HVTN 702 Confidential Do Not Distribute 1 HVTN

HVTN 702 Confidential – Do Not Distribute 1

HVTN 702 Protocol Team Confidential – Do Not Distribute 2
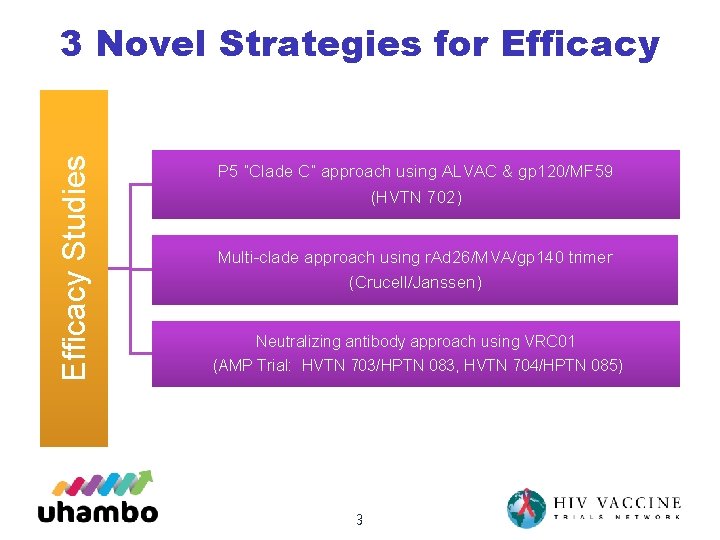
Efficacy Studies 3 Novel Strategies for Efficacy P 5 “Clade C” approach using ALVAC & gp 120/MF 59 (HVTN 702) Multi-clade approach using r. Ad 26/MVA/gp 140 trimer (Crucell/Janssen) Neutralizing antibody approach using VRC 01 (AMP Trial: HVTN 703/HPTN 083, HVTN 704/HPTN 085) 3
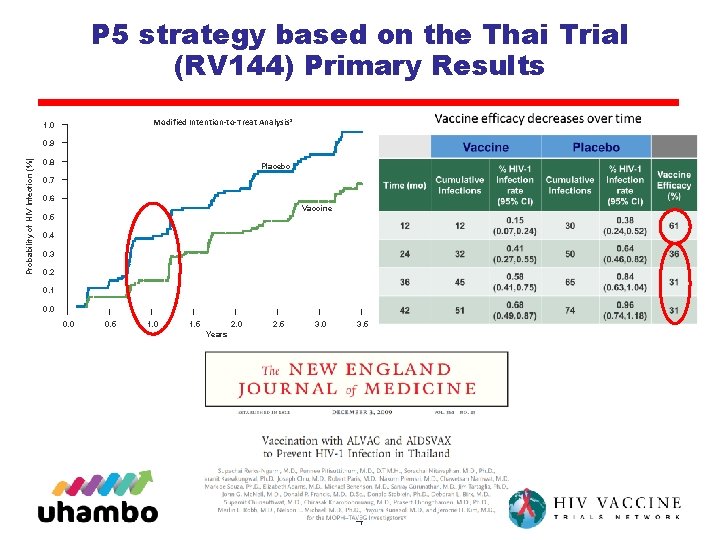
P 5 strategy based on the Thai Trial (RV 144) Primary Results Modified Intention-to-Treat Analysis* 1. 0 Probability of HIV Infection (%) 0. 9 0. 8 Placebo 0. 7 0. 6 Vaccine 0. 5 0. 4 0. 3 0. 2 0. 1 0. 0 0. 5 1. 0 1. 5 2. 0 2. 5 3. 0 3. 5 Years 4
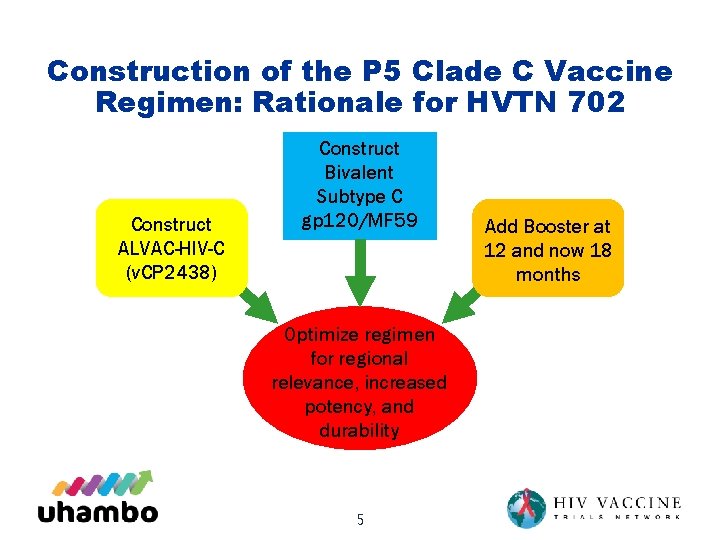
