Hybridization Carbon configuration Carbon in excited state can
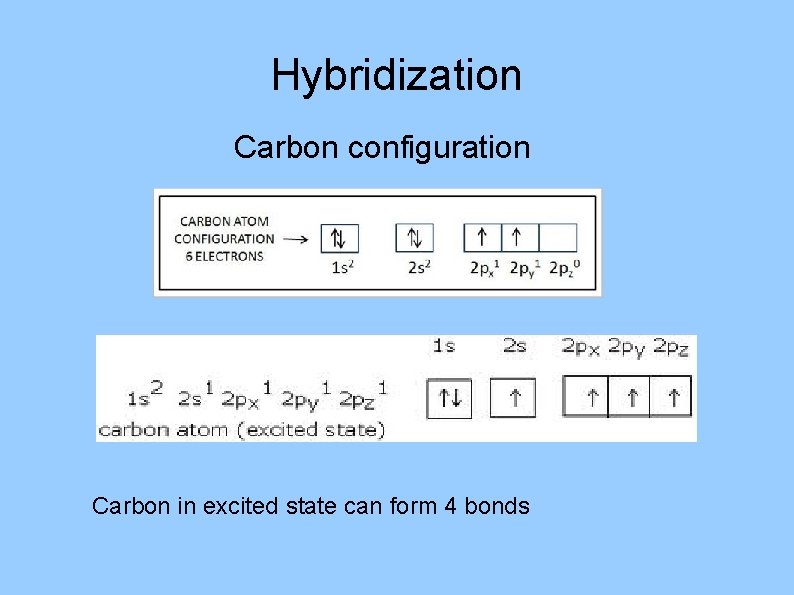
Hybridization Carbon configuration Carbon in excited state can form 4 bonds
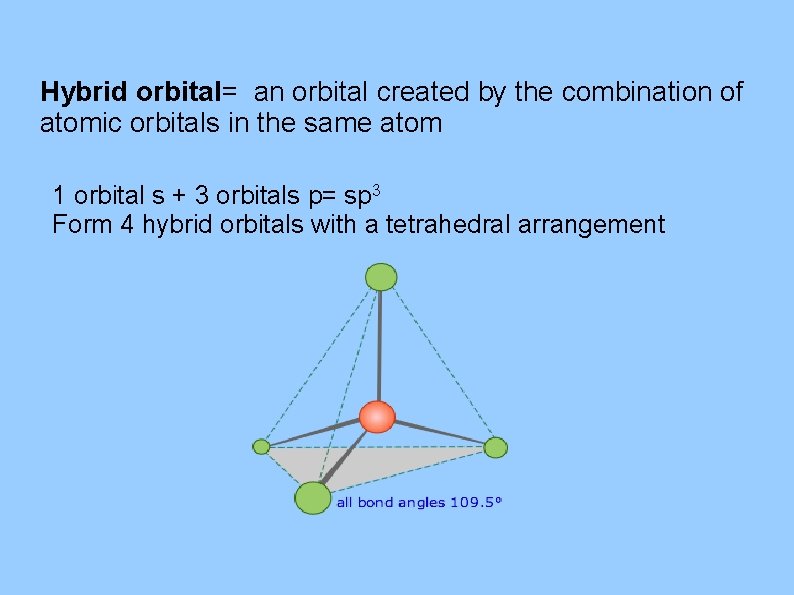
Hybrid orbital= an orbital created by the combination of atomic orbitals in the same atom 1 orbital s + 3 orbitals p= sp 3 Form 4 hybrid orbitals with a tetrahedral arrangement

Energy changes occurring in hybridization Hybridization of an s orbital with all three p orbitals (px , py, and pz) results in four sp 3 hybrid orbitals are oriented at bond angle of 109. 5 degrees from each other. This 109. 5 degree arrangement gives tetrahedral geometry
