HUMAN RENAL SYSTEM PHYSIOLOGY Lecture 11 12 BY
HUMAN RENAL SYSTEM PHYSIOLOGY Lecture 11, 12 BY: LECT. DR. ZAINAB AL-AMILY

Objectives 1. Describe metabolic acidosis and its compensatory mechanism 2. Describe metabolic alkalosis and its compensatory mechanism 3. Describe respiratory acidosis and its compensatory mechanism 4. Describe respiratory alkalosis and its compensatory mechanism

• Acid-base disorders • METABOLIC ACIDOSIS • is caused by a decreased HCO 3 concentration in the blood. • can result from: 1. 2. 3. 4. increased production of fixed acids such as ketoacids or lactic acid; ingestion of fixed acids such as salicylic acid; Methanol/formaldehyde poisoning; Ethylene glycol poisoning the inability of the kidneys to excrete the fixed acids produced from normal metabolism; or loss of HCO 3 via the kidneys or the gastrointestinal tract (Diarrhea)

• The arterial blood profile seen in metabolic acidosis is: • p. H ↓ • [HCO 3 ] ↓ • Pco 2 ↓ • Although metabolic acidosis can be caused by a frank loss of HCO 3 , as in diarrhea most often it is caused by an excess of fixed acid in the body.

The following sequence of events occurs in generation of metabolic acidosis to produce this blood profile. 1. Gain of fixed H+. • Excess fixed H is accumulated in the body either through increased production or ingestion of fixed acid or from decreased excretion of fixed acid. 2. Buffering. The excess fixed H is buffered in both ECF and ICF. • In ECF, the H is buffered primarily by HCO 3, which produces a decrease in HCO 3 concentration. • The decrease in HCO 3 concentration causes a decrease in p. H, • In ICF, the excess fixed H is buffered by organic phosphates and proteins.

• To utilize these intracellular buffers, H first must enter the cells. • H can enter the cells with an organic anion such as ketoanion, lactate, or formate, or it can enter the cells in exchange for K. • When the H is exchanged for K, hyperkalemia occurs 3. Respiratory compensation. • Decreased arterial p. H stimulates peripheral chemoreceptors in the carotid bodies, which respond by causing hyperventilation. • In turn, hyperventilation produces a decreased PCO 2, which is the respiratory compensation for metabolic acidosis. [HCO 3 ] ( ↓ Primary disturbance) Pco 2 ( ↓ Respiratory compensation)

• The primary disturbance is decreased HCO 3 concentration, which, by itself, would lead to a profound decrease in p. H. • The respiratory compensation, ( hyperventilation), decreases the PCO 2, which tends to normalize the ratio of HCO 3/ PCO 2 and to normalize the p. H. 4. Renal correction. • Buffering and respiratory compensation occur quickly. • The ultimate correction of metabolic acidosis occurs in the kidneys and takes several days. • The excess fixed H will be excreted as titratable acid and NH 4 • Simultaneously, new HCO 3 will be synthesized and reabsorbed by the kidneys to replace the HCO 3 that was consumed earlier in buffering. • In this way, the blood HCO 3 concentration will be returned to normal.


• METABOLIC ALKALOSIS • is caused by an increased HCO 3 -concentration in the blood. • is the result of 1. loss of fixed H from the gastrointestinal tract; 2. loss of fixed H from the kidney (e. g. , hyperaldosteronism); 3. administration of solutions containing HCO 3 4. or ECF volume contraction (e. g. , administration of diuretics)

• The arterial blood profile seen in metabolic alkalosis is: • p. H ↑ • [HCO 3 ] ↑ • Pco 2 ↑ • Although metabolic alkalosis can be caused by administration of HCO 3, most often it is caused by loss of fixed acid from the body