Construction of the P 5 Clade C Vaccine Regimen: Rationale for HVTN 702 Construct ALVAC-HIV-C (v. CP 2438) Construct Bivalent Subtype C gp 120/MF 59 Optimize regimen for regional relevance, increased potency, and durability 5 Add Booster at 12 and now 18 months
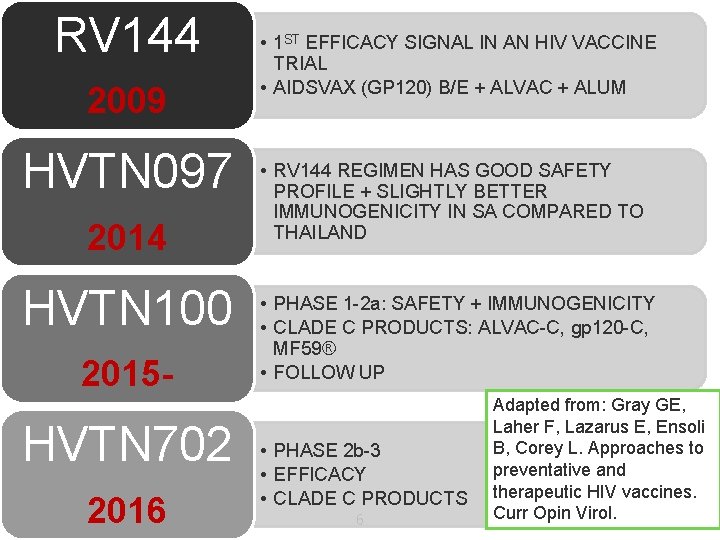
RV 144 2009 HVTN 097 2014 HVTN 100 2015 - HVTN 702 2016 • 1 ST EFFICACY SIGNAL IN AN HIV VACCINE TRIAL • AIDSVAX (GP 120) B/E + ALVAC + ALUM • RV 144 REGIMEN HAS GOOD SAFETY PROFILE + SLIGHTLY BETTER IMMUNOGENICITY IN SA COMPARED TO THAILAND • PHASE 1 -2 a: SAFETY + IMMUNOGENICITY • CLADE C PRODUCTS: ALVAC-C, gp 120 -C, MF 59® • FOLLOW UP • PHASE 2 b-3 • EFFICACY • CLADE C PRODUCTS 6 Adapted from: Gray GE, Laher F, Lazarus E, Ensoli B, Corey L. Approaches to preventative and therapeutic HIV vaccines. Curr Opin Virol.
