Human Physiology Renal Physiology by Talib F Abbas

Human Physiology Renal Physiology by Talib F. Abbas
Interventions of the renal vessels In the Kidneys, a fluid that resembles plasma is filtered through a collection of capillary tubes in a nephron by a process of glomerular filtration. The resulted urine expelled to the exterior by micturition. Each human kidney has approximately 1. 3 million nephron. The glomerulus, which is about 200μm in diameter, is formed by the invagination of a tuft of capillaries into the dilated, blind end of the nephron (Bowman’s capsule).

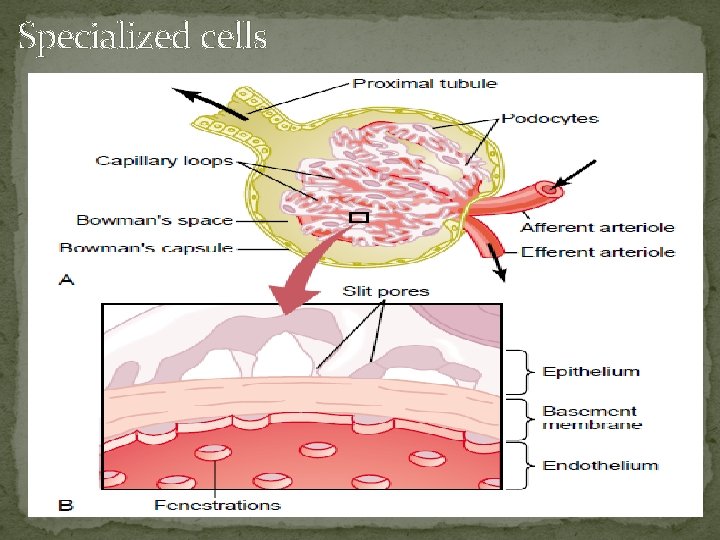
Glomerular slites and podocytes Two cellular layers separate the blood from the glomerular filtrate in Bowman’s capsule: the capillary endothelium and the specialized epithelium of the capsule. The endothelium of the glomerular capillaries is fenestrated, with pores that are 70 to 90 nm in diameter. The endothelium of the glomerular capillaries is completely surrounded by the glomerular basement membrane along with specialized cells called podocytes. Podocytes have numerous pseudopodia that interdigitate to form filtration slits along the capillary wall. The slits are approximately 25 nm wide, and each is closed by a thin membrane. The glomerular basement membrane, the basal lamina, does not contain visible gaps or pores.

Mesangial cells in Capilaries Stellate cells called mesangial cells are located between the basal lamina and the endothelium. They are similar to cells called pericytes, which are found in the walls of capillaries elsewhere in the body. Its happen by active transport pumbs and cotransportation. The thiazide diuretics, which are widely used to treat disorders such as hypertension and heart failure, inhibit the sodiumchloride co-transporter.

Functions of Mesangial cells are especially common between two neighboring capillaries, and in these locations the basal membrane forms a sheath shared by both capillaries. The mesangial cells are contractile and play a role in the regulation of glomerular filtration. Mesangial cells secrete the extracellular matrix, take up immune complexes, and are involved in the progression of glomerular disease. Functionally, the area of filtration in kidney Bowmans is like the Whiteman filter paper 0. 8 m 2. Filtration of plasma and reabsorption of ions Na, K, Cl, and Glucose occur in proximal convoluted tubules, loop of henle, distal convoluted tubules and collecting tubules.

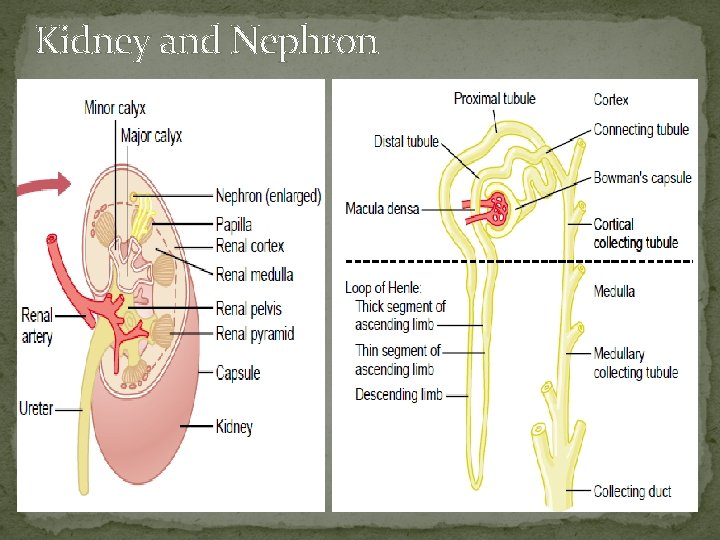
juxtaglomerular apparatus in Macula Densa The thick end of the ascending limb of the loop of Henle reaches the glomerulus of the nephron from which the tubule arose and nestles between its afferent and efferent arterioles. Specialized cells at the end form the macula densa, which is close to the efferent and particularly the afferent arteriole. The macula, the neighboring lacis cells, and the renin-secreting juxtaglomerular cells in the afferent arteriole form the juxtaglomerular apparatus.

