Humboldt Universitt zu Berlin Sommersemester 2010 Systembiologie 3

Humboldt. Universität zu Berlin Sommersemester 2010 Systembiologie 3 - Stoichiometry Edda Klipp Humboldt-Universität zu Berlin Institut für Biologie Theoretische Biophysik 1
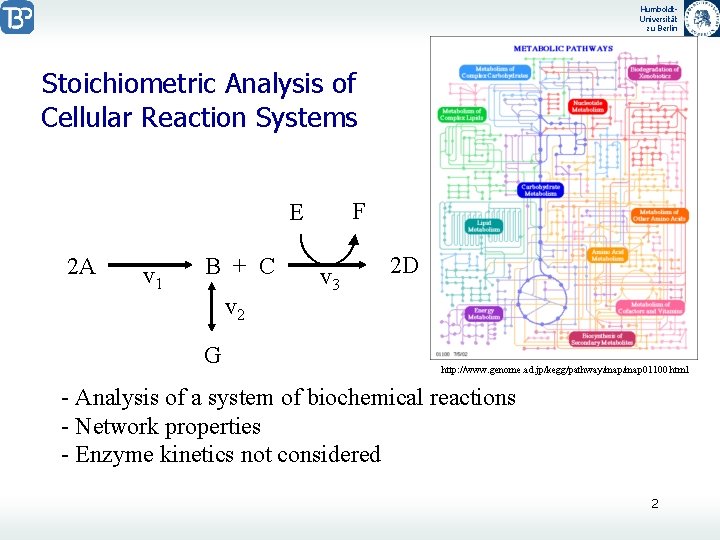
Humboldt. Universität zu Berlin Stoichiometric Analysis of Cellular Reaction Systems F E 2 A v 1 B + C v 2 G v 3 2 D http: //www. genome. ad. jp/kegg/pathway/map 01100. html - Analysis of a system of biochemical reactions - Network properties - Enzyme kinetics not considered 2

Humboldt. Universität zu Berlin Stoichiometric Coefficients Stoichiometric coefficients denote the proportions, with which the molecules of substrates and products enter the biochemical reactions. Example Catalase Stoichiometric coefficients for Hydrogenperoxid, water, oxygen Stoichiometric coefficients can be chosen such that they agree with molecularity, but not necessarily. Their signs depend on the chosen reaction direction. Since reactions are usually reversible, one cannot distinguish between „substrate“ and „product“. -2 2 1 -1 1 1/2 2 -2 -1 v -v 3

Humboldt. Universität zu Berlin Time Course of Concentrations Usually described by ordinary differential equations (ODE) Example catalase for this choice of stoichiometric coefficienten: -2 2 1 4

Humboldt. Universität zu Berlin Time Course of Concentrations Usually described by ordinary differential equations (ODE) Several reactions at the same time all rate equations must be considered at the same time. 1 S 1 4 2 S 2 3 æ 1 -1 0 -1ö S 1 ç ÷ = N ç 0 1 1 0 ÷ S 2 ç 0 0 0 1 ÷ S è ø 3 S 3 1 2 3 Row: Substance 4 Column: reaction External metabolites are not included in N. One can summarize the stoichiometric coefficients in matrix N. The rows refer to the substances, the columns refer to the reactions. 5

Balance equations/Systems equations Humboldt. Universität zu Berlin In general: We consider the substances Si and their stoichiometric coefficients nij in the respective reaction j. r – number of reaction Si – metabolite concentration vj – reaction rate nij – stoichiometric coefficient If the biochemical reactions are the only reason for the change of concentration of metabolites, i. e. if there is no mass flow by convection, diffusion or similar Then one can express the temporal behavior of concentrations by the balance equations. 6
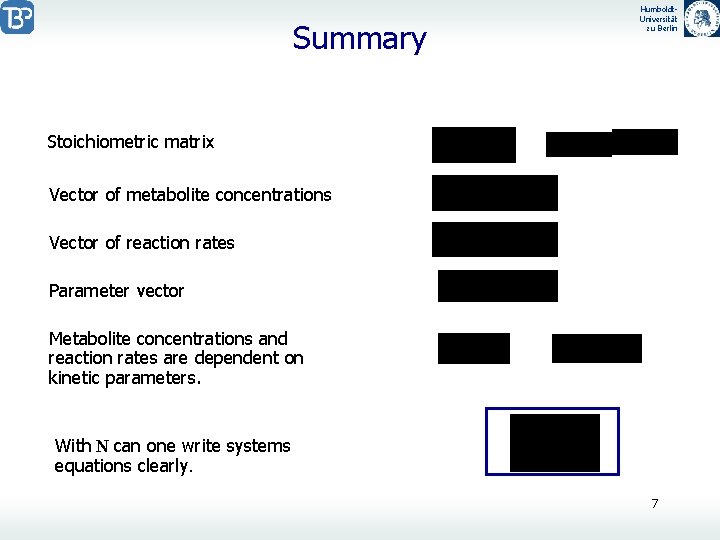
Summary Humboldt. Universität zu Berlin Stoichiometric matrix Vector of metabolite concentrations Vector of reaction rates Parameter vector Metabolite concentrations and reaction rates are dependent on kinetic parameters. With N can one write systems equations clearly. 7

