Hybridization College Chemistry VSEPR Theory How can I

Hybridization College Chemistry
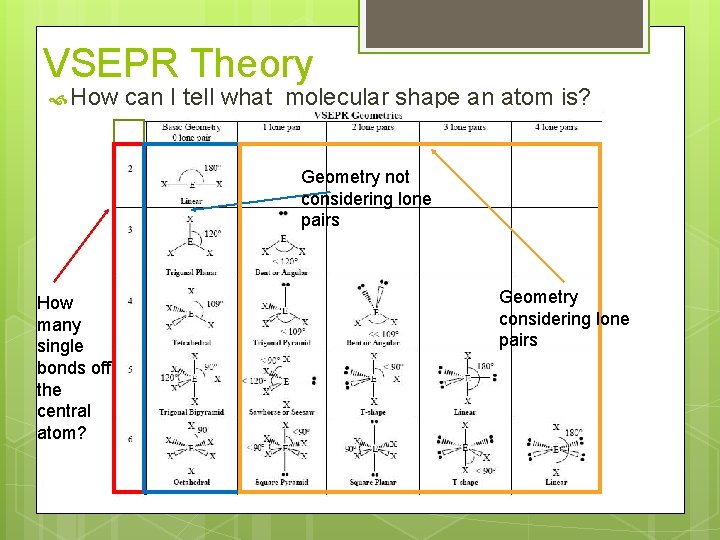
VSEPR Theory How can I tell what molecular shape an atom is? Geometry not considering lone pairs How many single bonds off the central atom? Geometry considering lone pairs
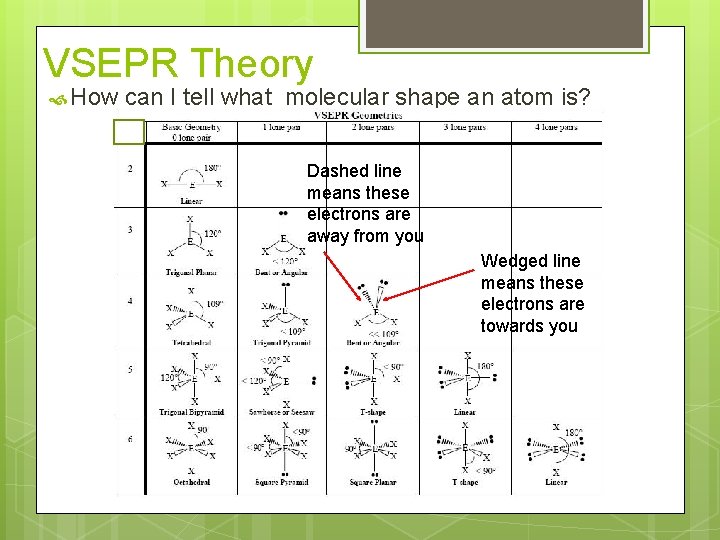
VSEPR Theory How can I tell what molecular shape an atom is? Dashed line means these electrons are away from you Wedged line means these electrons are towards you
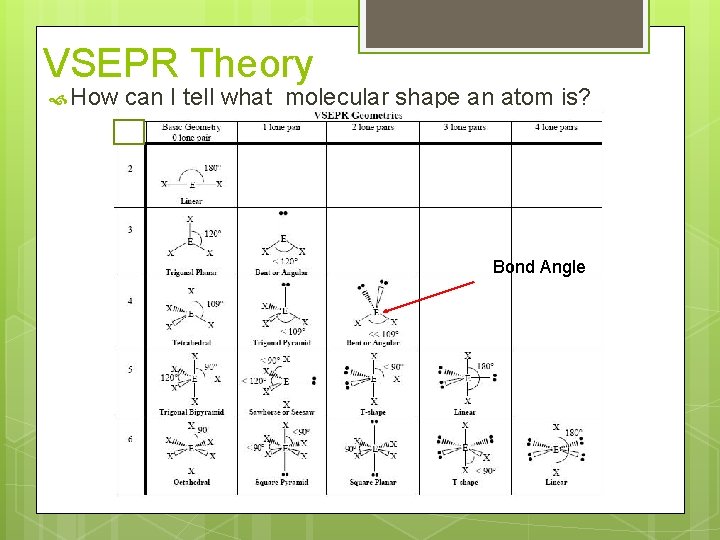
VSEPR Theory How can I tell what molecular shape an atom is? Bond Angle
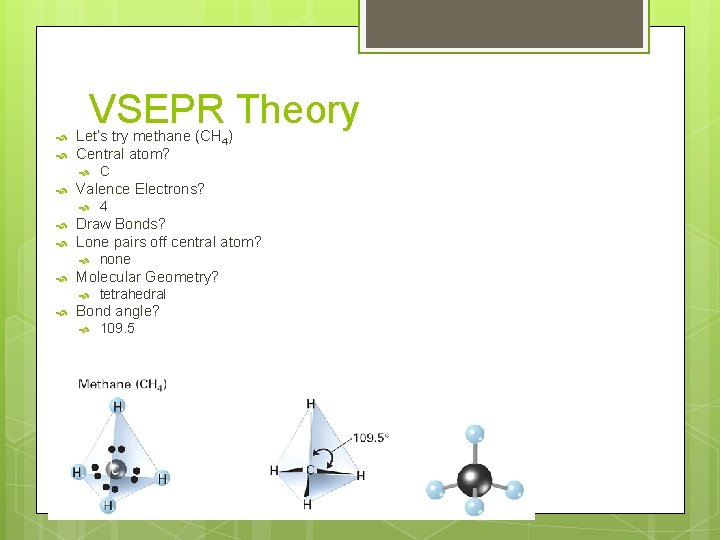
VSEPR Theory Let’s try methane (CH 4) Central atom? Valence Electrons? none Molecular Geometry? 4 Draw Bonds? Lone pairs off central atom? C tetrahedral Bond angle? 109. 5
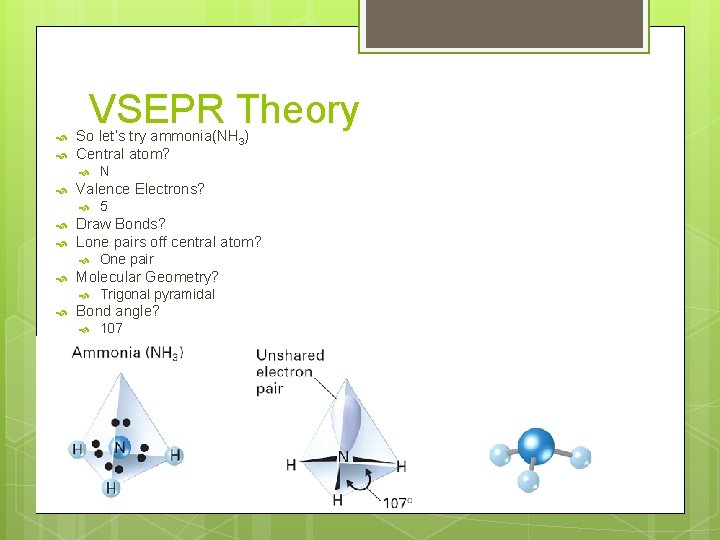