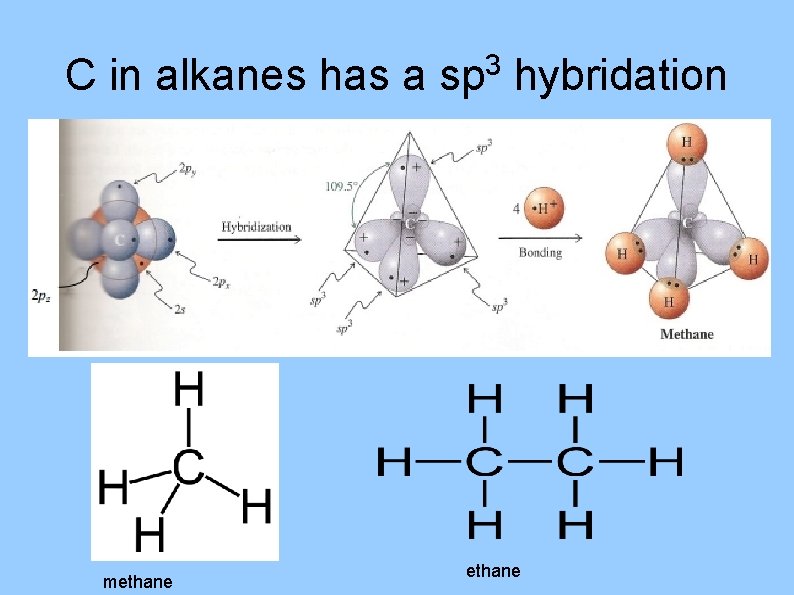
C in alkanes has a methane 3 sp hybridation ethane
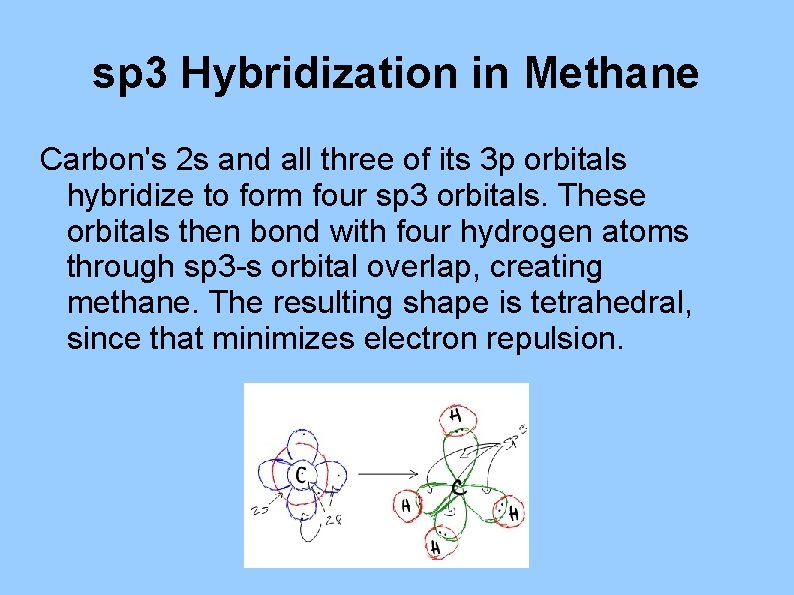
sp 3 Hybridization in Methane Carbon's 2 s and all three of its 3 p orbitals hybridize to form four sp 3 orbitals. These orbitals then bond with four hydrogen atoms through sp 3 -s orbital overlap, creating methane. The resulting shape is tetrahedral, since that minimizes electron repulsion.
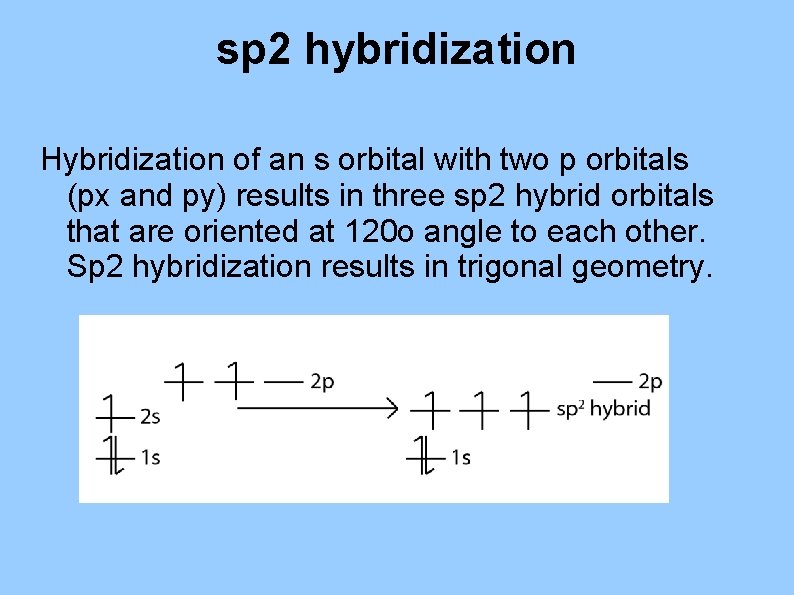
sp 2 hybridization Hybridization of an s orbital with two p orbitals (px and py) results in three sp 2 hybrid orbitals that are oriented at 120 o angle to each other. Sp 2 hybridization results in trigonal geometry.