The Steady State Humboldt. Universität zu Berlin Reaction systems are frequently considered in steady state, Where metabolite concentrations change do not change with time. This describes an implicite dependency of concentrations and fluxes on the parameters. b. z. w. The flux in steady state is 8
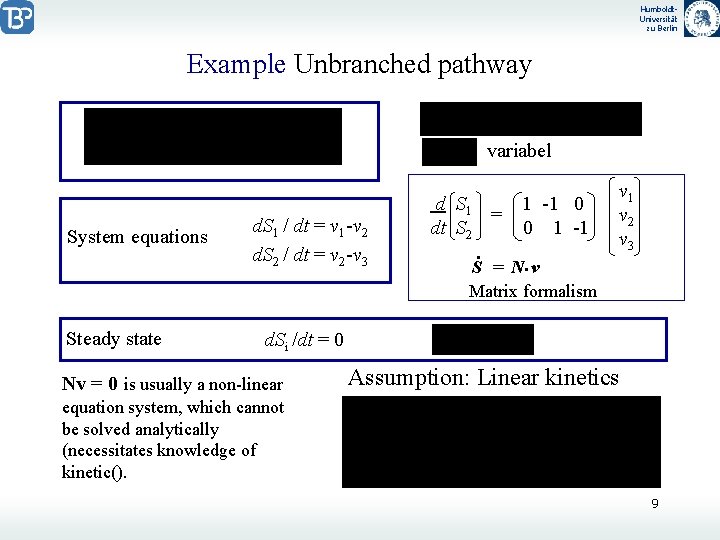
Humboldt. Universität zu Berlin Example Unbranched pathway variabel System equations Steady state d. S 1 / dt = v 1 -v 2 d. S 2 / dt = v 2 -v 3 d S 1 1 -1 0 = dt S 2 0 1 -1. S = N. v Matrix formalism v 1 v 2 v 3 d. Si /dt = 0 Nv = 0 is usually a non-linear Assumption: Linear kinetics equation system, which cannot be solved analytically (necessitates knowledge of kinetic(). 9

Humboldt. Universität zu Berlin The Stoichiometric Matrix N - Characterizes the network of all reactions in the system - Contains information about possible pathways 10

The Kernel Matrix K Humboldt. Universität zu Berlin In steady state holds Non-trivial solutions exist only if the columns of N are linearly dependent. Mathematically, the linear dependencies can be expressed by a matrix K with the columns k which each solve K – null space (Kernel) of N The number of basis vectors of the kernel of N is 11

Calculation of the Kernel Matrix Humboldt. Universität zu Berlin The Kernel matrix K can be calculated with the Gaussian Elimination Algorith for the solution of homogeneous linear equation systems. Example Alternative: calculate with computer programmes Such as „Null. Space[matrix]“ in Mathematica. 12

Representation of Kernel Matrix Humboldt. Universität zu Berlin The Kernel matrix K is not uniquely determined. Every linear combination of columns is also a Possible solution. Matrix multiplication with a regular Matrix Q „from right“ gives another Kernel matrix. For some applications one needs a simple ("kanonical") representation of the Kernel matrix. A possible and appropriate choice is K contains many zeros. I – Identity matrix 13

Informations from Kernel Matrix K Humboldt. Universität zu Berlin -Admissible fluxes in steady state -Equilibrium reactions -Unbranched reaction sequences -Elementary modes 14
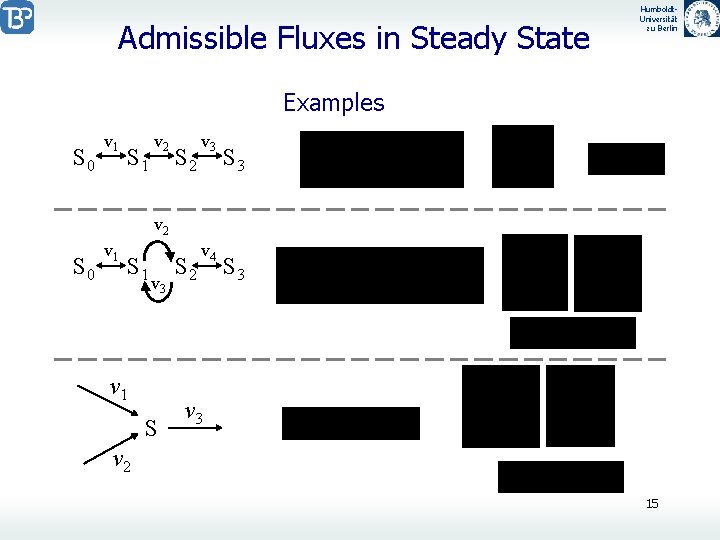
Admissible Fluxes in Steady State Humboldt. Universität zu Berlin Examples S 0 v 1 S 1 v 2 S 2 v 3 S 3 v 2 S 0 v 1 S 1 v 3 v 1 S S 2 v 4 S 3 v 2 15

