HYBRID A Prospective Randomized Trial of Hybrid Coronary

HYBRID: A Prospective, Randomized Trial of Hybrid Coronary Revascularization Versus Standard Surgical Revascularization in Multivessel Disease Michal Hawranek on behalf of the HYBRID Trial Investigators

Disclosure Statement of Financial Interest Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement or affiliation with the organization(s) listed below. Affiliation/Financial Relationship • • Grant/Research Support Consulting Fees/Honoraria Major Stock Shareholder/Equity Royalty Income Ownership/Founder Intellectual Property Rights Other Financial Benefit Company • Balton

Hybrid Revascularization for Multivessel Coronary Artery Disease POL-MIDES Cardiac surgery PI: Marian Zembala Cardiology PI: Mariusz Gasior Study Investigators: Cardiac surgery: M. Zembala Jr. , T. Hrapkowicz, K. Filipiak Cardiology: M. Tajstra, M. Hawranek, M. Gierlotka, L. Poloński Prospective Randomized Pil. Ot Study Eva. Luating the Safety and Efficacy of Hybrid Revascularization in Mult. Ivessel Coronary Artery Dis. Ea. Se (POLMIDES) ((Clinical. Trials. gov number, NCT 01035567
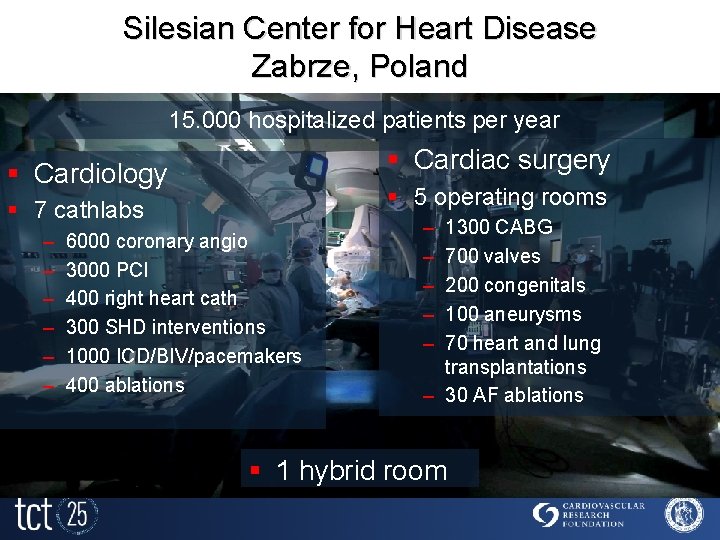
Silesian Center for Heart Disease Zabrze, Poland 15. 000 hospitalized patients per year § Cardiac surgery § Cardiology § 5 operating rooms § 7 cathlabs – – – 6000 coronary angio 3000 PCI 400 right heart cath 300 SHD interventions 1000 ICD/BIV/pacemakers 400 ablations – – – 1300 CABG 700 valves 200 congenitals 100 aneurysms 70 heart and lung transplantations – 30 AF ablations § 1 hybrid room
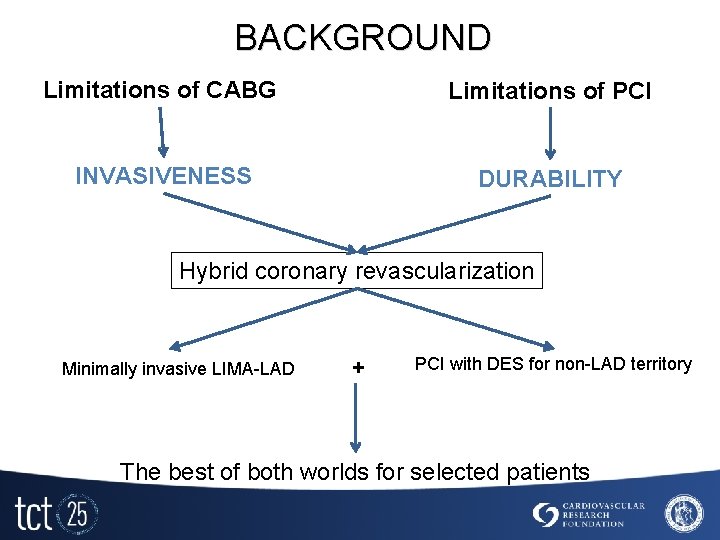
BACKGROUND Limitations of CABG Limitations of PCI INVASIVENESS DURABILITY Hybrid coronary revascularization Minimally invasive LIMA-LAD + PCI with DES for non-LAD territory The best of both worlds for selected patients
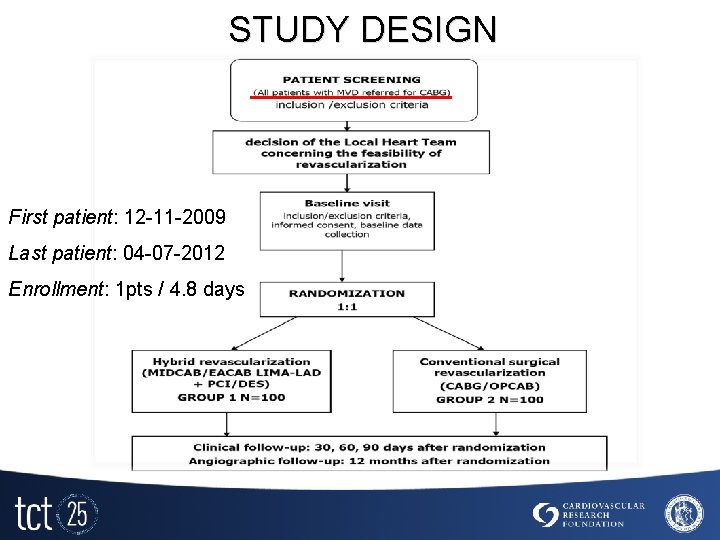
STUDY DESIGN First patient: 12 -11 -2009 Last patient: 04 -07 -2012 Enrollment: 1 pts / 4. 8 days

