Hybrid phatogenesis of MDS Myeloproliferative diseases de novo

Hybrid phatogenesis of MDS Myeloproliferative diseases “de novo” Acute Leukemias Defect in the proliferative process Defect in the differentiation process MDS: Defect in the proliferative and differentiative processes
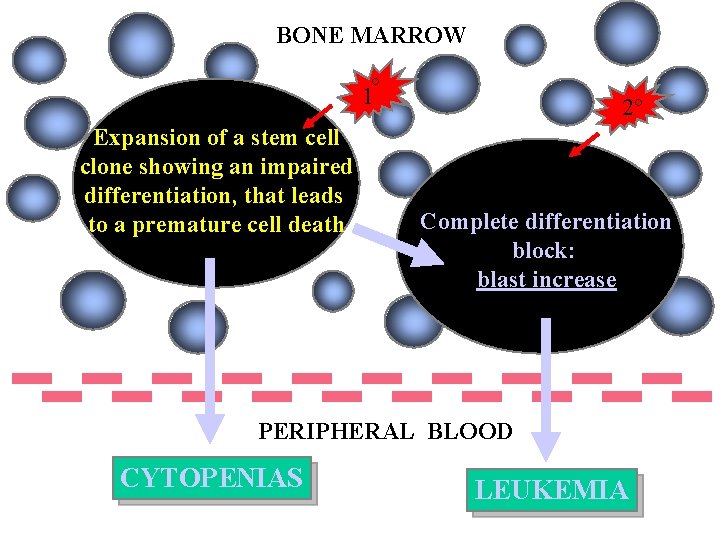
BONE MARROW 1° Expansion of a stem cell clone showing an impaired differentiation, that leads to a premature cell death 2° Complete differentiation block: blast increase PERIPHERAL BLOOD CYTOPENIAS LEUKEMIA
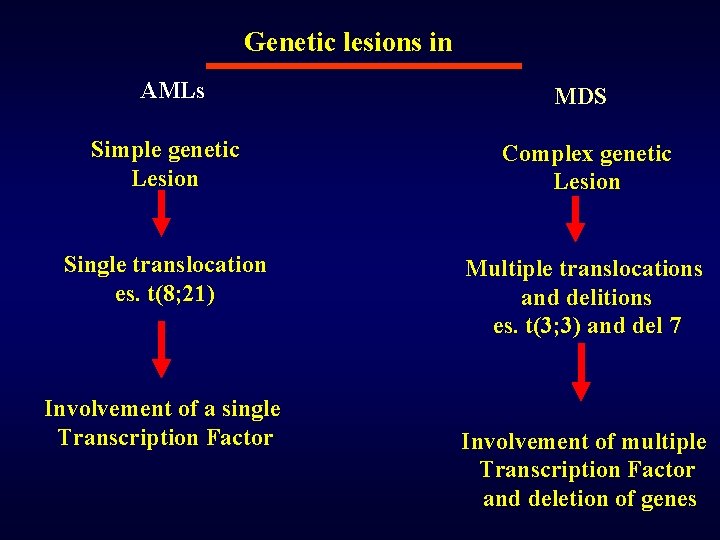
Genetic lesions in AMLs MDS Simple genetic Lesion Complex genetic Lesion Single translocation es. t(8; 21) Multiple translocations and delitions es. t(3; 3) and del 7 Involvement of a single Transcription Factor Involvement of multiple Transcription Factor and deletion of genes
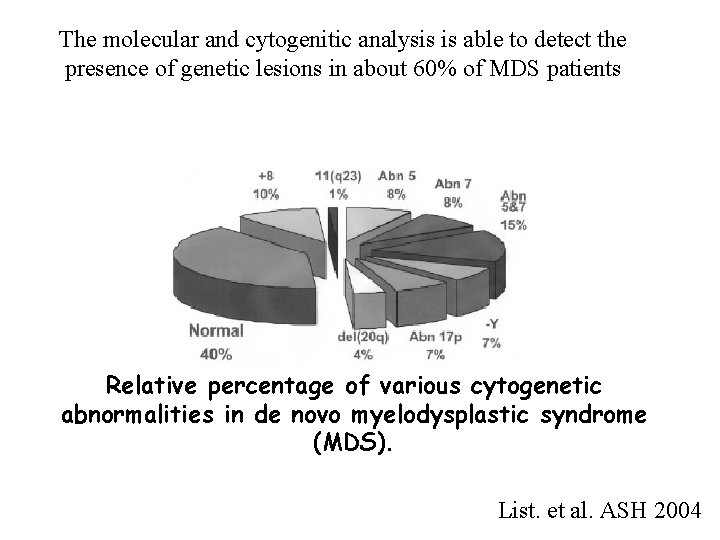
The molecular and cytogenitic analysis is able to detect the presence of genetic lesions in about 60% of MDS patients Relative percentage of various cytogenetic abnormalities in de novo myelodysplastic syndrome (MDS). List. et al. ASH 2004
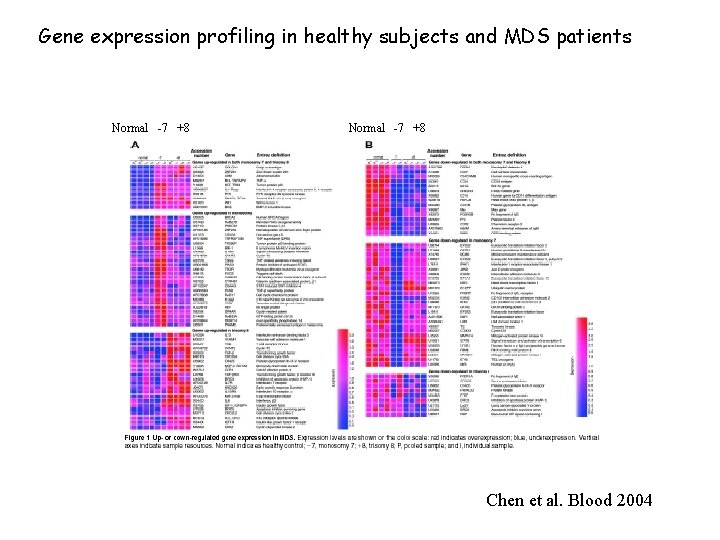
Gene expression profiling in healthy subjects and MDS patients Normal -7 +8 Chen et al. Blood 2004
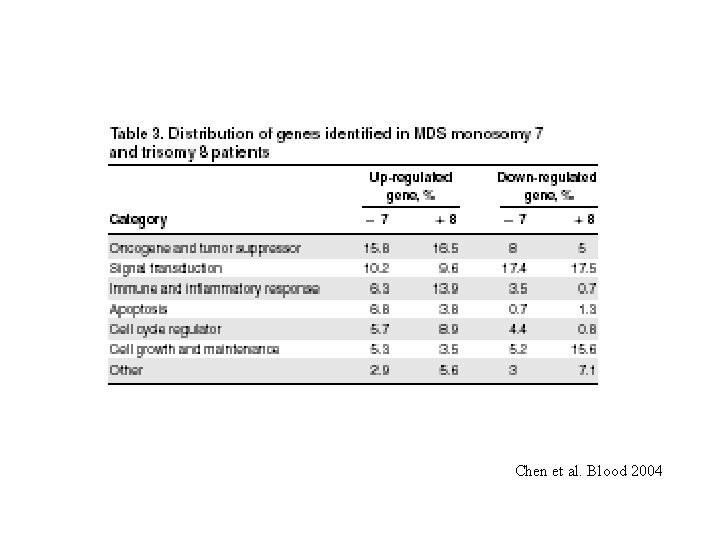
Chen et al. Blood 2004