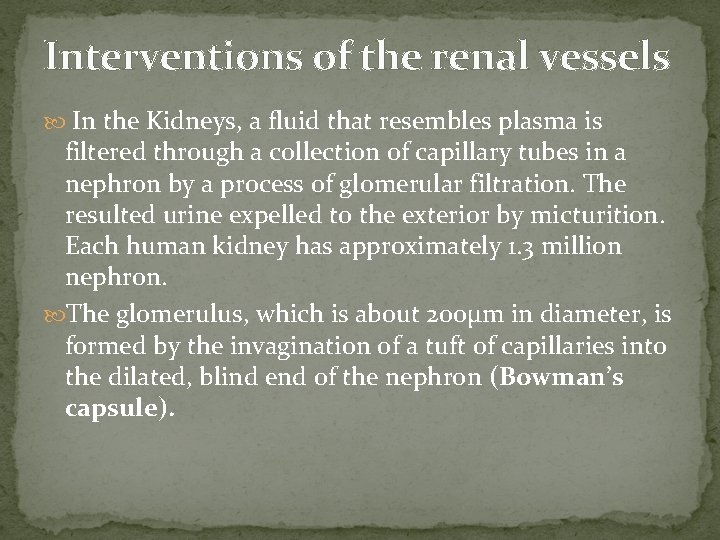
Collacting duct specialized cells The epithelium of the collecting ducts is made up of principal cells (P cells) and intercalated cells (I cells). The P cells, which predominate, are relatively tall and have few organelles. They are involved in Na+ reabsorption and vasopressinstimulated water reabsorption. The I cells, which are present in smaller numbers and are also found in the distal tubules, have more microvilli, cytoplasmic vesicles, and mitochondria. They are concerned with acid secretion and HCO 3– transport. The total length of the nephrons, including the collecting ducts, ranges from 45 to 65 mm.
Kidney and Nephron
Specialized cells

Neural control and Micturation The capillaries draining the tubules of the cortical nephrons form a peritubular network, whereas the efferent arterioles from the juxtamedullary glomeruli drain not only into a peritubular network, but also into vessels that form hairpin loops (the vasa recta). These loops dip into the medullary pyramids alongside the loops of Henle. The renal nerves travel along the renal blood vessels as they enter the kidney. They contain many postganglionic sympathetic efferent fibers and a few afferent fibers. There also appears to be a cholinergic innervation via the vagus nerve, but its function is uncertain. The sympathetic preganglionic innervation comes primarily from the lower thoracic and upper lumbar segments of the spinal cord, and the cell bodies of the postganglionic neurons are in the sympathetic ganglion chain, in the superior mesenteric ganglion, and along the renal artery.

Renorenal Reflex Other renal afferents presumably mediate a renorenal reflex by which an increase in ureteral pressure in one kidney leads to a decrease in efferent nerve activity to the contralateral kidney, and this decrease permits an increase in its excretion of Na+ and water.
Renal Clearance In a resting adult, the kidneys receive 1. 2 to 1. 3 L of blood per minute, or just under 25% of the cardiac output. Renal flow measure through: 1 - Flow meter 2 - electromagnetic 3 - Fick Principle Because the kidney filters plasma, the renal plasma flow equals the amount of a substance excreted per unit of time divided by the renal arteriovenous difference as long as the amount in the red cells is unaltered during passage through the kidney.

Substances used in Renal clearance Any excreted substance can be used If: its concentration in arterial and renal venous plasma can be measured and if it is not metabolized, stored, or produced by the kidney. PAH: p aminohippuric acid (Cpah). Good morning Creatinine Inulin The value obtained should be called the effective renal plasma flow (ERPF) to indicate that the level in renal venous plasma was not measured.

