Human Physiology Renal Physiology by Talib F Abbas

Human Physiology Renal Physiology by Talib F. Abbas

Role of Urea in ECF � Urea contributes to the establishment of the osmotic gradient in the medullary pyramids and to the ability to form a concentrated urine in the collecting ducts. Urea transport is mediated by urea transporters, presumably by facilitated diffusion. There at least four isoforms of the transport protein UT-A in the kidneys (UT-A 1 to UT-A 4); UT-B is found in erythrocytes. �a high-protein diet increases the ability of the kidneys to concentrate the urine.
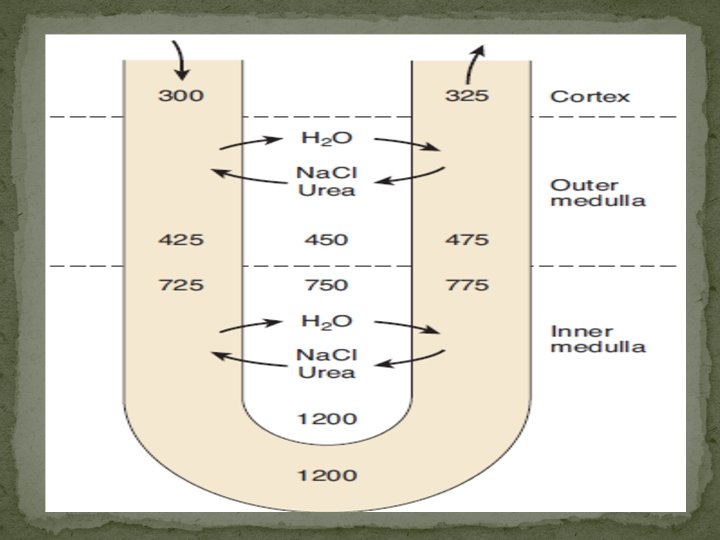

Water diuresis � The osmotic gradient in the medullary pyramids would not last long if the Na+ and urea in the interstitial spaces were removed by the circulation. These solutes remain in the pyramids primarily because the vasa recta operate as countercurrent exchangers. �the solutes tend to recirculate in the medulla and water tends to bypass it, so that hypertonicity is maintained. The water removed from the collecting ducts in the pyramids is also removed by the vasa recta and enters the general circulation. ? Nilesat swees role

Osmotic diuresis �The presence of large quantities of unreabsorbed solutes in the renal tubules causes an increase in urine volume called osmotic diuresis. �Therefore, they “hold water in the tubules. ” In addition, the concentration gradient against which Na+ can be pumped out of the proximal tubules is limited. Osmotic diuresis is produced by the administration of compounds such as mannitol and related polysaccharides that are filtered but not reabsorbed. �For example, in diabetes mellitus, if blood glucose is high, glucose in the glomerular filtrate is high, thus the glucose will remain in the tubules causing polyuria. Osmotic diuresis can also be produced by the infusion of large amounts of sodium chloride or urea.

Free water clearance � In order to quantitate the gain or loss of water by excretion of a concentrated or dilute urine, the "free water clearance"(CH 2 O) is sometimes calculated. This is the difference between the urine volume and the clearance of osmoles (COsm): � where V • is the urine flow rate and UOsm and POsm the urine and plasma osmolality, respectively. COsm is the amount of water necessary to excrete the osmotic load in a urine that is isotonic with plasma. Therefore, CH 2 O is negative when the urine is hypertonic and positive when the urine is hypotonic. ﺍﻻﻫﻮﺍﺭ ﺿﻤﻦ ﺍﻟﺘﺮﺍﺙ ﺍﻟﻌﺎﻟﻤﻲ ﻭﺿﻊ