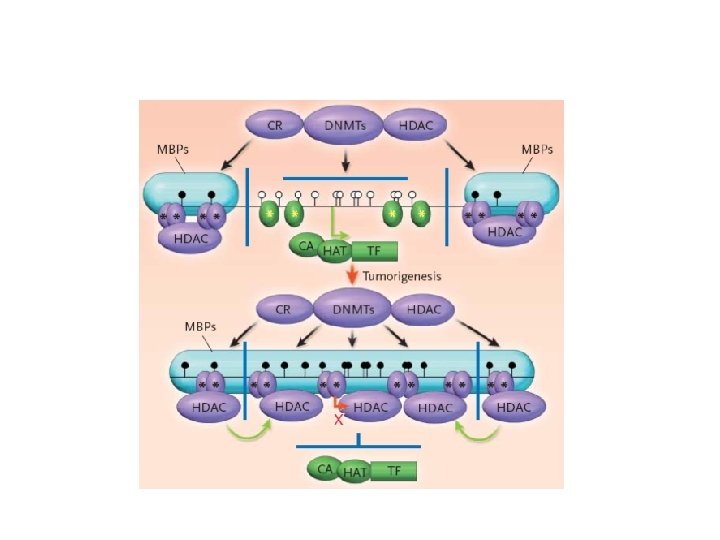
Physiopathology of MDS Genetic Abnormalities -Chromosomal (+8, -7, -5, 5 q-, 7 q-, 20 q-) -Point Mutations (RAS) Epigenetic Modifications -Aberrant DNA methylation -Aberrant acetylation Dysregulation of cytokine production -Abnormal rate of apoptosis & proliferation

Epigenetic modifications Potentially reversible DNA and chromatin modifications transmissed from a cell to its progeny, able to induce altered gene expression without changing DNA sequence and without any “new” genetic information

Epigenetic modifications DNA Methylation: embryogenesis, differentiation, imprinting, X inactivation, cancer Post translational modifications of proteins with subsequent chromatin alteration: Histone acetylation H. Methylation H. Phosphorylation H. Ubiquitination

Nucleasome structure • Acetylation of lysines • Methylation of lysines and arginines • Phosphorylation of serines and threonines • Ubiquitination and sumulyation of lysines
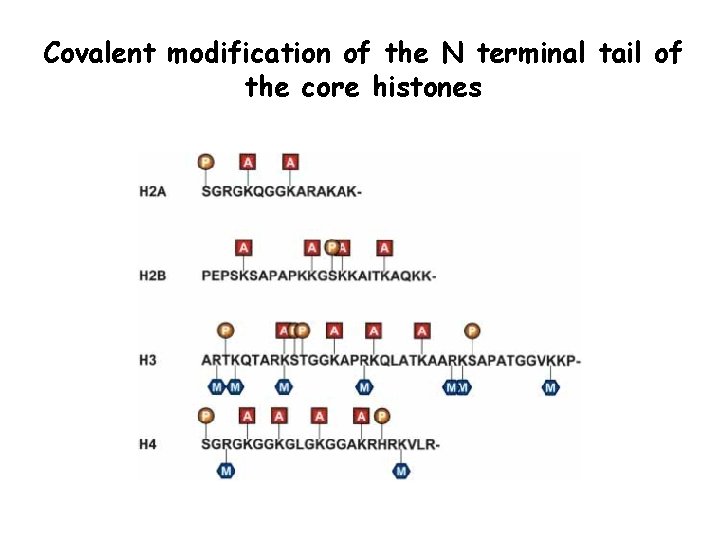
Covalent modification of the N terminal tail of the core histones
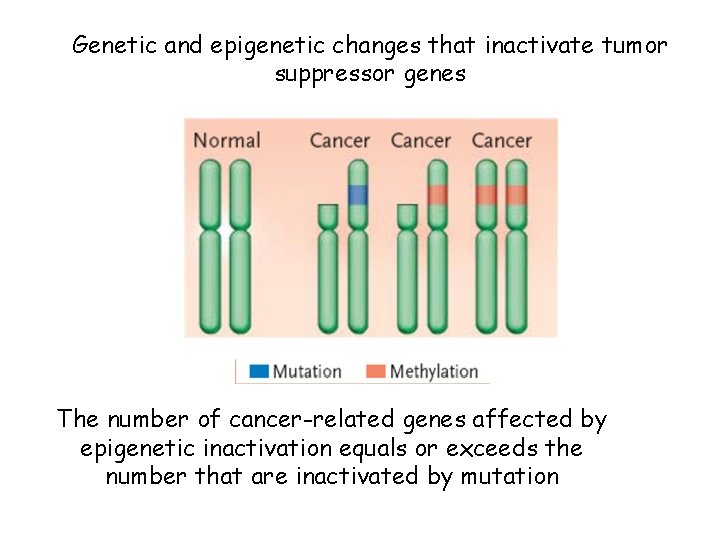
Genetic and epigenetic changes that inactivate tumor suppressor genes The number of cancer-related genes affected by epigenetic inactivation equals or exceeds the number that are inactivated by mutation