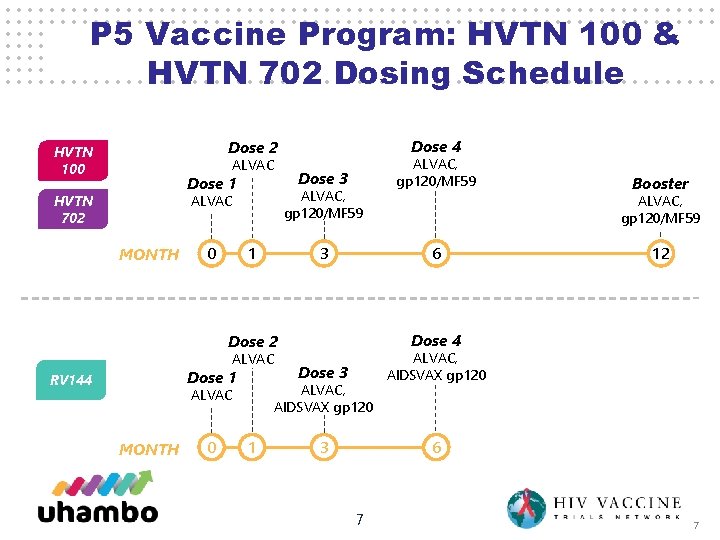
P 5 Vaccine Program: HVTN 100 & HVTN 702 Dosing Schedule Dose 4 Dose 2 HVTN 100 ALVAC Dose 1 HVTN 702 ALVAC, gp 120/MF 59 ALVAC MONTH 0 Dose 3 1 3 Dose 1 RV 144 MONTH 0 1 12 Dose 4 Dose 3 ALVAC, AIDSVAX gp 120 3 6 ALVAC, AIDSVAX gp 120 ALVAC Booster ALVAC, gp 120/MF 59 6 Dose 2 ALVAC, gp 120/MF 59 7 7

Criteria required to move into efficacy testing Criterion 1: Prevalence of antibodies to gp 120 vaccine antigens to be ≥ 75% • 100% of HVTN 100 subjects developed Env binding antibodies to the Clade C strain in the ALVAC vector, as well as the two Clade C strains in the bivalent gp 120 protein boost. Criterion 2: Magnitude of Env antibody response to be ≥ 50% of that seen in RV 144 • HVTN 100 binding antibody responses were 3. 6 to 8. 8 fold higher than in RV 144. Criterion 3: CD 4 T cell response to Env will be at least equal to that seen in RV 144 • HVTN 100 Env specific CD 4 T cell responses exceeded those in RV 144 (58% vs. 41%, respectively) and showed considerably higher polyfunctionality than in RV 144. (3 B. The polyfunctionality score was shown to be an independent correlate of VE in RV 144. ) Criterion 4: Prevalence of Ig. G antibodies to the V 1 V 2 loop to be ≥ 63% • Prevalence of Ig. G V 1 V 2 to the 1086 Clade C strain in the vaccine was 71% • The cumulative prevalence of Ig. G antibodies to the V 1 V 2 loop was 80%; this level exceeds the threshold predicted for a VE=50% based upon RV 144 Confidential – Do Not Distribute 8 8
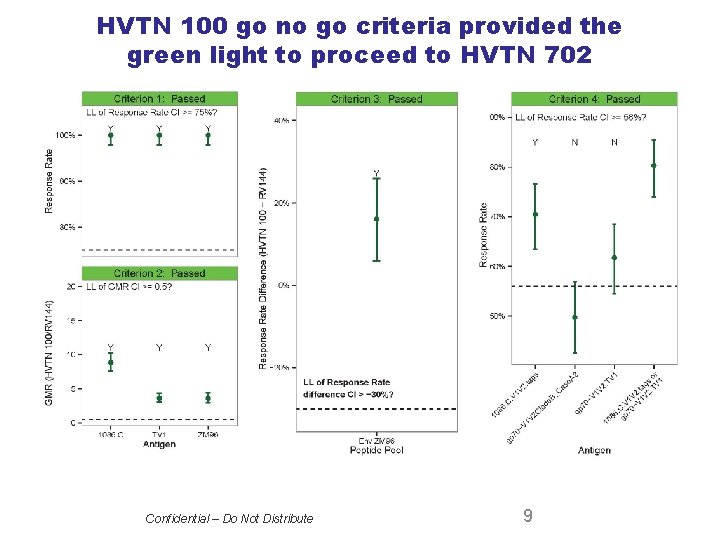
HVTN 100 go no go criteria provided the green light to proceed to HVTN 702 Confidential – Do Not Distribute 9 9