Renal Hydrogen secretion The cells of the proximal and distal tubules secrete hydrogen ions. �Acidification also occurs in the collecting ducts. The reaction that is primarily responsible for H+ secretion in the proximal tubules is Na–H exchange. This is an example of secondary active transport; extrusion of Na+ from the cells into the interstitium by Na, K ATPase lowers intracellular Na+, and this causes Na+ to enter the cell from the tubular lumen, with coupled extrusion of H+. The H+ comes from intracellular dissociation of H 2 CO 3, and the HCO 3– that is formed diffuses into the interstitial fluid. Thus, for each H+ ion secreted, one Na+ ion and one HCO 3– ion enter the interstitial fluid. �
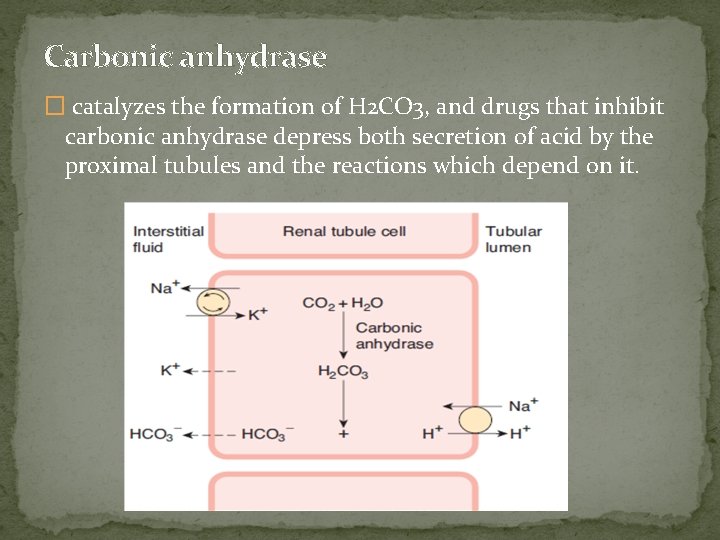
Carbonic anhydrase � catalyzes the formation of H 2 CO 3, and drugs that inhibit carbonic anhydrase depress both secretion of acid by the proximal tubules and the reactions which depend on it.

Ammonia secretion � Reactions in the renal tubular cells produce NH 4+ and HCO 3–. NH 4+ is in equilibrium with NH 3 and H+ in the cells. Because the p. K' of this reaction is 9. 0, the ratio of NH 3 to NH 4+ at p. H 7. 0 is 1: 100. However, NH 3 is lipidsoluble and diffuses across the cell membranes down its concentration gradient into the interstitial fluid and tubular urine. In the urine it reacts with H+ to form NH 4+, and the NH 4+ remains in the urine.

Ammonia secreation �The principal reaction producing NH 4+ in cells is conversion of glutamine to glutamate. This reaction is catalyzed by the enzyme glutaminase, which is abundant in renal tubular cells. Glutamic dehydrogenase catalyzes the conversion of glutamate to α-ketoglutarate, with the production of more NH 4+. Subsequent metabolism of αketoglutarate utilizes 2 H+, freeing 2 HCO 3–.
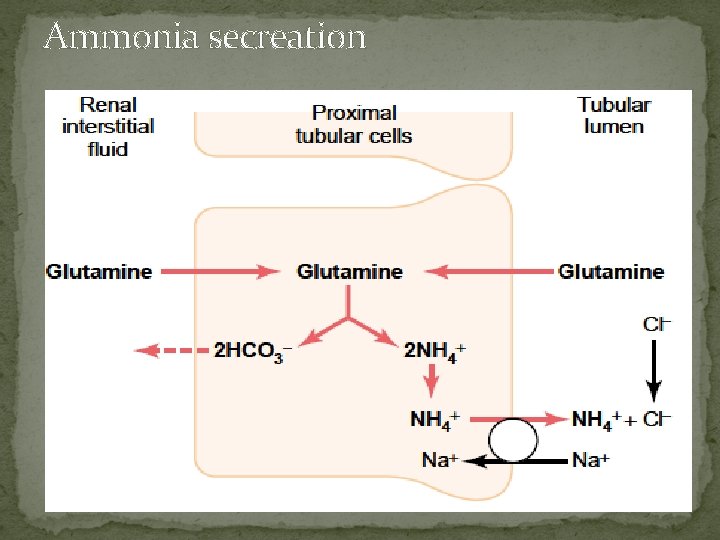
Ammonia secreation
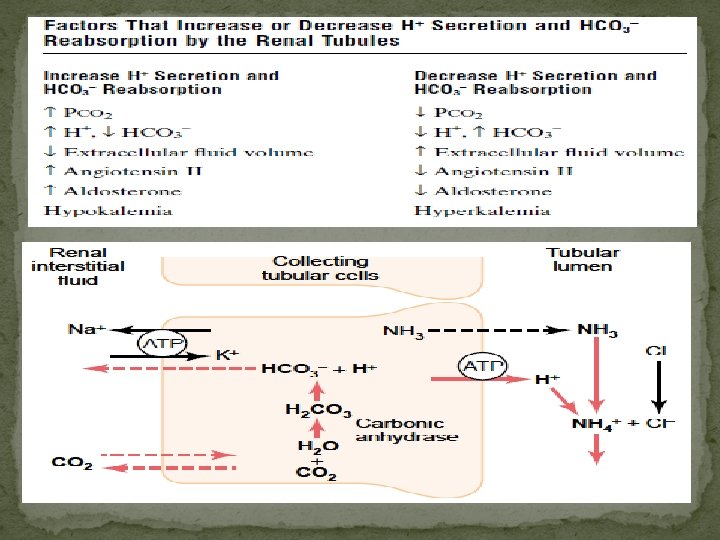