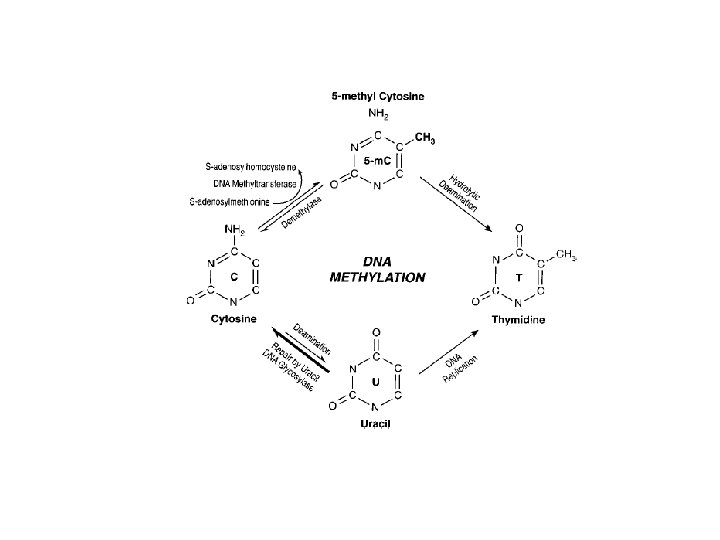
DNA Methylation • DNA methylation is a covalent chemical modification, adding a CH 3 group at the carbon 5 position of cytosine situated in the sequence contest 5’CG 3’. Its frequence is lower than predictable by sequence. • Cp. G dinucleotide clustered in promoter regions of 50% of human genes (Cp. G Islands 0. 5 – 5 kb every 100 kb). • Cp. G islands are generally unmethylated in normal cells • Methylation of Cp. G islands determines gene silencing
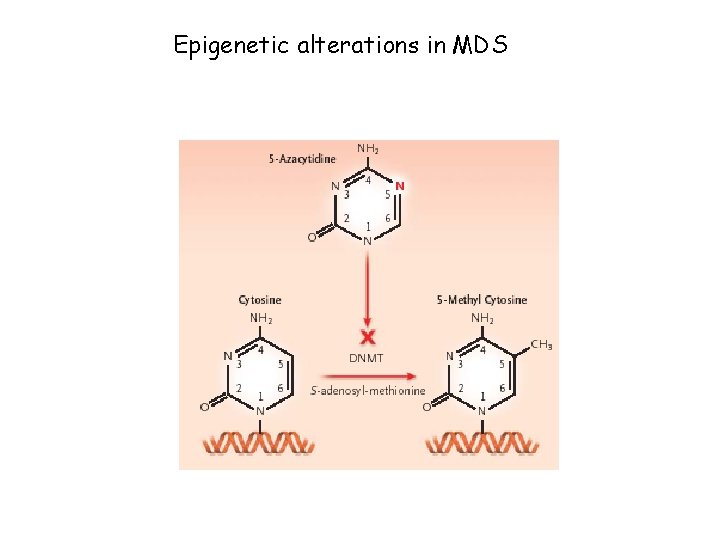
Epigenetic alterations in MDS