Humboldt. Universität zu Berlin Admissible Fluxes in Steady State With the vectors ki (k 1, k 2, …) is also every linear combination A possible columns of K. for example: instead and also In steady state holds All admissible fluxes in steady state can be written as linear combinations of vectors ki : v 1 für v 3 S v 2 The coefficients i have the respective units, eg. or . 16

Equilibrium Reactions Humboldt. Universität zu Berlin Case: all elements of a row in K are 0 Then: the respective reaction is in every steady state in equilibrium. Example 17

Unbranched Reaction Steps S 0 v 1 S 1 v 2 v 4 S 2 v 3 Humboldt. Universität zu Berlin S 3 S 4 The basis vectors of nullspace have the same entries for unbranched reaction sequences. Unbranched reaction sequences can be lumped for further analysis. 18

Kernel Matrix –Dead Ends S 0 v 1 S 1 v 2 v 4 S 2 v 3 S 4 Humboldt. Universität zu Berlin S 1, S 2, S 3 intern, S 0, S 4 extern S 3 Necessary and sufficient condition for a „Dead end“: One metabolite has only one entry in the stoichiometric matrix (is only once Substrate or product). Flux in steady state through this reaction must vanish in steady state (J 4 = 0). Model reduction: one can neglect those reactants for steady state analyses. 19

Kernel Matrix – Irreversibility S 0 v 1 S 1 v 2 v 4 S 2 v 3 S 3 Humboldt. Universität zu Berlin S 1, S 2 internal, S 0 , S 3, S 4 external S 4 Other choice of basis vectors Mathematically possible, biologically not feasible The basis vectors of a null space are not unique. The direction of fluxes (signs) do not necessarily agree with the direction of irreversible reactions. (Irreversibility limits the space of possible steady state fluxes. ) 20

Conservation Relations: Matrix G Humboldt. Universität zu Berlin If compounds or groups are not added to or deprived of a Reaction system, then must their total amount remain constant. Examples Michaelis-Menten kinetics Isolated reaction: Pyruvatkinase, Na/K-ATPase 21

Conservation Relations - Calculation Humboldt. Universität zu Berlin If there exist linear dependencies between the rows of the stoichiometric matrix, then one can find a matrix G such as N – stoichiometric matrix Due to holds The integration of this equation yields the conservation relations. 22

Conservation Relations – Properties of G Humboldt. Universität zu Berlin The number of independent row vectors g (= number of Independent conservation relations) is given by (n = number of rows of the stoichiometric matrix = number of metabolites) GT is the Kernel matrix of NT, and can be found in the same way as K. (Gaussian elimination algorithm) The matrix G is not unique, with P regular quadratic matrix is again conservation matrix. Separated conservation conditions: 23
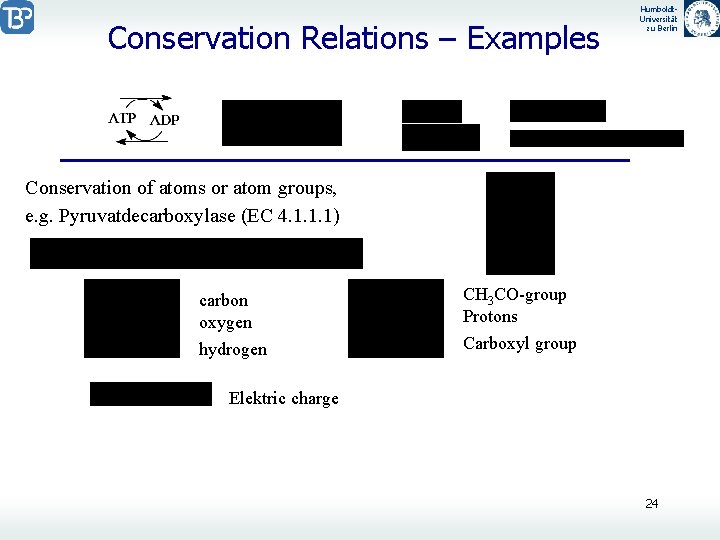
Conservation Relations – Examples Humboldt. Universität zu Berlin Conservation of atoms or atom groups, e. g. Pyruvatdecarboxylase (EC 4. 1. 1. 1) carbon oxygen CH 3 CO-group Protons hydrogen Carboxyl group Elektric charge 24