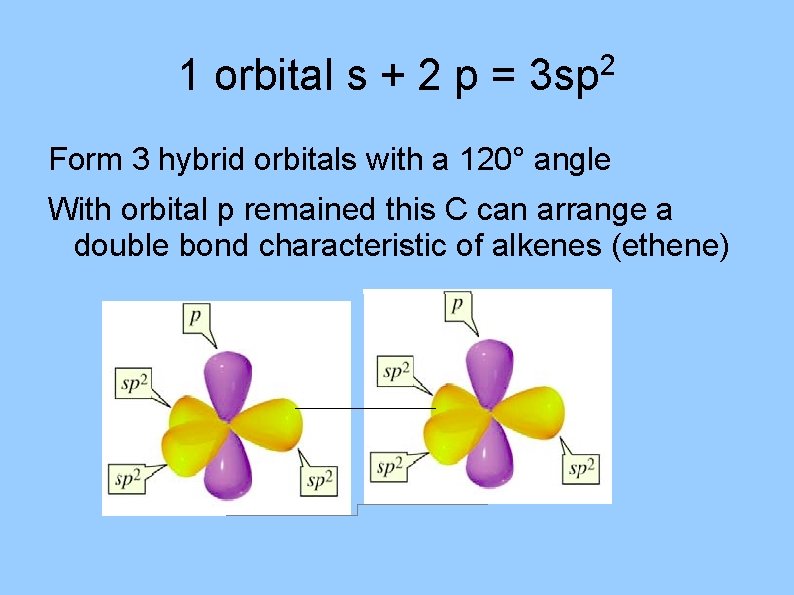
1 orbital s + 2 p = 2 3 sp Form 3 hybrid orbitals with a 120° angle With orbital p remained this C can arrange a double bond characteristic of alkenes (ethene)
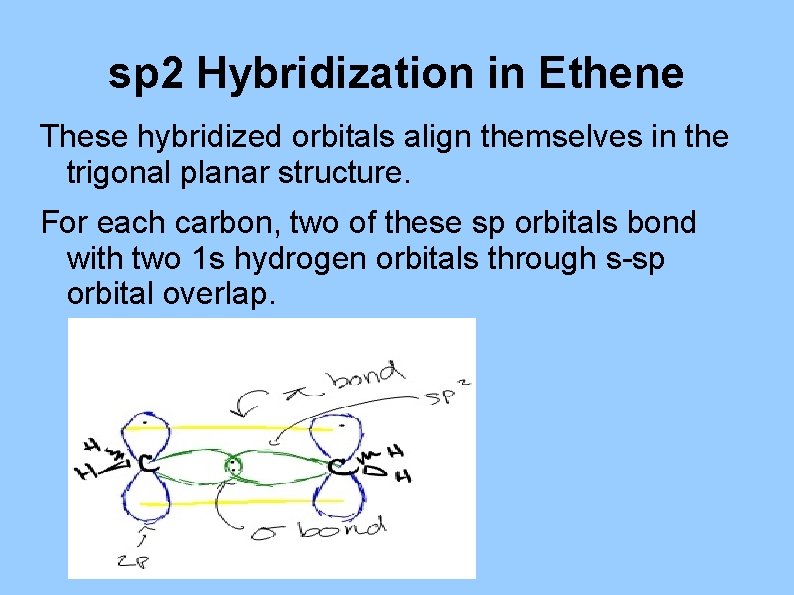
sp 2 Hybridization in Ethene These hybridized orbitals align themselves in the trigonal planar structure. For each carbon, two of these sp orbitals bond with two 1 s hydrogen orbitals through s-sp orbital overlap.