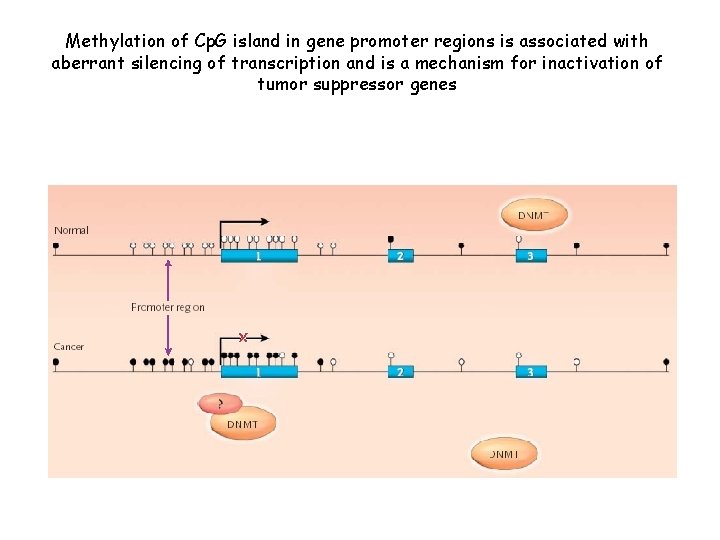
Methylation of Cp. G island in gene promoter regions is associated with aberrant silencing of transcription and is a mechanism for inactivation of tumor suppressor genes
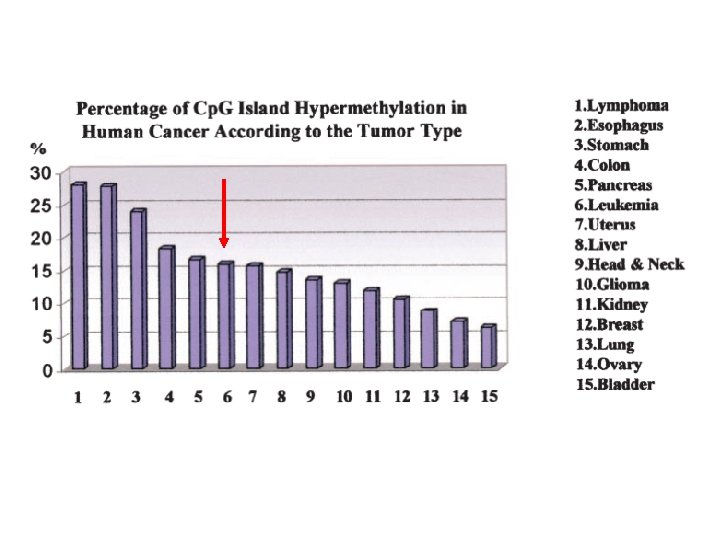
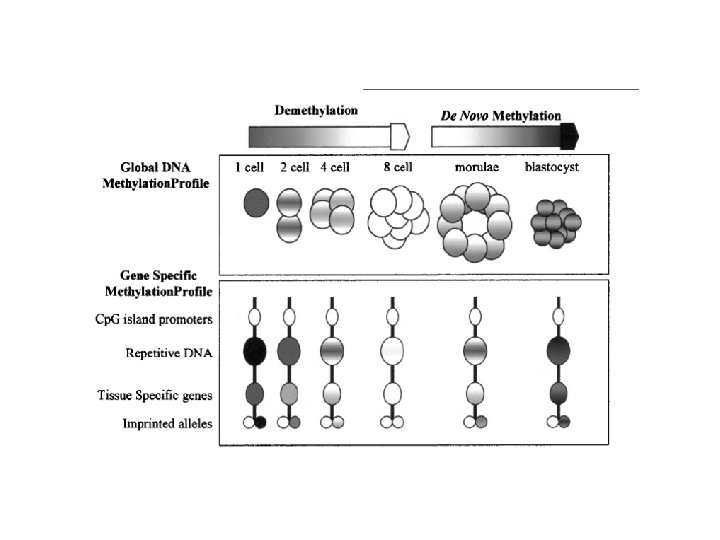
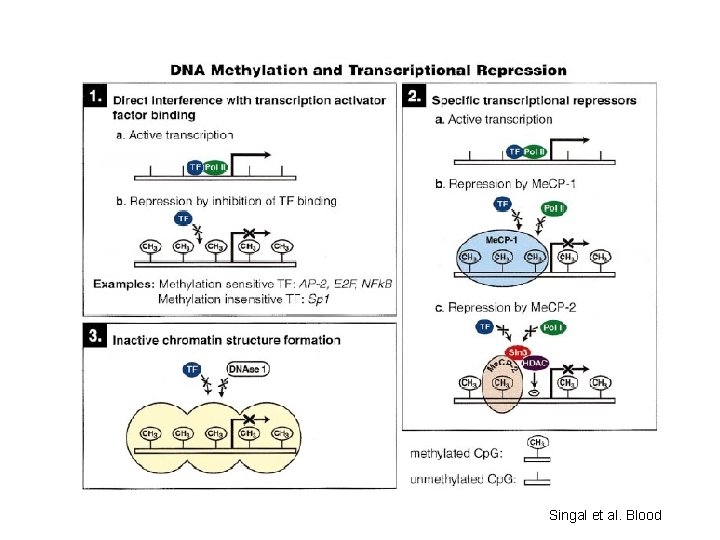
Singal et al. Blood
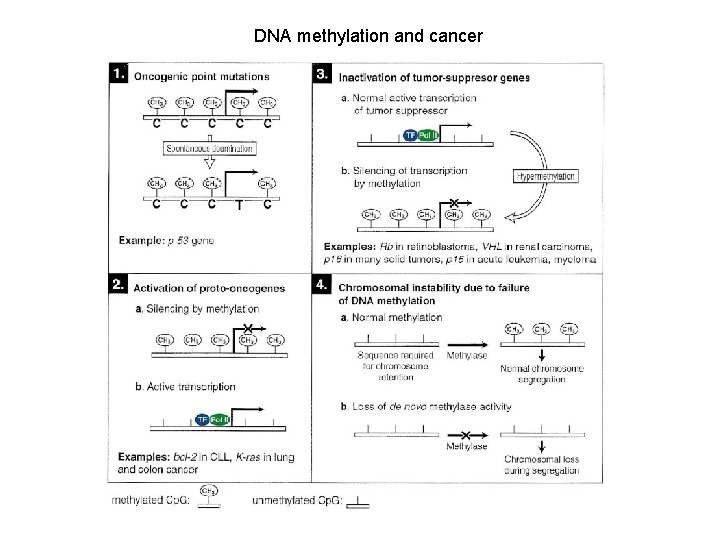
DNA methylation and cancer
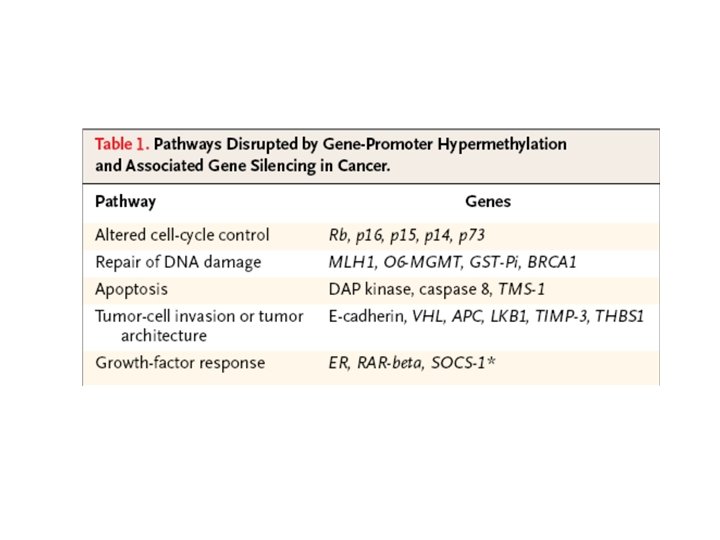
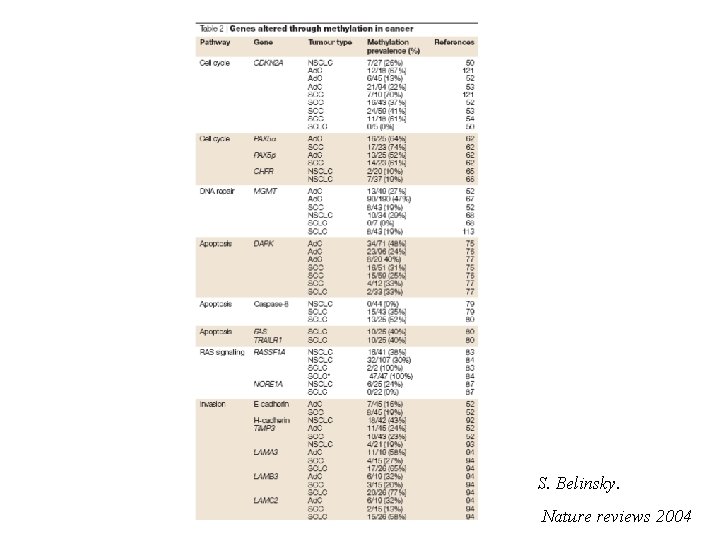
S. Belinsky. Nature reviews 2004
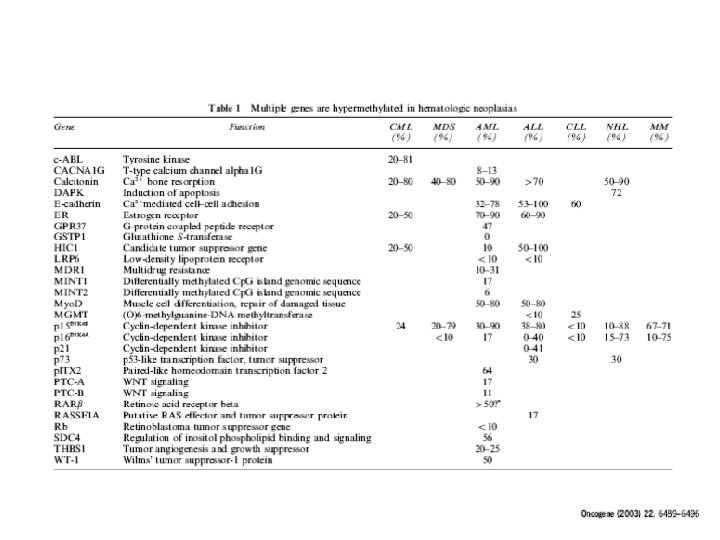