HVTN 702 A pivotal phase 2 b/3 multi-site, randomized, double-blind, placebo-controlled clinical trial to evaluate the safety and efficacy of ALVAC-HIV (v. CP 2438) and Bivalent Subtype C gp 120/MF 59 in preventing HIV-1 infection in adults in South Africa § Started in Nov 2016 in RSA § Glenda Gray, Chair § Co-Chairs: Linda Gail Bekker, Fatima Laher, Mookh Malaheha Womandla! 10

Study Schema: HVTN 702 Open for 39 weeks: 997 participants enrolled, averaging 25. 6/week N (total 5400) Primary Vaccine Regimen Booster Month 0 Month 1 Month 3 Month 6 Month 12 2700 ALVAC-HIV (v. CP 2438) ALVAC-HIV+ Bivalent Subtype C gp 120/MF 59® 2700 Placebo Placebo + Placebo Estimated Total Study duration 72 months: • Stage 1: 60 months-18 months for enrolment, 24 months of follow-up for HIV-1 uninfected individuals, 18 months follow up for HIV-1 infected individuals) • Stage 2: an additional 12 months of follow up for uninfected individuals 11
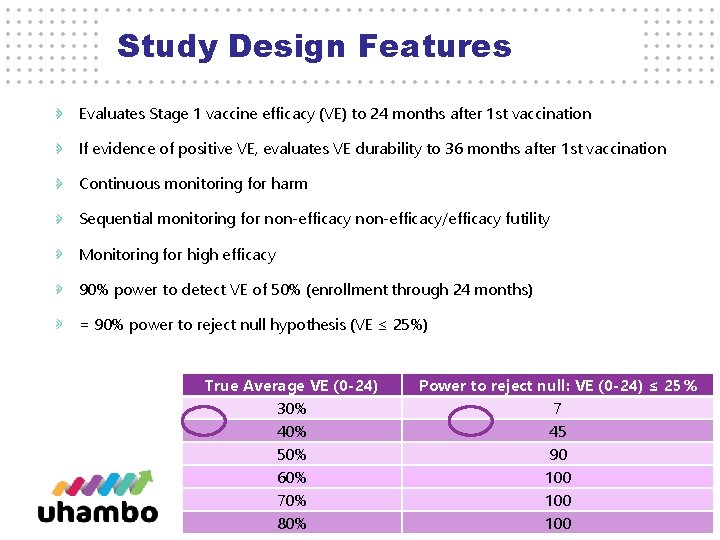
Study Design Features Evaluates Stage 1 vaccine efficacy (VE) to 24 months after 1 st vaccination If evidence of positive VE, evaluates VE durability to 36 months after 1 st vaccination Continuous monitoring for harm Sequential monitoring for non-efficacy/efficacy futility Monitoring for high efficacy 90% power to detect VE of 50% (enrollment through 24 months) = 90% power to reject null hypothesis (VE ≤ 25%) True Average VE (0 -24) 30% 40% 50% 60% 70% 12 80% Power to reject null: VE (0 -24) ≤ 25% 7 45 90 100 100 12
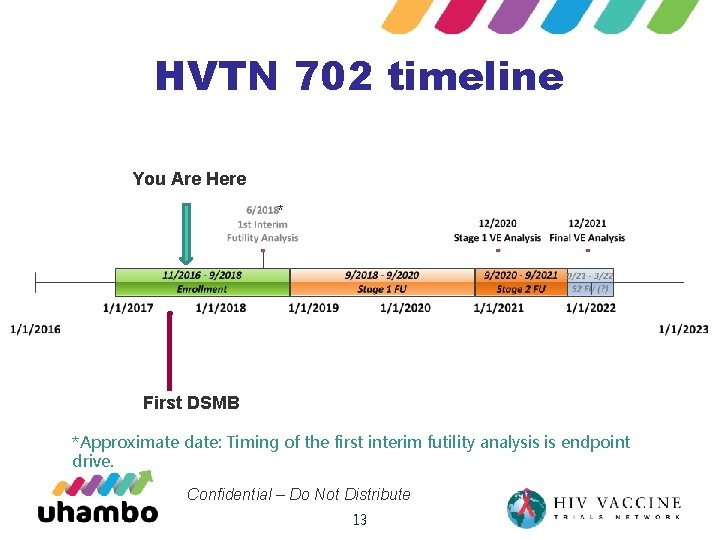
HVTN 702 timeline You Are Here ** First DSMB *Approximate date: Timing of the first interim futility analysis is endpoint drive. Confidential – Do Not Distribute 13