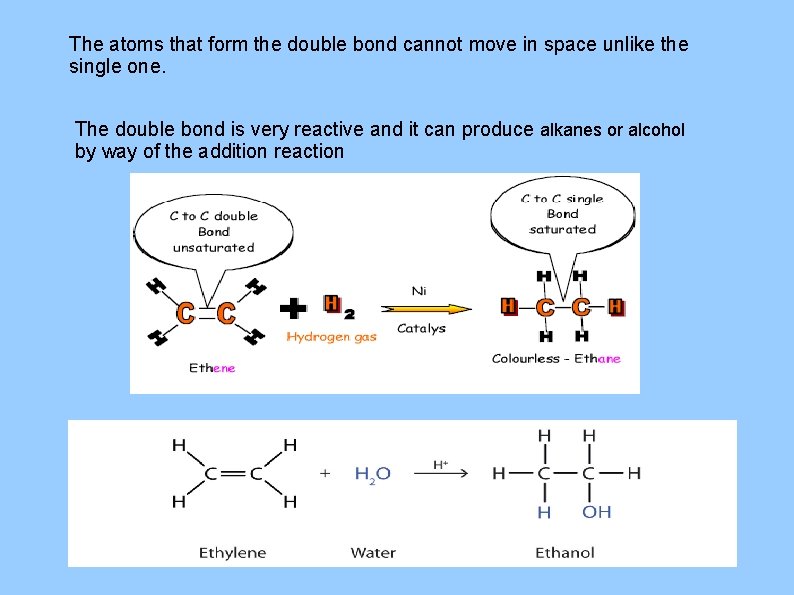
The atoms that form the double bond cannot move in space unlike the single one. The double bond is very reactive and it can produce alkanes or alcohol by way of the addition reaction
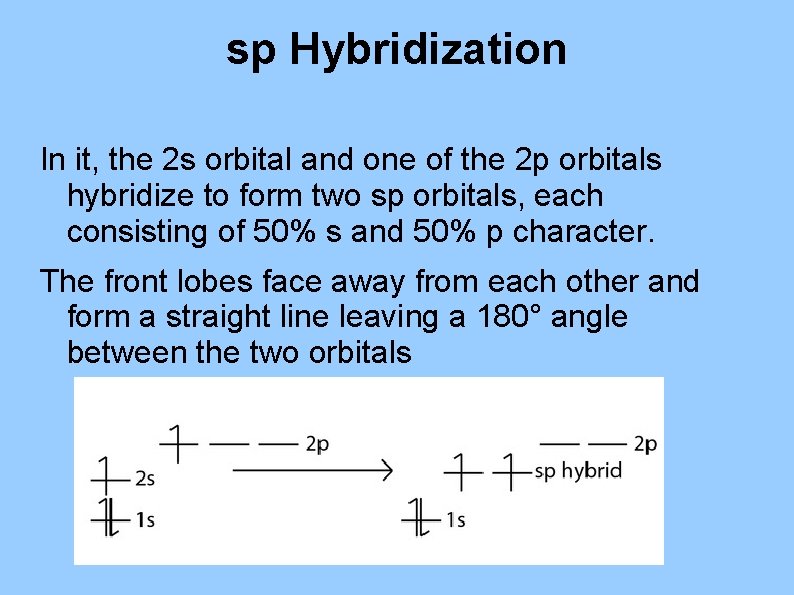
sp Hybridization In it, the 2 s orbital and one of the 2 p orbitals hybridize to form two sp orbitals, each consisting of 50% s and 50% p character. The front lobes face away from each other and form a straight line leaving a 180° angle between the two orbitals
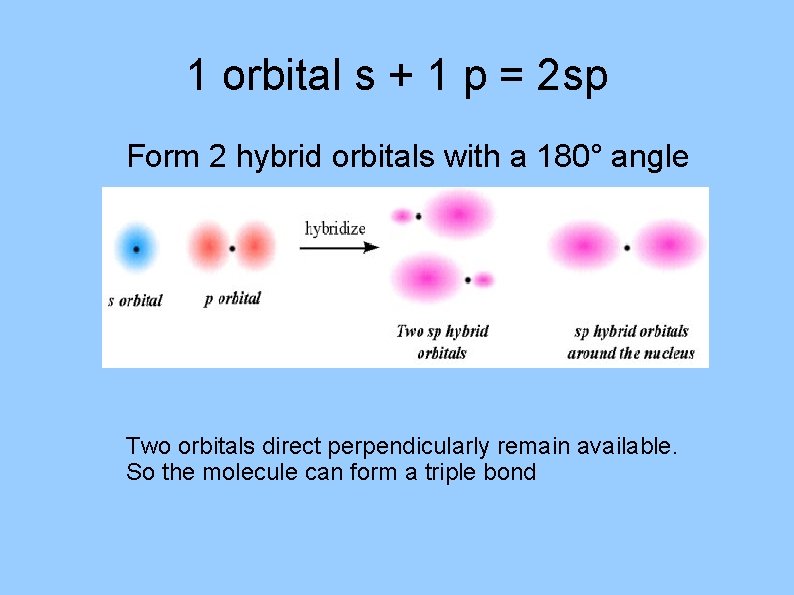
1 orbital s + 1 p = 2 sp Form 2 hybrid orbitals with a 180° angle Two orbitals direct perpendicularly remain available. So the molecule can form a triple bond