Hypermethylated genes in MDS P 15 ink 4 a (65%) DAPkinase (47%) SOCS 1 E-cadherin calcitonin

p 15 ink 4 a Negatively regulates cell cycle (G 1 -S phase) Inhibits cyclin dependent kinase 4 and 5 Gene hypermethylation correlates with shorter MDS survival and disease progression
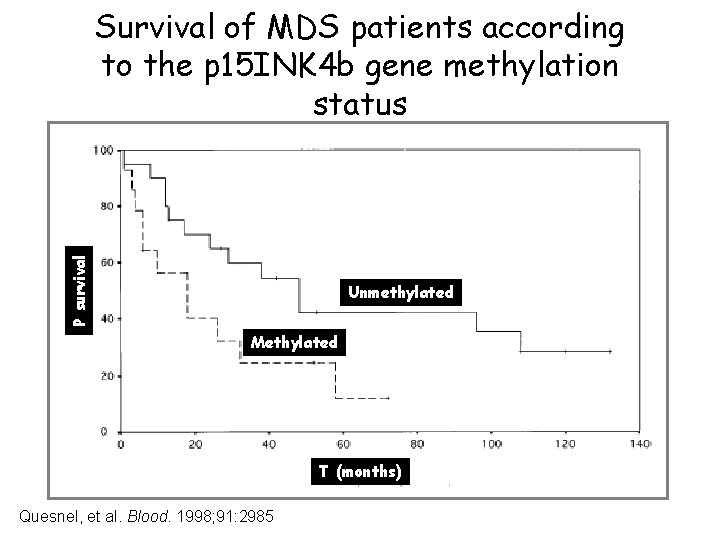
P survival Survival of MDS patients according to the p 15 INK 4 b gene methylation status Unmethylated Methylated T (months) Quesnel, et al. Blood. 1998; 91: 2985
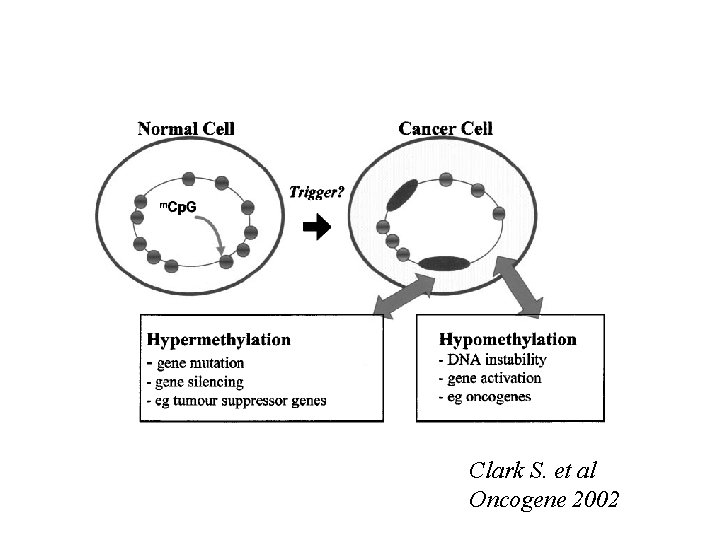
Clark S. et al Oncogene 2002
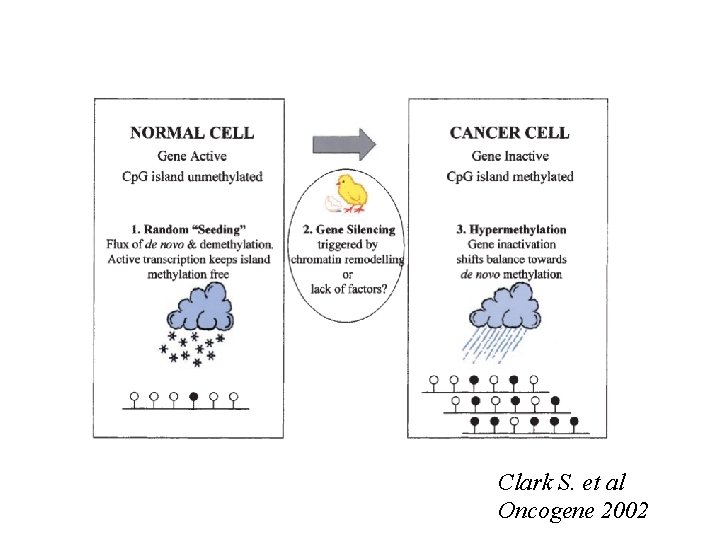
Clark S. et al Oncogene 2002
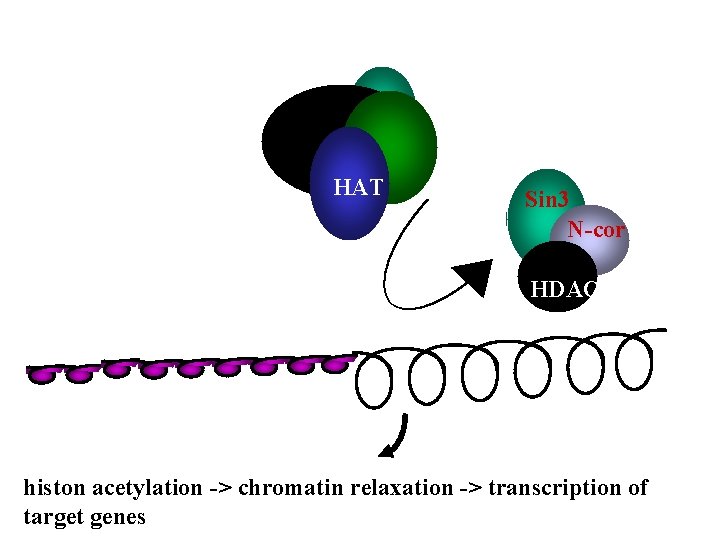
HAT Sin 3 N-cor HDAC histon acetylation -> chromatin relaxation -> transcription of target genes
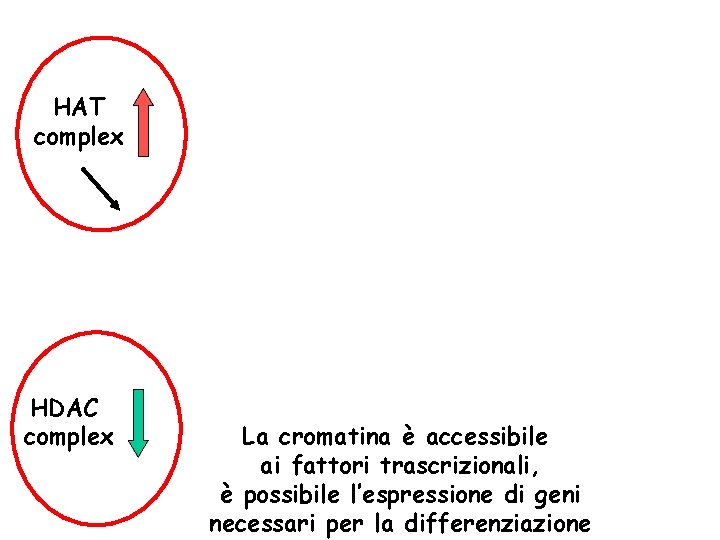
HAT complex HDAC complex La cromatina è accessibile ai fattori trascrizionali, è possibile l’espressione di geni necessari per la differenziazione
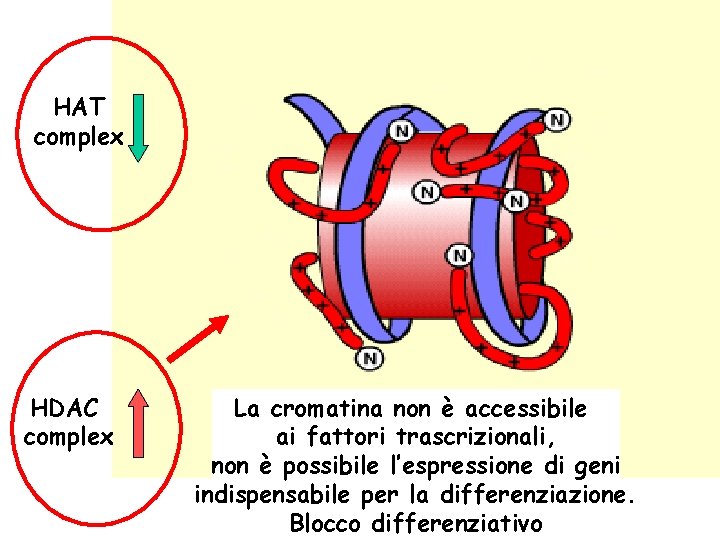
HAT complex HDAC complex La cromatina non è accessibile ai fattori trascrizionali, non è possibile l’espressione di geni indispensabile per la differenziazione. Blocco differenziativo