HVTN 100 data to support 18 month boost Confidential – Do Not Distribute

Month 18 booster HVTN 702 designed, in part, to test hypothesis that Correlates of Risk (Co. R) identified in RV 144 are, in fact, Correlates of vaccine Protection (Co. P) • Specifically, Ig. G antibodies to V 1 V 2 epitopes in Env • Ab titers drop rapidly from peak after vaccination • 12/8/2020 Confidential – Do Not Distribute 15 15
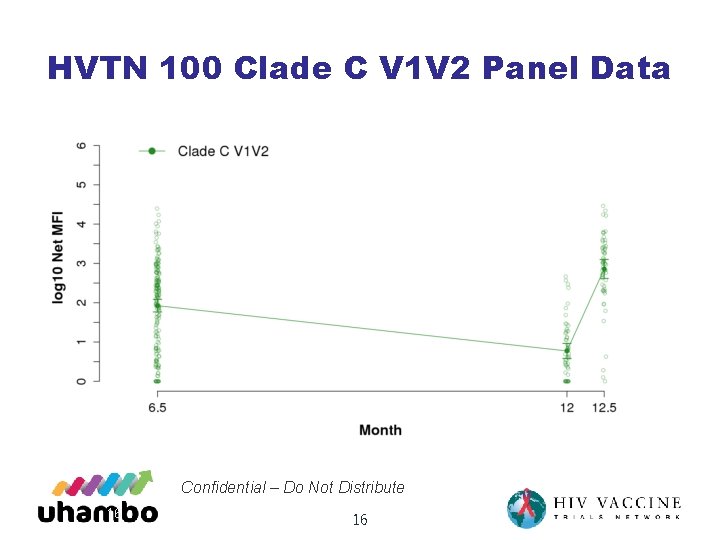
HVTN 100 Clade C V 1 V 2 Panel Data Confidential – Do Not Distribute 16 16
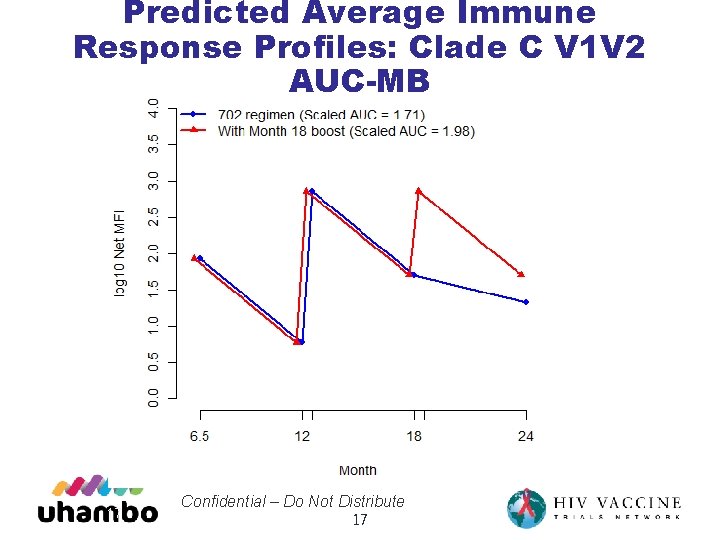
Predicted Average Immune Response Profiles: Clade C V 1 V 2 AUC-MB 17 Confidential – Do Not Distribute 17
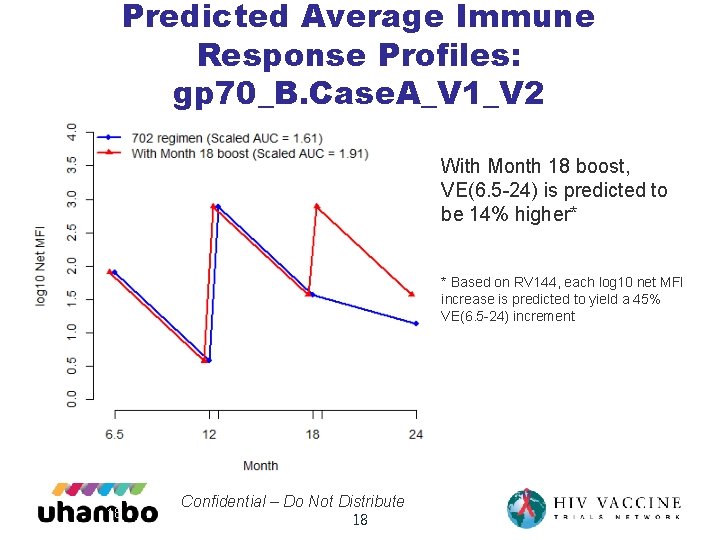
Predicted Average Immune Response Profiles: gp 70_B. Case. A_V 1_V 2 With Month 18 boost, VE(6. 5 -24) is predicted to be 14% higher* * Based on RV 144, each log 10 net MFI increase is predicted to yield a 45% VE(6. 5 -24) increment 18 Confidential – Do Not Distribute 18
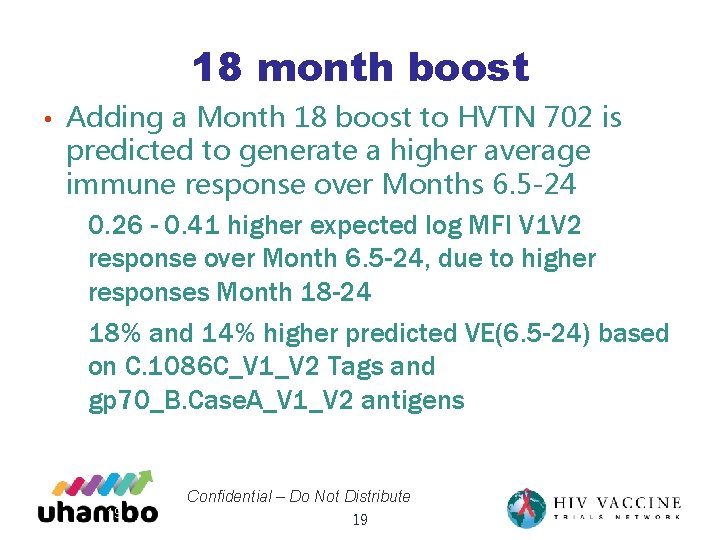
18 month boost • Adding a Month 18 boost to HVTN 702 is predicted to generate a higher average immune response over Months 6. 5 -24 • 0. 26 - 0. 41 higher expected log MFI V 1 V 2 response over Month 6. 5 -24, due to higher responses Month 18 -24 • 18% and 14% higher predicted VE(6. 5 -24) based on C. 1086 C_V 1_V 2 Tags and gp 70_B. Case. A_V 1_V 2 antigens 19 Confidential – Do Not Distribute 19

Summary Using human clinical trials with intense evaluation of the Correlates of Protection is in the end - true “rational vaccine design”. • For the first time, the basic science agenda in HIV vaccine development will be based on human clinical trials. • 20