Renal Calculations of GFR In humans, ERPF averages about 625 m. L/min. Example: Concentration of PAH in urine (U PAH): 14 mg/m. L Urine flow (V • ): 0. 9 m. L/min Concentration of PAH in plasma (P PAH): 0. 02 mg/m. L

Renal Calculations of GFR It should be noted that the ERPF determined in this way is the clearance of PAH. The concept of clearance is discussed in detail below. ERPF can be converted to actual renal plasma flow (RPF): Average PAH extraction ratio: 0. 9 From the renal plasma flow, the renal blood flow can be calculated by dividing by 1 minus the hematocrit: Hematocrit (Hct): 45%

GFR if the substance is designated by the letter X, the GFR is equal to the concentration of X in urine (UX) times the urine flow per unit of time (V) divided by the arterial plasma level of X (PX), or UXV • /PX. This value is called the clearance of X (CX). A substance suitable for measuring the GFR should be nontoxic and not metabolized by the body. Inulin, a polymer of fructose with a molecular weight of 5200 that is found in Jerusalem artichokes (Helian-thus tuber-osus), meets these criteria in humans and most animals and is extensively used to measure GFR. Plasma and urinary inulin concentrations are determined and the clearance calculated:


Renal Calculations of GFR In dogs, cats, rabbits, and a number of other mammalian species, clearance of creatinine (CCr) can also be used to determine the precise GFR, but in primates, including humans, some creatinine is secreted by the tubules and some may be reabsorbed. The values agree quite well with the GFR values measured with inulin because, although the value for UCr. V • is high as a result of tubular secretion, the value for PCr is also high as a result of nonspecific chromogens, and the errors thus tend to cancel. The GFR in a healthy person of average size is approximately 125 m. L/min ($).

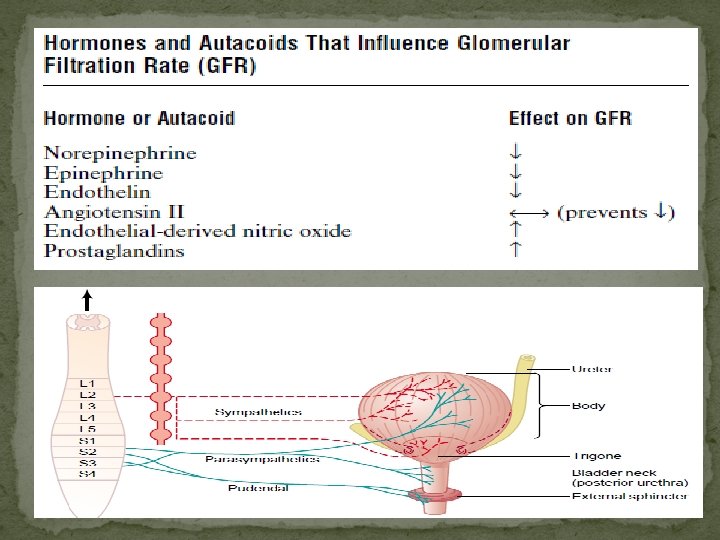
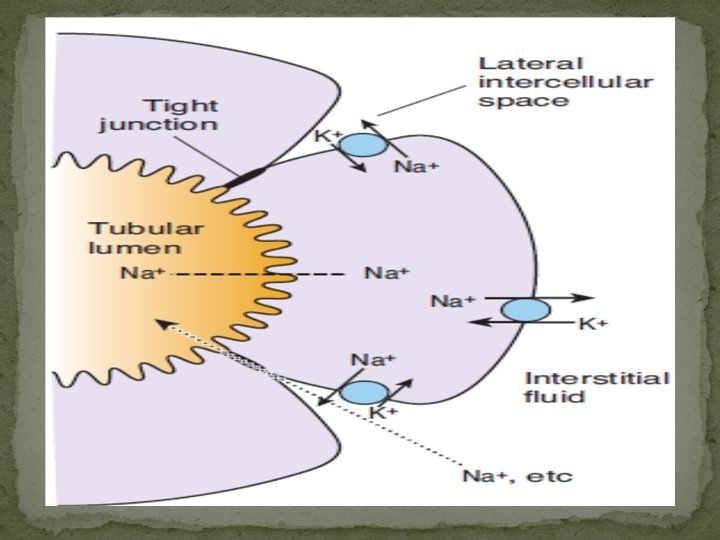
Sodium Reabsorption The reabsorption of Na+ and Cl– plays a major role in body electrolyte and water homeostasis. In addition, Na+ transport is coupled to the movement of H+, glucose, amino acids, organic acids, phosphate, and other electrolytes and substances across the tubule walls. Normally about 60% of the filtered Na+ is reabsorbed in the proximal tubule, primarily by Na–H exchange. Another 30% is absorbed via the Na– 2 Cl–K cotransporter in the thick ascending limb of the loop of Henle, and about 7% is absorbed by Na–Cl cotransporter in the distal convoluted tubule. The remainder of the filtered Na+, about 3%, is absorbed via the ENa. C channels in the collecting ducts, and this is the portion that is regulated by aldosterone in the production of homeostatic adjustments in Na+ balance.