Factors effecting acid secretion � Renal acid secretion is altered by changes in the intracellular PCO 2, K+ concentration, carbonic anhydrase level, and adrenocortical hormone concentration. When the PCO 2 is high (respiratory acidosis), more intracellular H 2 CO 3 is available to buffer the hydroxyl ions and acid secretion is enhanced, whereas the reverse is true when the PCO 2 falls. K+ depletion enhances secretion, apparently because the loss of K+ causes intracellular acidosis even though the plasma p. H may be elevated. Conversely, K+ excess in the cells inhibits acid secretion. When carbonic anhydrase is inhibited, acid secretion is inhibited because the formation of H 2 CO 3 is decreased. Aldosterone and the other adrenocortical steroids that enhance tubular reabsorption of Na+ also increase the secretion of H+ and K+.
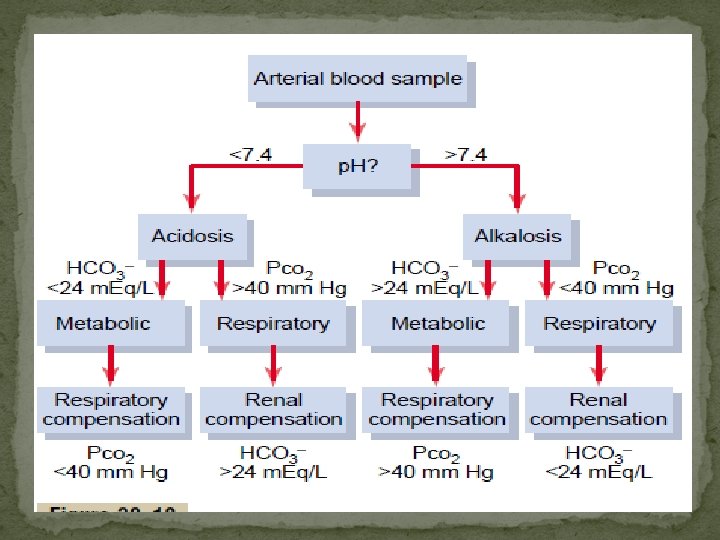

Bicarbonate excretion �Although the process of HCO 3– reabsorption does not actually involve transport of this ion into the tubular cells, HCO 3– reabsorption is proportional to the amount filtered over a relatively wide range. �When the plasma HCO 3– concentration is low, all the filtered HCO 3– is reabsorbed; but when the plasma HCO 3– concentration is high; that is, above 26 to 28 m. Eq/L (the renal threshold for HCO 3–), HCO 3– appears in the urine and the urine becomes alkaline.

Bicarbonate excreation �when the plasma HCO 3– falls below about 26 m. Eq/L, the value at which all the secreted H+ is being used to reabsorb HCO 3–, more H+ becomes available to combine with other buffer anions. �Therefore, the lower the plasma HCO 3– concentration drops, the more acidic the urine becomes and the greater its NH 4+ content.

Defense of Hydrogen concentration �Defense of H+ concentration: The mystique that envelopes the subject of acid–base balance makes it necessary to point out that the core of the problem is not “buffer base” or “fixed cation” or the like, but simply the maintenance of the H+ concentration of the ECF.
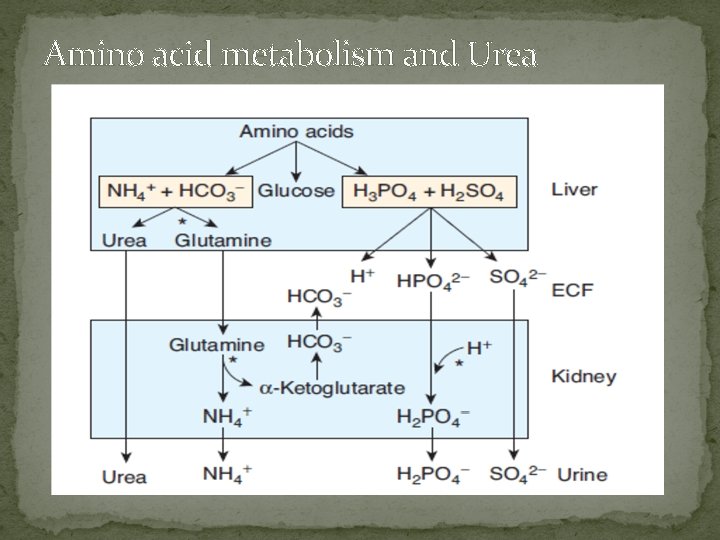
Amino acid metabolism and Urea