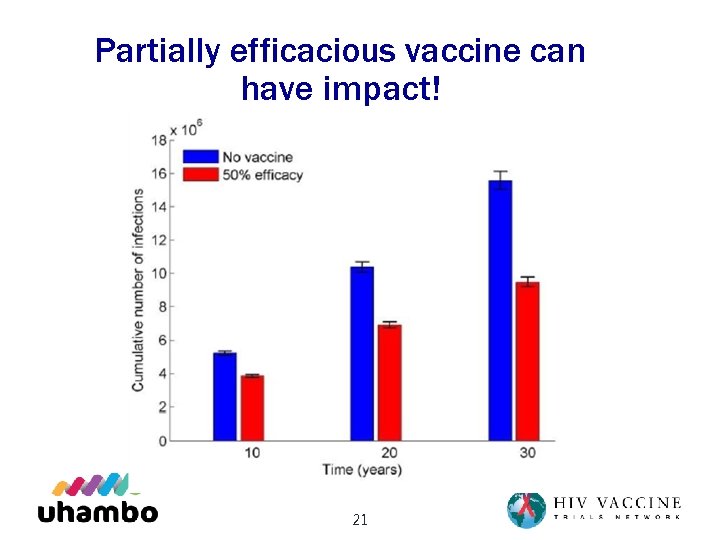
Partially efficacious vaccine can have impact! 21

HVTN 702 Protocol Team Acknowledgments Chair Glenda Gray, PHRU Cochair Fatima Laher, PHRU Linda-Gail Bekker, Univ of Cape Town Mookho Malahleha, Setshaba Research Center PTL / CMM Nicole Grunenberg, HVTN Lab Lead John Hural, HVTN Statistician Holly Janes, SCHARP Yunda Huang, SCHARP Zoe Moodie, SCHARP Medical Officer Mary Allen, DAIDS, NIAID 12/8/2020 Community Engagement Unit Nandi Luthuli, HVTN CSS RCSL Keitumetse Diphoko, HCRISA REG Megan Jones, HVTN CTM Shelly Ramirez, HVTN Developer Carlos Diaz. Granados, Sanofi Corinne Lecomte, GSK Marguerite Koutsoukos, GSK Elizabeth Briesemeister, HVTN RML Simba Takuva, HCRISA SCDM Gina Escamilla, SCHARP Social Scientist Michele Andrasik, HVTN Olivier Van Der Meeren, GSK Sanjay Phogat, Sanofi Millicent Atujuna, Univ of Cape Town SRA Lab On Ho, HVTN Lead Clinic Coordinator Brittany Sanchez, SCHARP Doug Grove, SCHARP CAB Rep Katlego Mapetla, Setshaba PDM Bulelwa Zono, Cape Town-Emavundleni Kagiso Mothwa, Setshaba Carter Bentley, HVTN Pharmacist Bijal Patel, DAIDS, NIAID Irene Rwakazina, DAIDS, NIAID Project Officer Michael Pensiero, DAIDS, NIAID Vijay Mehra, DAIDS, NIAID Themboko Maduna, PHRU-Soweto CER Rep Phumla Madi, PHRU - Kliptown Technical Editor Erik Schwab, HVTN 22 22

HVTN 702 - Acknowledgements Stats Brittany Sanchez, SCHARP Doug Grove, SCHARP Holly Janes, SCHARP Tahereh Nourbakhsh, SCHARP Yunda Huang, SCHARP Zoe Moodie, SCHARP Funders & Other Collaborators NIAID/DAIDS BMGF South African Medical Research Council (SAMRC) GSK (formerly Novartis) Sanofi Pasteur Data Management Gina Escamilla, SCHARP Hannah Leingang, SCHARP 12/8/2020 23 23

