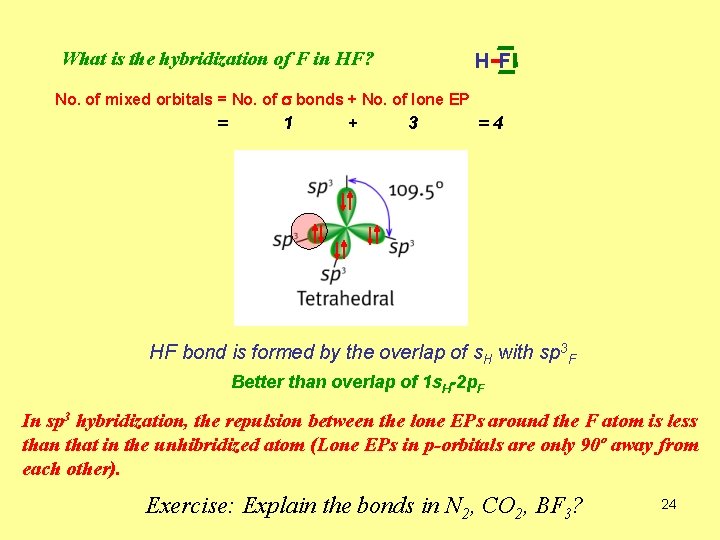
H -F What is the hybridization of F in HF? No. of mixed orbitals = No. of s bonds + No. of lone EP = 1 + 3 =4 HF bond is formed by the overlap of s. H with sp 3 F Better than overlap of 1 s. H-2 p. F In sp 3 hybridization, the repulsion between the lone EPs around the F atom is less than that in the unhibridized atom (Lone EPs in p-orbitals are only 90º away from each other). Exercise: Explain the bonds in N 2, CO 2, BF 3? 24
- Slides: 24