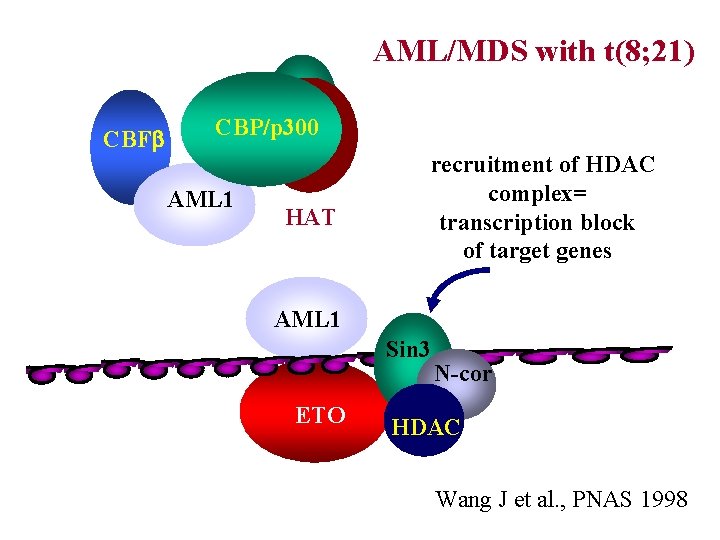
AML/MDS with t(8; 21) CBFb CBP/p 300 AML 1 HAT recruitment of HDAC complex= transcription block of target genes AML 1 Sin 3 ETO N-cor HDAC Wang J et al. , PNAS 1998
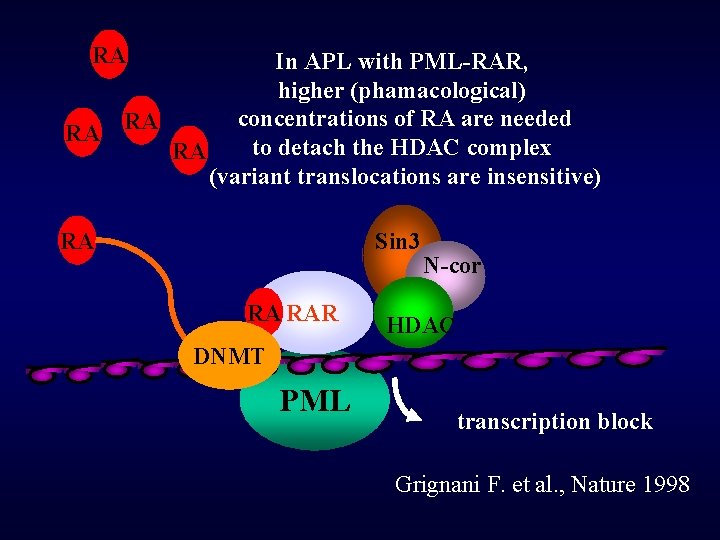
RA In APL with PML-RAR, higher (phamacological) concentrations of RA are needed RA RA to detach the HDAC complex RA (variant translocations are insensitive) RA Sin 3 RA RAR N-cor HDAC DNMT PML transcription block Grignani F. et al. , Nature 1998
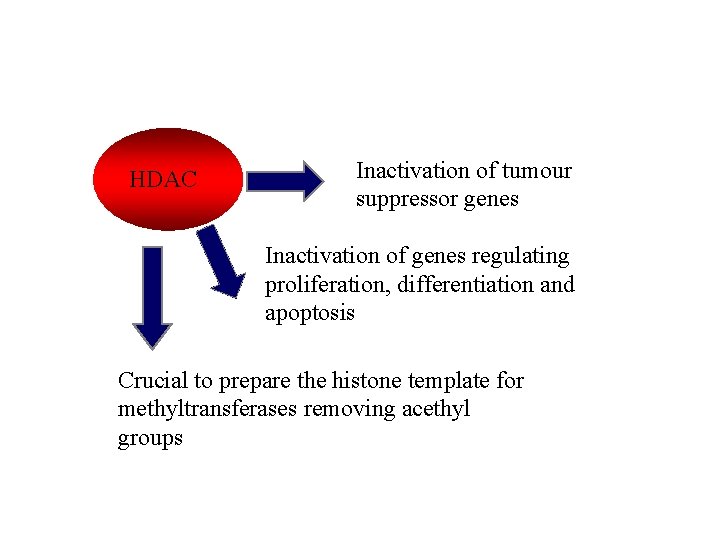
HDAC Inactivation of tumour suppressor genes Inactivation of genes regulating proliferation, differentiation and apoptosis Crucial to prepare the histone template for methyltransferases removing acethyl groups
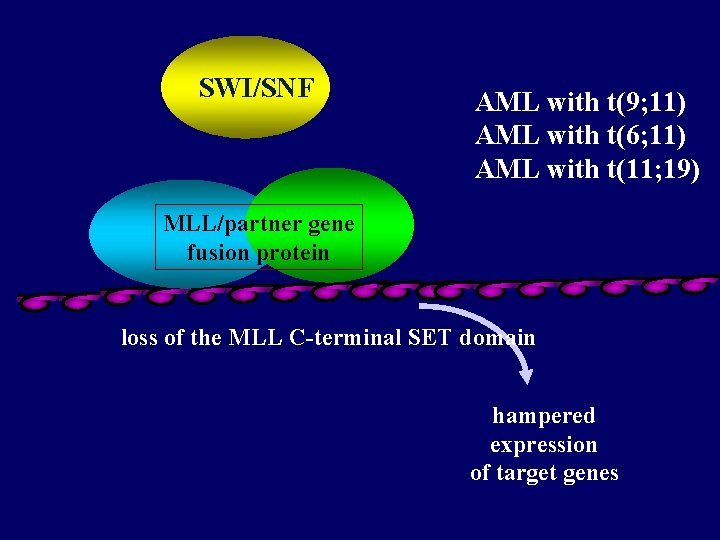
SWI/SNF AML with t(9; 11) AML with t(6; 11) AML with t(11; 19) MLL/partner gene fusion protein loss of the MLL C-terminal SET domain hampered expression of target genes
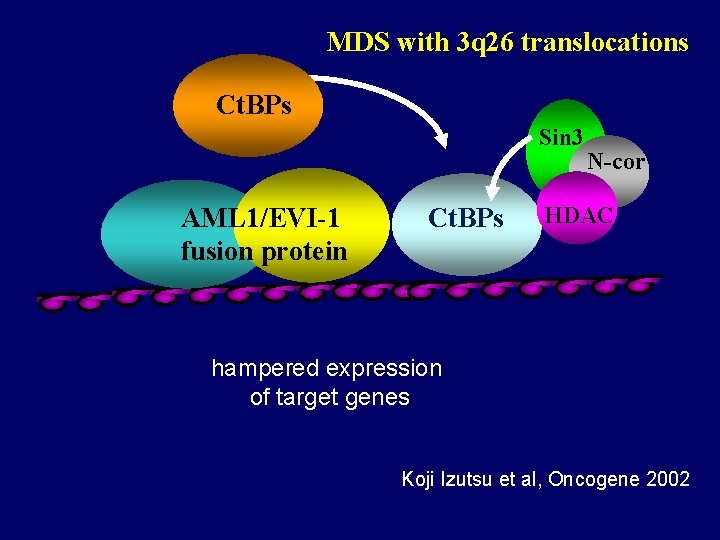
MDS with 3 q 26 translocations Ct. BPs Sin 3 AML 1/EVI-1 fusion protein Ct. BPs N-cor HDAC hampered expression of target genes Koji Izutsu et al, Oncogene 2002