Thank you to all the study participants, site investigators, clinic coordinators, CER teams, and site pharmacists. HVTN 702 Sites: Cape Town - Khayelitsha Durban - e. Thekwini Durban - Isipingo Durban - Verulam Klerksdorp Ladysmith Medunsa 12/8/2020 24 24 Mthatha Rustenburg Soshanguve Soweto - Bara Soweto - Kliptown Tembisa
Acknowledgements NIH, NIAID, DAIDS • • Anthony Fauci Carl Dieffenbach Mary Marovich Dale Hu Mary Allen Mary Ann Luzar Bijal Patel Michael Pensiero • Participants • Clinical Research Site staff • CAB members • Community Stakeholders HVTN CORE • • • • Larry Corey Jim Kublin Nicole Grunenberg Carter Bentley Shelly Ramirez Niles Eaton Megan Jones Simba Takuva Nandi Luthuli Emilder Chihota Michelle Nebergall Kate Hopkins Liz Briesemeister Rachel Mc. Clennan Jim Maynard Bert Le. Roux Co-Chairs • • • Linda-Gail Bekkker Mookho Malahleha Fatima Laher HVTN LAB • • • Julie Mc. Elrath John Hural On Ho Mike Stirewalt Andries Engelbrecht MRC (SA) SCHARP • Holly Janes • Zoe Moodie • Yunda Huang • Doug Grove • Shannon Grant • April Randhawa • Jessica Andriesen • Gina Escamilla • Peter Gilbert 25 25 BMGF • • • Emilio Emini Nina Russell Peggy Johnston Pervin Anklesaria Silvija Staprans GSK • • Marguerite Koutsoukos Olivier Van-der-Meeren SANOFI PASTEUR • • Sanjay Gurunathan Jim Tartaglia Sanjay Phogat Carlos Diaz. Granados Protocol CAB • Kagiso Mothwa • Thembeka Maduna Clinic coordinator • Katlego Mapetla CER • Phumla Madi Social scientists • Michele Peake-Andrasik • Millicent Atujuna
- Slides: 25