Conservation relations – Simplification of the ODE system Humboldt. Universität zu Berlin If conservation relations hold for a reaction system, then the ODE system can be reduced, since some equations are linearly dependent. Rearrange N, L – Linkmatrix (independent upper rows, dependent lower rows) Rearrange S respectively (indep upper rows, dep lower rows) For dependent concentrations hold Reduced ODE system 25
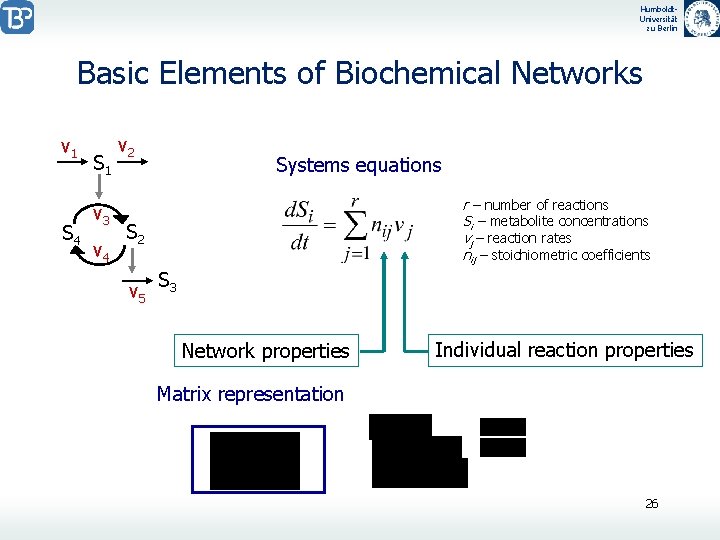
Humboldt. Universität zu Berlin Basic Elements of Biochemical Networks v 1 S 4 S 1 v 3 v 4 v 2 Systems equations r – number of reactions Si – metabolite concentrations vj – reaction rates nij – stoichiometric coefficients S 2 v 5 S 3 Network properties Individual reaction properties Matrix representation 26

Humboldt. Universität zu Berlin Nachtrag vom 10. Mai 2010 27

Humboldt. Universität zu Berlin Non-negative Flux Vectors In many biologically relevant situations have fluxes fixed signs. We can define their direction such that Example: opposite uni-directional rates instead of net rates, - Description of tracer kinetics or dynamics of NMR labels - different isoenzymes for different directions of reactions - for (quasi) irreversible reactions Sometimes is the value of individual rates fixed. Both conditions restrict the freedom for the choice of Basis vectors for K. 28

Humboldt. Universität zu Berlin Elementary Flux Modes Situation: some fluxes have fixed signes, others can operate in both directions. Which (simple) pathes connect external substrats? P 1 v 1 v 2 S v 3 P 2 P 3 29

Elementary Flux Modus Humboldt. Universität zu Berlin -An elementary flux mode comprises all reaction steps, Leading from a substrate S to a product P. -Each of these steps in necessary to maintain a steady state. -The directions of fluxes in elementary modes fulfill the demands for irreversibility 30
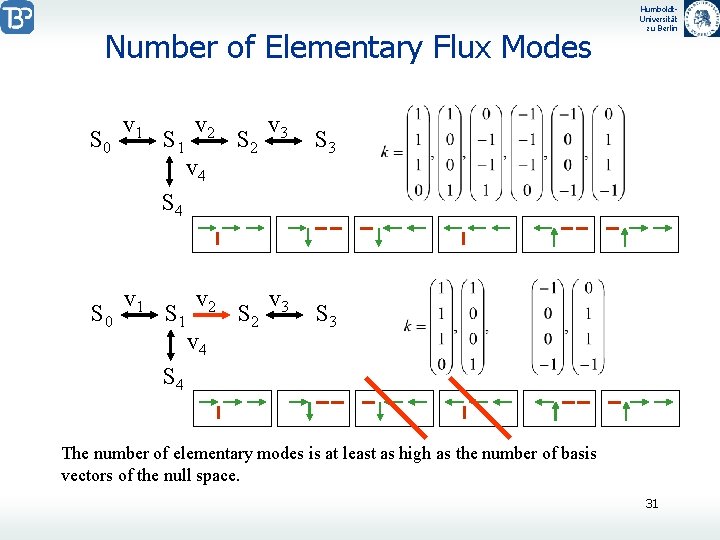
Number of Elementary Flux Modes S 0 v 1 S 1 v 2 v 4 S 2 v 3 Humboldt. Universität zu Berlin S 3 S 4 S 0 v 1 S 1 v 2 v 4 S 2 v 3 S 4 The number of elementary modes is at least as high as the number of basis vectors of the null space. 31
- Slides: 31