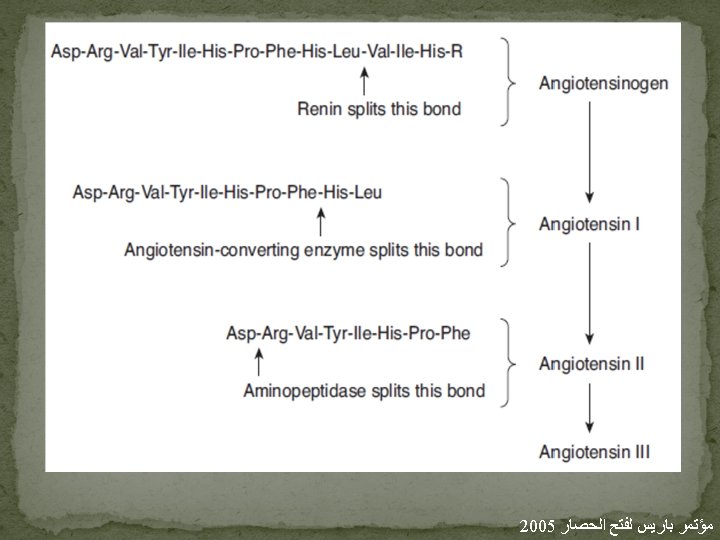
Renin-Angiotensin system �The rise in blood pressure produced by injection of kidney extracts is due to renin, an acid protease secreted by the kidneys into the bloodstream. �This enzyme acts in concert with angiotensinconverting enzyme to form angiotensin II. �plasma renin concentration (PRC): exogenous angiotensinogen is often added, Deficiency of angiotensinogen as well as renin cause low plasma renin activity (PRA). �The plasma angiotensin II concentration in such subjects is about 25 pg/m. L (approximately 25 pmol/L).
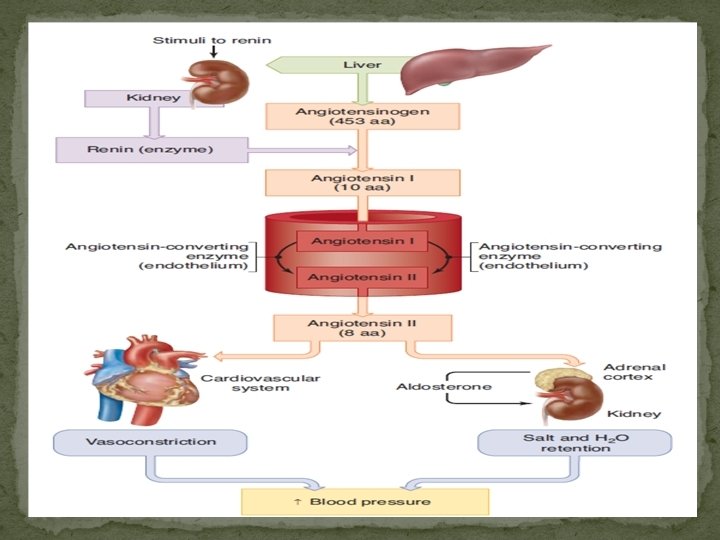

The Juxtaglomerular Apparatus TV �The renin in kidney extracts and the bloodstream is produced by the juxtaglomerular cells (JG cells). These epitheloid cells are located in the media of the afferent arterioles as they enter the glomeruli. �The membrane-lined secretory granules in them have been shown to contain renin. �Renin is also found in agranular lacis cells that are located in the junction between the afferent and efferent arterioles, but its significance in this location is unknown. �The lacis cells, the JG cells, and the macula densa constitute the juxtaglomerular apparatus.

Renin Regulation Factors �intrarenal baroreceptor mechanism that causes renin secretion to decrease when arteriolar pressure at the level of the JG cells increases. �Renin secretion is inversely proportional to the amount of Na+ and Cl– entering the distal renal tubules from the loop of Henle. �NO and K+ level effect the Renin secreation. �Angiotensin II feeds back to inhibit renin secretion by a direct action on the JG cells. �Vassopressin inhibit renin. �increased activity of the sympathetic nervous system increases renin secretion � ﺧﻼﻳﺎ ﺍﻟﺴﻴﺲ ﺳﻼﻟﻢ ﺿﻤﻦ ﺑﺮﻧﺎﻣﺞ ﺍﻻﺭﺽ ﻓﻲ ﺍﻟﻨﺠﻒ ﻣﻊ ﺑﻨﺎﺀ ﺣﻤﺎﻡ ﺗﺤﺖ

Thank you
- Slides: 24