STUDY DESIGN STUDY ENDPIONTS Primary endpoint § § Feasibility defined by means of - a % of patients with completed hybrid procedure according to study protocol, and - a % of conversion to standard CABG. Safety defined as a occurrence of MACE such as death, MI, stroke, TVR, or major bleeding within 12 months follow up Secondary endpoint § § § Postprocedure and follow up angiographic measurements as patency of grafts and restenosis in revascularized segments Assessment of quality of life of alive study participants according to SF-36 Health Survey version 2 Cost-effectiveness defined as a cost of revascularization procedure and costs of hospitalizations in both groups.

RESULTS PRIMARY ENDPOINT - FEASIBILITY • % of patients with completed hybrid revascularization procedure 93. 9% • % of conversion to standard CABG 6. 1% Reasons: • LAD not visible through the minithoracothomy incision – 2 pts. • Hemodynamically unstable when preparing LAD for grafting. Recurrent VTs. Emergency conversion to full sternotomy – 1 pts. • LIMA damaged during endoscopic harvesting – 1 pts. • Patient did not tolerate single lung ventilation – 1 pts. • Surgeons decision ’on table’– 1 pts.
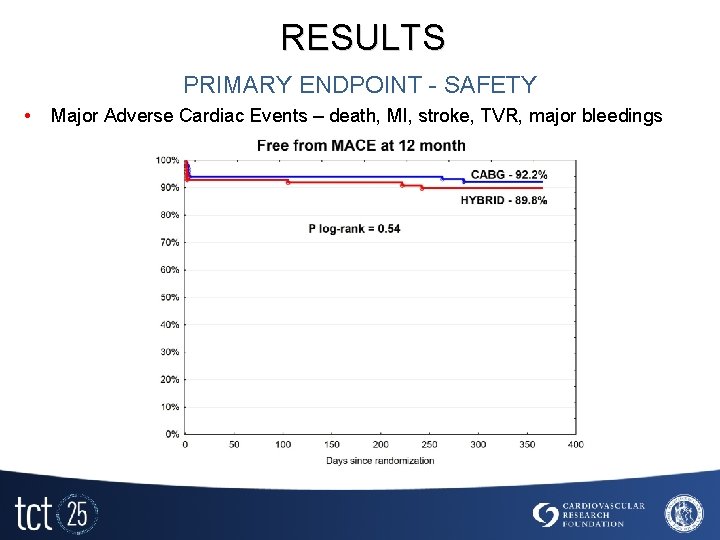
RESULTS PRIMARY ENDPOINT - SAFETY • Major Adverse Cardiac Events – death, MI, stroke, TVR, major bleedings

CONCLUSION § Hybrid coronary revascularization is feasible and safe in selected population of patients with MVD § MIDCAB as a first stage procedure in patients with MVD considered for hybrid revascularization was not associated with significantly more adverse events § This first randomized pilot study on hybrid coronary revascularization shows promising results supporting the idea of hybrid coronary revascularization in patients with MVD
- Slides: 10