VSEPR Theory So let’s try ammonia(NH 3) Central atom? Valence Electrons? One pair Molecular Geometry? 5 Draw Bonds? Lone pairs off central atom? N Trigonal pyramidal Bond angle? 107

VSEPR Theory Remember those double and triple bonds? We can draw those in VSEPR theory too Only the first bond is counted for bonding purposes (i. e. : a double and triple bond is counted only once) The first bond/single bond – sigma bond (s) Second/third bond – pi bond (p)
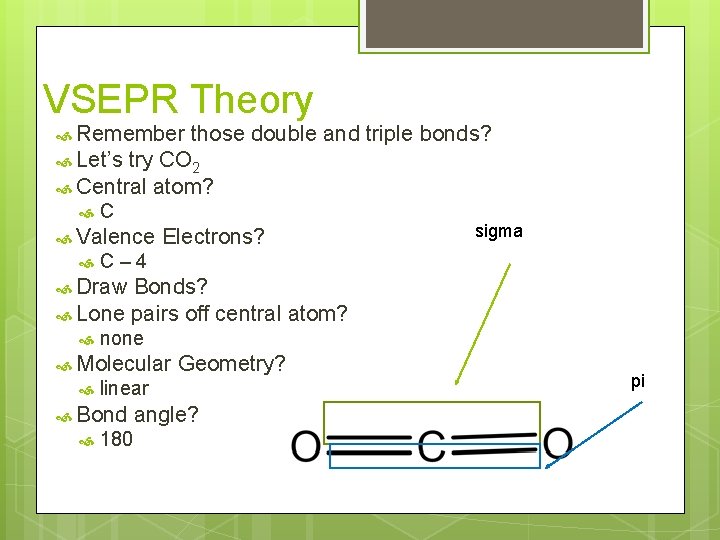
VSEPR Theory Remember those double and triple bonds? Let’s try CO 2 Central atom? C Valence Electrons? sigma C– 4 Draw Bonds? Lone pairs off central atom? none Molecular linear Bond Geometry? 180 angle? pi

Hybridization Hydridization – mix of several atomic orbitals (s and p subshells) to form hybrid oribals Describes the type of bonds formed, which the VSEPR theory CANNOT do!!