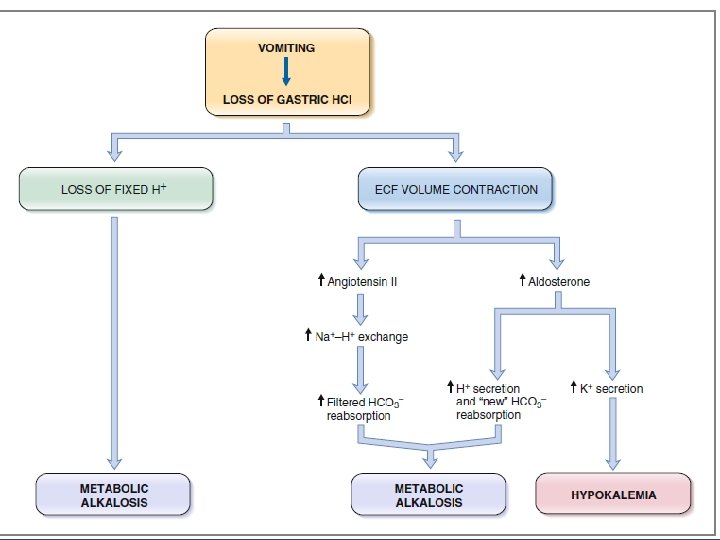
• The following sequence of events occurs in the generation of metabolic alkalosis to produce this blood profile. • 1. Loss of fixed acid. The classic example of metabolic alkalosis is vomiting, in which HCl is lost from the stomach. • The gastric parietal cells produce H and HCO 3 from CO 2 and H 2 O. • The H is secreted with Cl into the lumen of the stomach to aid in digestion, and the HCO 3 enters the blood. • In normal persons, the secreted H moves from the stomach to the small intestine, where a low p. H triggers the secretion of HCO 3 by the pancreas. • Thus, normally, the HCO 3 added to blood by the parietal cells is later removed from blood in the pancreatic secretions.

• when vomiting occurs, H is lost from the stomach and never reaches the small intestine. • HCO 3 secretion from the pancreas, therefore, is not stimulated, and the HCO 3 - remains in the blood, resulting in an increase in HCO 3 - concentration. • The increase in HCO 3 concentration causes an increase in p. H,

• Which of the following is a cause of metabolic alkalosis? (A) vomiting (B) Chronic renal failure (C) Ethylene glycol ingestion (D) diarrhea (E) Hypoaldosteronism

• 2. Buffering. occurs in both ECF and ICF. • To utilize ICF buffers, H leaves the cells in exchange for K, and hypokalemia occurs. 3. Respiratory compensation. • Increased arterial p. H inhibits the peripheral chemoreceptors, which respond by causing hypoventilation. • In turn, hypoventilation produces an increased PCO 2, which is the respiratory compensation for metabolic alkalosis. [HCO 3 ] ( ↑ Primary disturbance) Pco 2 ( ↑ Respiratory compensation

• The primary disturbance in metabolic alkalosis is an increased HCO 3 concentration that, by itself, would lead to a profound increase in p. H • The respiratory compensation, hypoventilation, increases the PCO 2, which tends to normalize the ratio of HCO 3 /CO 2 and to normalize the p. H. 4. Renal correction. The correction of metabolic alkalosis should be the most straight forward of all the acid-base disorders.

• Since the primary disturbance is increased HCO 3 concentration, restoration of acid-base balance will take place when the excess HCO 3 is excreted by the kidneys. • This can be accomplished because the renal tubule has a finite reabsorptive capacity for filtered HCO 3 • When the filtered load of HCO 3 exceeds the reabsorptive capacity, HCO 3 is excreted in the urine, eventually reducing the HCO 3 concentration to normal.
. A patient arrives at the emergency room with low arterial pressure, reduced tissue turgor, and the following arterial blood values: p. H = 7. 69 [HCO 3 –] = 57 m. Eq/L PCO 2 = 48 mm Hg Which of the following responses would also be expected to occur in this patient? (A) Hyperventilation (B) increased K+ secretion by the distal tubules (C) Increased ratio of H 2 PO 4 – to HPO 4– 2 in urine (D) Exchange of intracellular H+ for extracellular K+ (E) Hyperaldosteronism


• RESPIRATORY ACIDOSIS • is caused by hypoventilation, which results in retention of CO 2. • The retention of CO 2 can be caused by 1. inhibition of the medullary respiratory center, 2. paralysis of respiratory muscles, 3. airway obstruction, 4. or failure to exchange CO 2 between pulmonary capillary blood and alveolar gas • The arterial blood profile seen in respiratory acidosis is • p. H ↓ • [HCO 3 ] ↑ • Pco 2 ↑