Glucose Reabsorption Glucose, amino acids, and bicarbonate are reabsorbed along with Na+ in the early portion of the proximal tubule. Glucose is typical of substances removed from the urine by secondary active transport. Glucose and Na+ bind to the sodium-dependentglucose transporter (SGLT) 2 in the apical membrane. Na+ is then pumped out of the cell into the interstitium, and the glucose is transported by glucose transporter (GLUT) 2 into the interstitial fluid.

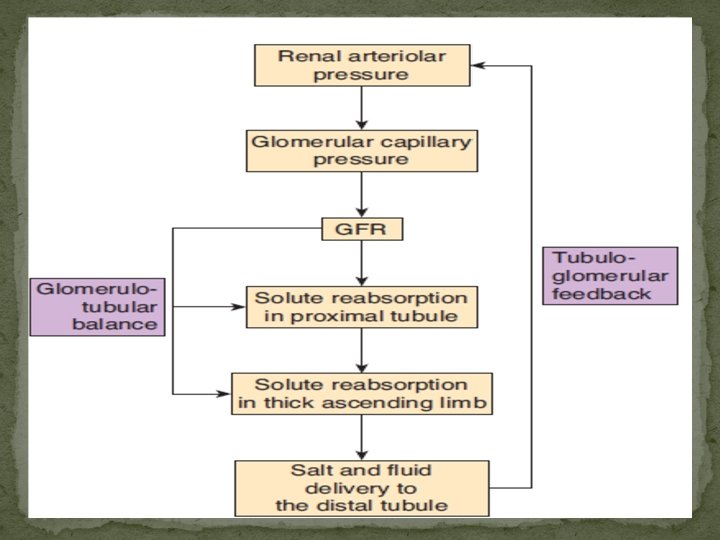
Tubuloglomerular Feedback and Glomerulotubular Balance Signals from the renal tubule in each nephron feed back to affect filtration in its glomerulus. As the rate of flow through the ascending limb of the loop of Henle and first part of the distal tubule increases, glomerular filtration in the same nephron decreases, and, conversely, a decrease in flow increases the GFR. This process, which is called tubuloglomerular feedback, tends to maintain the constancy of the load delivered to the distal tubule. The sensor for this response is the macula densa. The amount of fluid entering the distal tubule at the end of the thick ascending limb of the loop of Henle depends on the amount of Na+ and Cl– in it. .

Water Transport Normally, 180 L of fluid is filtered through the glomeruli each day, while the average daily urine volume is about 1 L. The same load of solute can be excreted per 24 h in a urine volume of 500 m. L with a concentration of 1400 m. Osm/kg or in a volume of 23. 3 L with a concentration of 30 m. Osm/kg Rapid diffusion of water across cell membranes depends on the presence of water channels, integral membrane proteins called aquaporins. To date, 13 aquaporins have been cloned; however, only 4 aquaporins (aquaporin-1, aquaporin-2, aquaporin- 3, and aquaporin-4) play a key role in the kidney. When aquaporin-1 was knocked out in mice, proximal tubular water permeability was reduced by 80%. When the mice were subjected to dehydration, their urine osmolality did not increase (<700 m. Osm/kg), even though other renal aquaporins were present.

