Hybridization A Blending of Orbitals Methane CH 4

Hybridization A Blending of Orbitals
Methane CH 4 n Sometimes called “natural gas, ” methane is used to heat homes. n

Shape of Methane n Tetrahedral geometry n 109. 5 o angle between bonds

Carbon’s atomic orbitals n 4 valence electrons n Outermost orbitals: 2 s, 2 px, 2 py, 2 pz
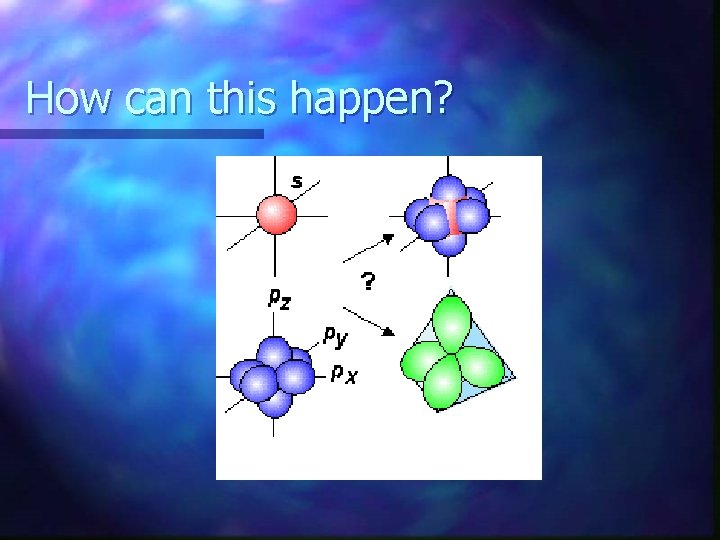
How can this happen?

2 possibilities n Maybe our quantum mechanical model of atomic orbitals is totally wrong. OR n Maybe carbon is doing something else with its orbitals to form this compound.

Hybridization occurs n. A mathematical blending of orbitals n Number of atomic orbitals blended = number of hybrid orbitals produced n Result: Identical orbitals – New shape – New orientation in space

sp 3 hybridization ns + px + Py + pz = 4 sp 3 orbitals

Sp 3 hybridization on carbon 4 identical sp 3 orbitals n 109. 5 o between orbitals n Tetrahedral orientation n
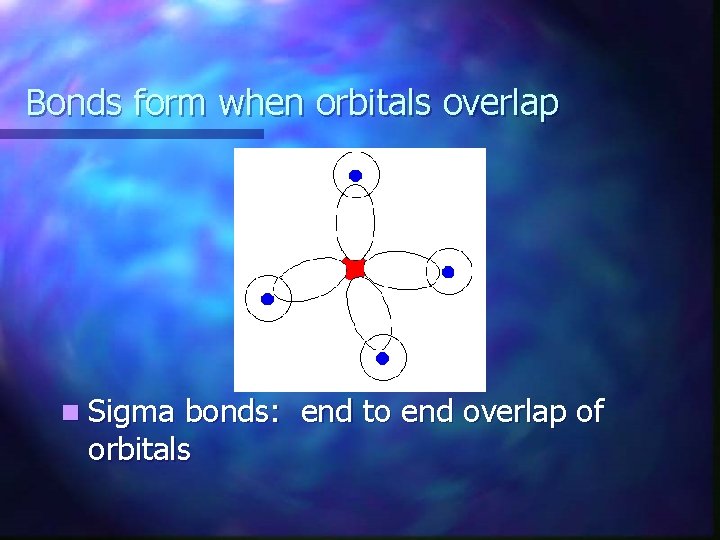
Bonds form when orbitals overlap n Sigma bonds: end to end overlap of orbitals

What happens in ethene? C 2 H 4 n Properties n Double bond between carbons n Trigonal planar geometry around C n 120 o bond angles

Sp 2 hybridization occurs n. S + px + py = 3 sp 2 orbitals n 1 unused p orbital left over
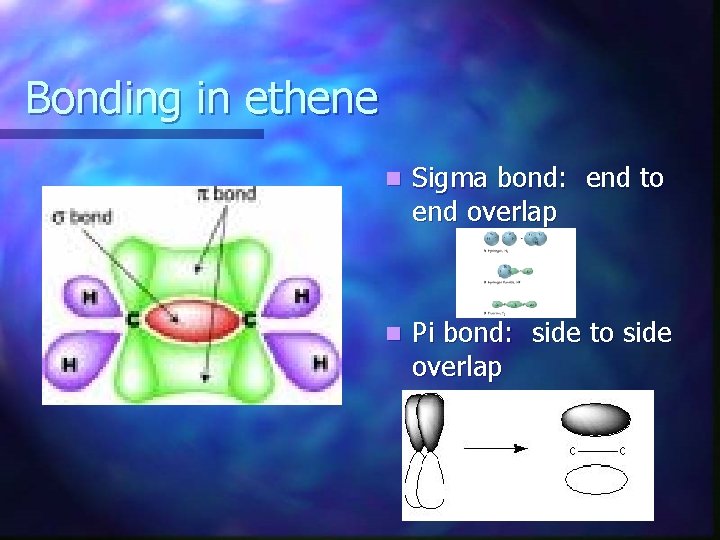
Bonding in ethene n Sigma bond: end to end overlap n Pi bond: side to side overlap

Putting ethene together Carbon-carbon double bond = 1 sigma bond and 1 Pi bond n 4 C-H sigma bonds n Trigonal planar geometry results n

What happens in ethyne? n C 2 H 2 n Linear geometry n What type of hybridization is involved? n What hybrid and atomic orbitals can it use? n What types and numbers of bonds are present in the molecule?
- Slides: 15