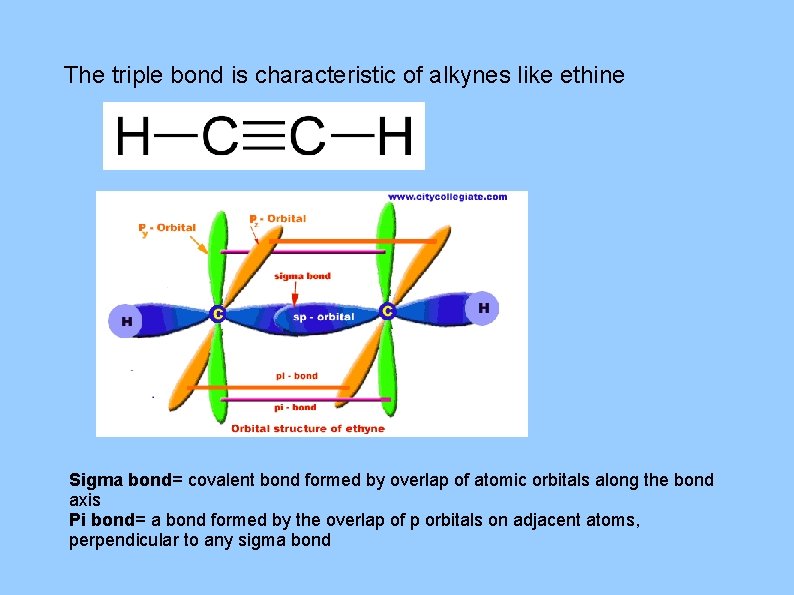
The triple bond is characteristic of alkynes like ethine Sigma bond= covalent bond formed by overlap of atomic orbitals along the bond axis Pi bond= a bond formed by the overlap of p orbitals on adjacent atoms, perpendicular to any sigma bond
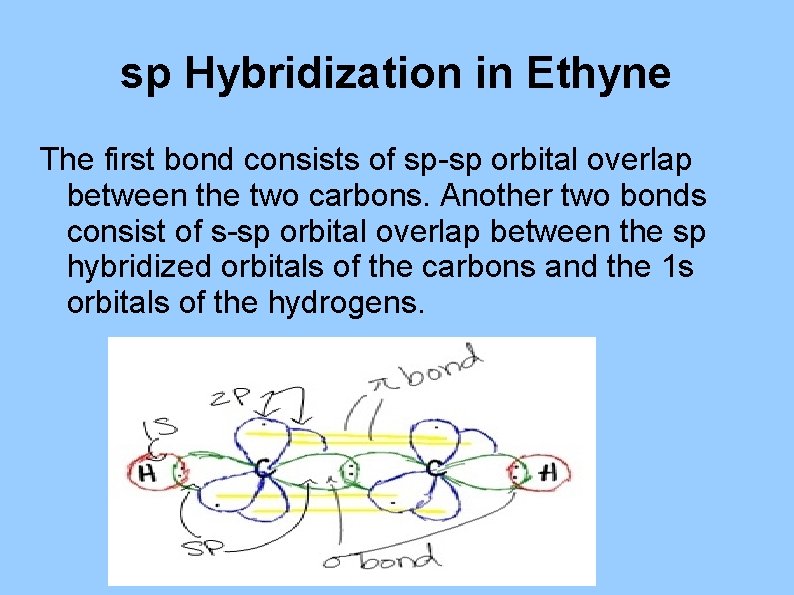
sp Hybridization in Ethyne The first bond consists of sp-sp orbital overlap between the two carbons. Another two bonds consist of s-sp orbital overlap between the sp hybridized orbitals of the carbons and the 1 s orbitals of the hydrogens.
- Slides: 13