CANDIDATE TARGETS FOR EPIGENETIC THERAPY DNMTs HDACs HATs

CANDIDATE DRUGS FOR EPIGENETIC THERAPY DNMT inhibitors azacitidine, decitabine, zebularine, procainamide DNMT antisense and si. RNA HDAC inhibitors


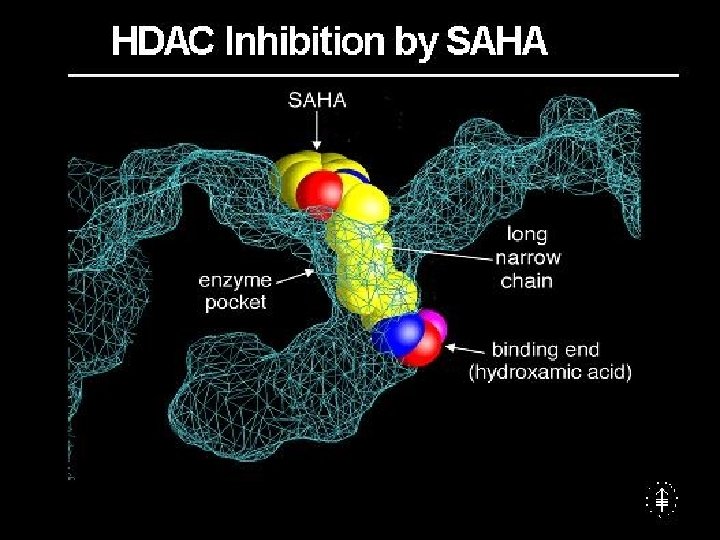

Valproic acid • Used in the treatment of epilepsy • At the dosage used : 10 mg/kg (serum levels 40 -100 mcg/ml) it acts as a potent HDAC inhibitor • Used in clinical trials for the treatment of AML and MDS patients – Refractory to conventional chemotherapy – Not candidate for conventional chemotherapy