Collecting ducts The collecting ducts have two portions: a cortical portion and a medullary portion. The changes in osmolality and volume in the collecting ducts depend on the amount of vasopressin acting on the ducts. This antidiuretic hormone from the posterior pituitary gland increases the permeability of the collecting ducts to water. The key to the action of vasopressin on the collecting ducts is aquaporin-2, which is mainly work through vasopressin V 2 receptor, mediated the c. AMP –protien kinase A.
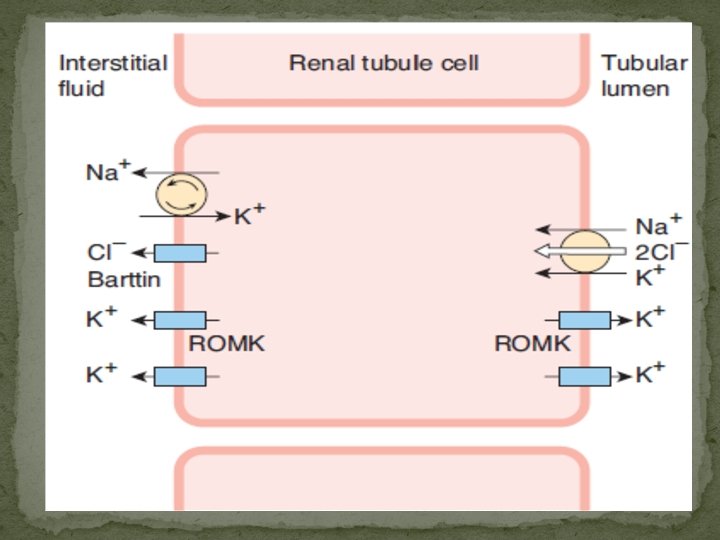

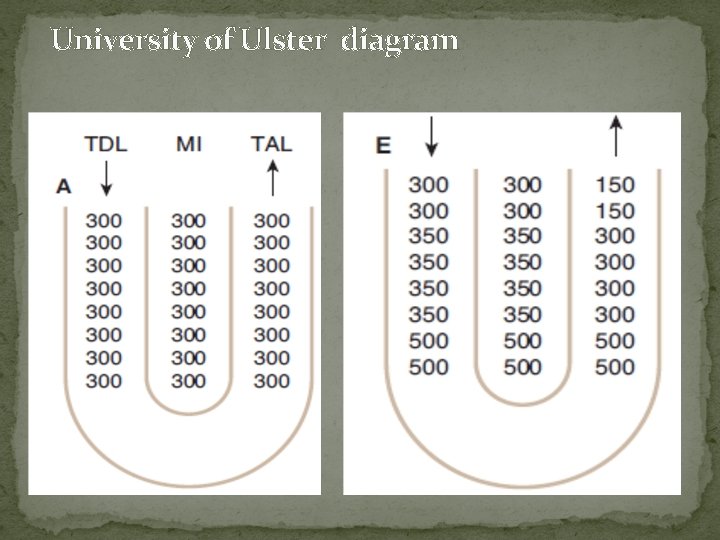
The countercurrent mechanism The concentrating mechanism depends upon the maintenance of a gradient of increasing osmolality along the medullary pyramids. This gradient is produced by the operation of the loops of Henle as countercurrent multipliers and maintained by the operation of the vasa recta as countercurrent exchangers. A countercurrent system is a system in which the inflow runs parallel to, counter to, and in close proximity to the outflow for some distance. This occurs for both the loops of Henle and the vasa recta in the renal medulla.

The countercurrent mechanism The operation of each loop of Henle as a countercurrent multiplier depends on the high permeability of the thin descending limb to water (via aquaporin-1), the active transport of Na+ and Cl– out of the thick ascending limb, and the inflow of tubular fluid from the proximal tubule, with outflow into the distal tubule. The process can be explained using hypothetical steps. In juxtamedullary nephrons with longer loops and thin ascending limbs, the osmotic gradient is spread over a greater distance and the osmolality at the tip of the loop is greater. This is because thin ascending limb is relatively impermeable to water but permeable to Na+ and Cl–. Therefore, Na+ and Cl–move down their concentration gradients into the interstitium, and there is additional passive countercurrent multiplication. The greater the length of the loop of Henle, the greater the osmolality that can be reached at

The countercurrent mechanism Therefore, the solutes tend to recirculate in the medulla and water tends to bypass it, so that hypertonicity is maintained. The water removed from the collecting ducts in the pyramids is also removed by the vasa recta and enters the general circulation. Countercurrent exchange is a passive process; it depends on movement of water and could not maintain the osmotic gradient along the pyramids if the process of countercurrent multiplication in the loops of Henle were to cease.
University of Ulster diagram

Thank you
- Slides: 33