8. 3 Hybridization
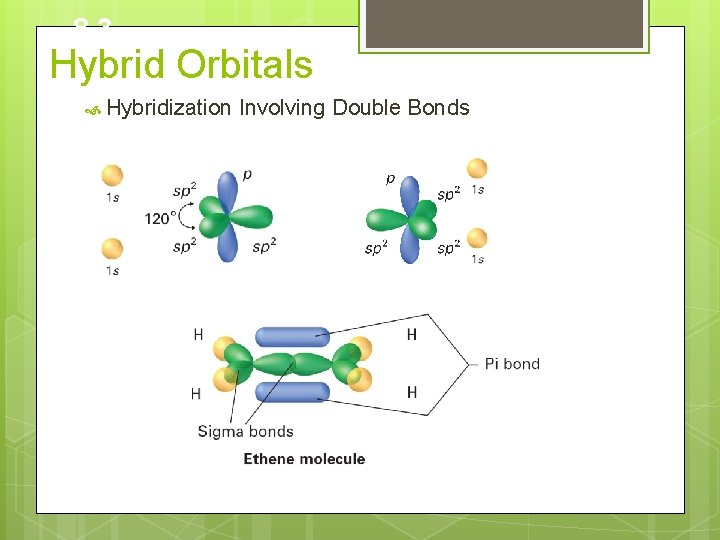
8. 3 Hybrid Orbitals Hybridization Involving Double Bonds
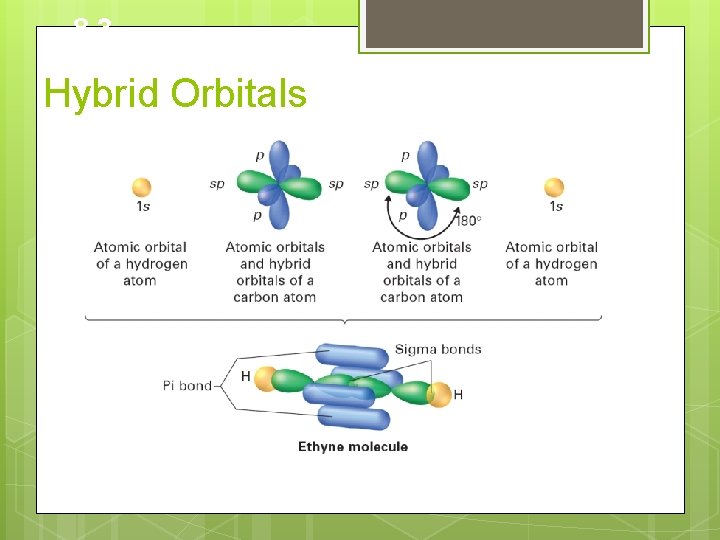
8. 3 Hybrid Orbitals Hybridization Involving Triple Bonds
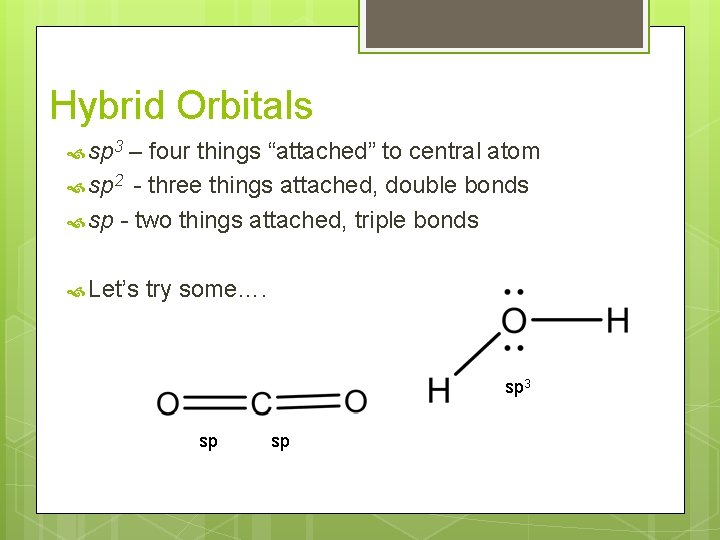
Hybrid Orbitals sp 3 – four things “attached” to central atom sp 2 - three things attached, double bonds sp - two things attached, triple bonds Let’s try some…. sp 3 sp sp
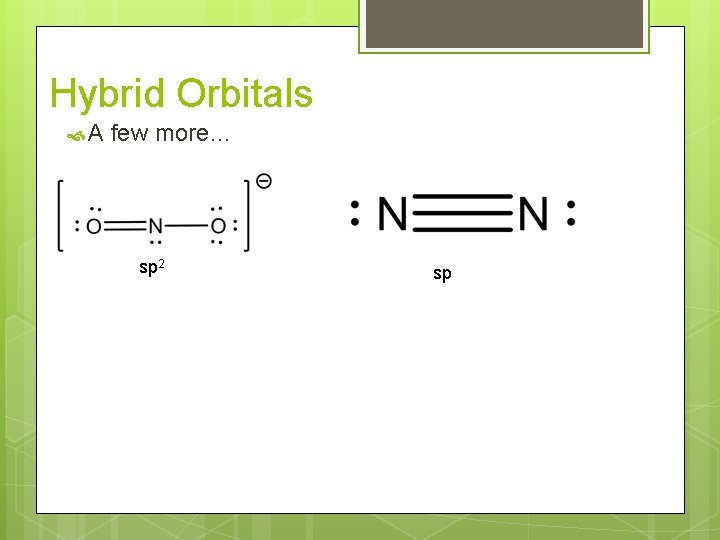
Hybrid Orbitals A few more… sp 2 sp
- Slides: 14