VPA & serum level (40 -100 mcg/ml) 1. 2. 3. 4. • • ATRA 45 mg/m 2 Patients (10 AML, 7 RAEB) Patients with <1 month of therapy Patients evaluable Patients responsive Plts before therapy: median 23 x 106/l range ( 9 -39) Plts after therapy: median 157 x 106/l range (76 -271) 5. Median duration of response: months 17 7 10 6 p<0. 01 6 -8 Responses in approximately 30% of the enrolled patients (ITT basis) and in 60% of those who were really treated
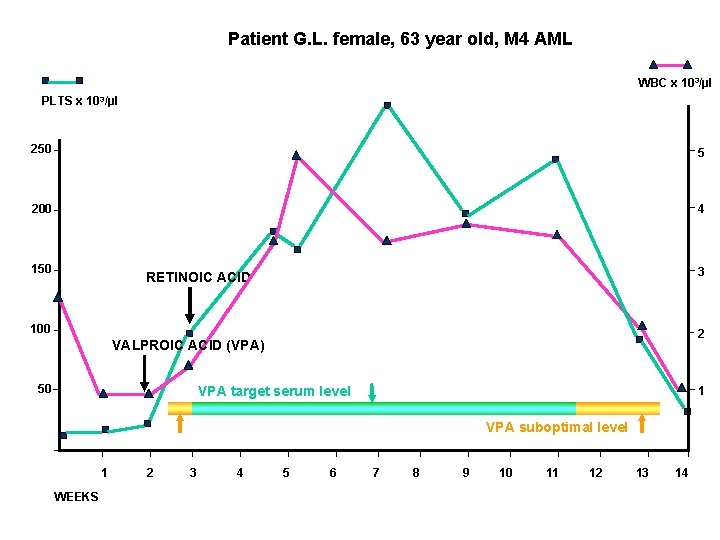
Patient G. L. female, 63 year old, M 4 AML WBC x 103/µl PLTS x 10 3/µl 250 5 200 4 150 3 RETINOIC ACID 100 2 VALPROIC ACID (VPA) 50 VPA target serum level 1 VPA suboptimal level 1 WEEKS 2 3 4 5 6 7 8 9 10 11 12 13 14

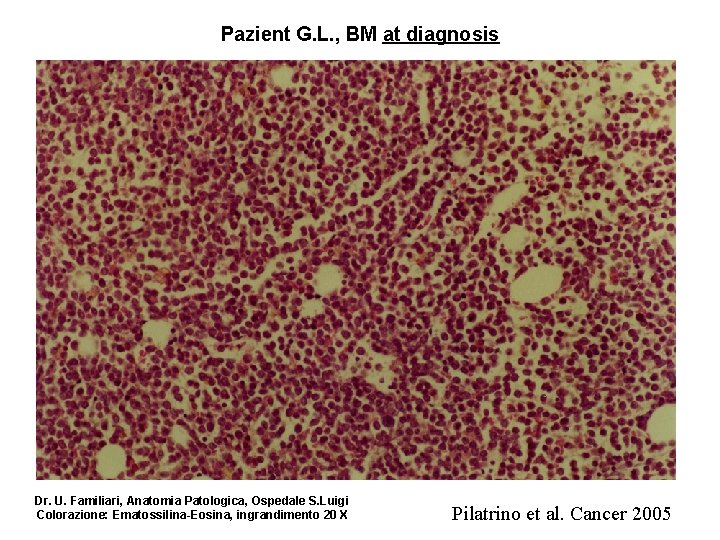
Pazient G. L. , BM at diagnosis Dr. U. Familiari, Anatomia Patologica, Ospedale S. Luigi Colorazione: Ematossilina-Eosina, ingrandimento 20 X Pilatrino et al. Cancer 2005
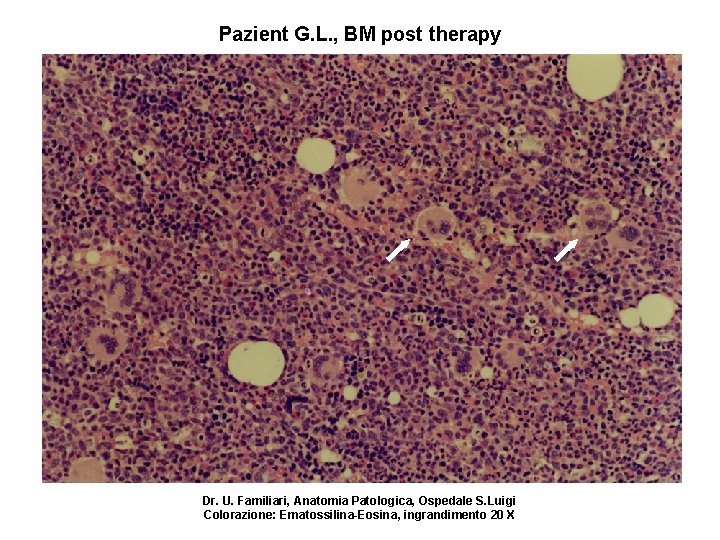
Pazient G. L. , BM post therapy Dr. U. Familiari, Anatomia Patologica, Ospedale S. Luigi Colorazione: Ematossilina-Eosina, ingrandimento 20 X
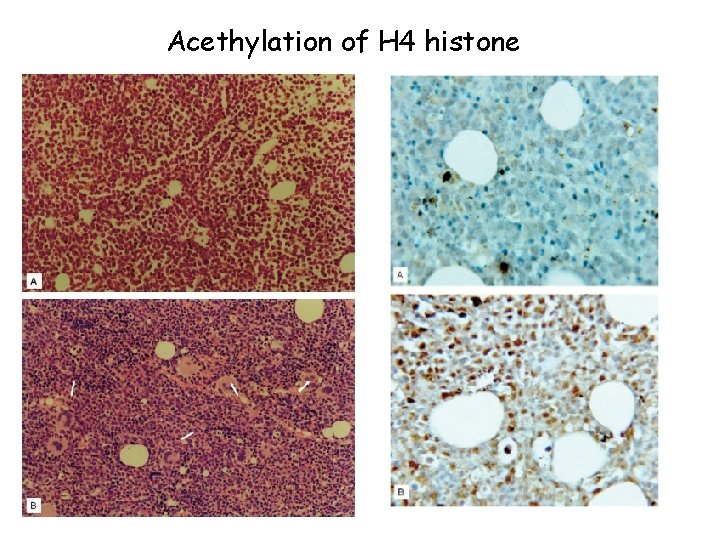
Acethylation of H 4 histone

New therapeutical strategies • Association of HDAC inhibitors and demethylating agents.
- Slides: 51