• The following sequence of events occurs in the generation of respiratory acidosis to produce this blood profile: • 1. Retention of CO 2. Hypoventilation causes retention of CO 2 and an increase in PCO 2. • The increased PCO 2 is the primary disturbance in respiratory acidosis

2. Buffering. • Buffering of the excess CO 2 occurs exclusively in ICF, especially in red blood cells. • To utilize these intracellular buffers, CO 2 diffuses across the cell membranes. Within the cells, CO 2 is converted to H and HCO 3, and the H is buffered by intracellular proteins (e. g. , hemoglobin) and by organic phosphates 3. Respiratory compensation. There is no respiratory compensation for respiratory acidosis, since respiration is the cause of this disorder.

• 4. Renal compensation. • Renal compensation for respiratory acidosis consists of increased H excretion as titratable acid and NH 4 and increased synthesis and reabsorption of new HCO 3. • Reabsorption of new HCO 3 increases the HCO 3 concentration • The Henderson-Hasselbalch equation can be used to understand why the increased HCO 3 concentration is a compensatory response [HCO 3 ] ( ↑ Renal compensation) Pco 2 ( ↑ Primary disturbance)

• In acute respiratory acidosis, renal compensation has not yet occurred, and the p. H tends to be quite low • On the other hand, in chronic respiratory acidosis, renal compensation is occurring, which increases the HCO 3 concentration and tends to normalize both the ratio of HCO 3/CO 2 and the p. H. • The difference between acute and chronic respiratory acidosis lies in the renal compensation

• RESPIRATORY ALKALOSIS • is caused by hyperventilation, which results in excessive loss of CO 2. Hyperventilation can be caused by: 1. direct stimulation of the medullary respiratory center, 2. hypoxemia (which stimulates peripheral chemoreceptors), 3. Mechanical ventilation The arterial blood profile seen in respiratory alkalosis is • p. H ↑ • [HCO 3 ] ↓ • Pco 2 ↓

• The following sequence of events occurs in the generation of respiratory alkalosis to produce this blood profile: • 1. Loss of CO 2. Hyperventilation causes an excessive loss of CO 2 and a decrease in PCO 2. • The decreased PCO 2 is the primary disturbance in respiratory alkalosis and, as predicted by the Henderson-Hasselbalch equation, causes an increase in p. H • The decreased PCO 2 also causes a decreased concentration of HCO 3

2. Buffering occurs exclusively in ICF, particularly in red blood cells. In this case, CO 2 leaves the cells and intracellular p. H increases. 3. Respiratory compensation. As with respiratory acidosis, there is no respiratory compensation for respiratory alkalosis, since respiration is the cause of the disorder.

• 4. Renal compensation for respiratory alkalosis consists of decreased excretion of H as titratable acid and NH 4 and decreased synthesis and reabsorption of new HCO 3 • Decreased reabsorption of HCO 3 decreases the HCO 3 concentration • The Henderson-Hasselbalch equation can be used to understand why the decreased HCO 3 concentration is a compensatory response [HCO 3 ] ( ↓ Renal compensation) Pco 2 ( ↓ Primary disturbance)

• In acute respiratory alkalosis, renal compensation has not yet occurred, and p. H is quite high • In chronic respiratory alkalosis, renal compensation is occurring, which further decreases the blood HCO 3 concentration and tends to normalize both the ratio of HCO 3/CO 2 and the p. H. • The difference between acute and chronic respiratory alkalosis lies in renal compensation
A patient has the following arterial blood values: p. H = 7. 52 PCO 2 = 20 mm Hg [HCO 3–] = 16 m. Eq/L Which of the following statements about this patient is most likely to be correct? (A) He is hypoventilating (B) He has decreased ionized [Ca 2+] in blood (C) He has almost complete respiratory compensation (D) He has an acid–base disorder caused by overproduction of fixed acid (E) Appropriate renal compensation would cause his arterial [HCO 3–] to increase • • •

- Slides: 31