Human Biology BIOL 104 Talk two Chemistry of

Human Biology (BIOL 104) Talk two: Chemistry of life The Chemical Foundation of Life (Chapter Two) Biological Macromolecules (Chapter Three) Don’t Panic!

WHY? !!!!!! • In studying human biology, it’s useful to understand some basic chemistry – Chemical reactions explain the effects – harmful or helpful – of substances we take into our bodies • This will actually help you to identify structures, functions, and processes of many of the human body systems. • It will help you understand how everything in the human body works together for healthy life to exist. – In other words – it will help you connect up all the dots

What are we looking at? • All matter, including living things, is made up of various combinations of elements. • Some of the most abundant elements in living organisms include carbon, hydrogen, nitrogen, oxygen, sulfur, and phosphorus. • These elements form the major biological molecules—nucleic acids, proteins, carbohydrates, and lipids— that are the fundamental components of living matter. • Biologists study these important molecules to understand their unique structures which determine their specialized functions. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University Credit: modification of work by Christian Guthier
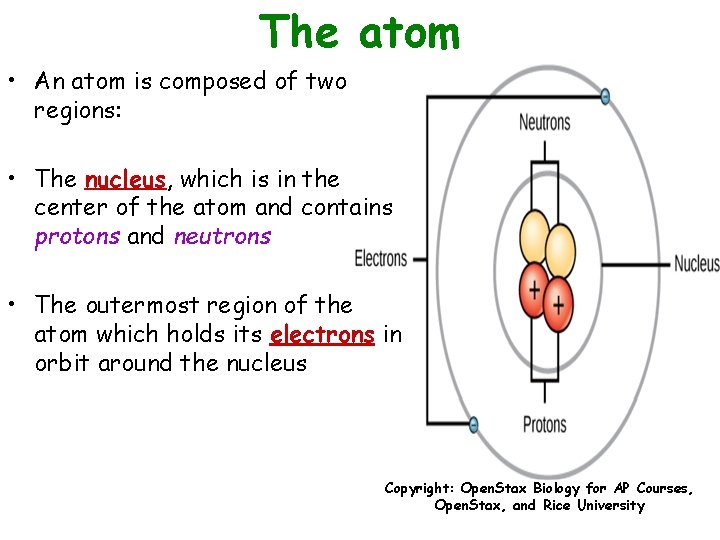
The atom • An atom is composed of two regions: • The nucleus, which is in the center of the atom and contains protons and neutrons • The outermost region of the atom which holds its electrons in orbit around the nucleus Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University
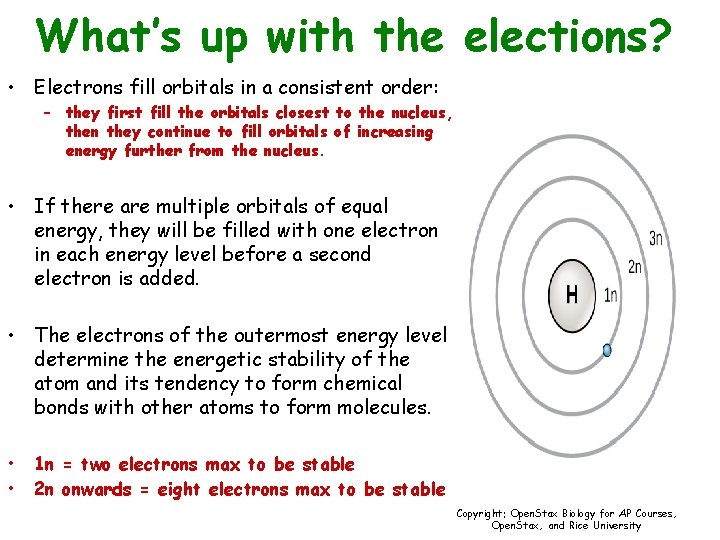
What’s up with the elections? • Electrons fill orbitals in a consistent order: – they first fill the orbitals closest to the nucleus, then they continue to fill orbitals of increasing energy further from the nucleus. • If there are multiple orbitals of equal energy, they will be filled with one electron in each energy level before a second electron is added. • The electrons of the outermost energy level determine the energetic stability of the atom and its tendency to form chemical bonds with other atoms to form molecules. • • 1 n = two electrons max to be stable 2 n onwards = eight electrons max to be stable Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University
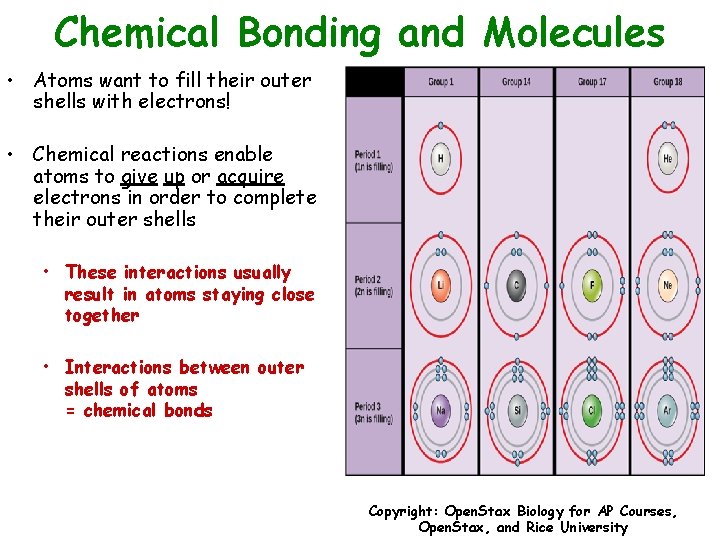
Chemical Bonding and Molecules • Atoms want to fill their outer shells with electrons! • Chemical reactions enable atoms to give up or acquire electrons in order to complete their outer shells • These interactions usually result in atoms staying close together • Interactions between outer shells of atoms = chemical bonds Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University
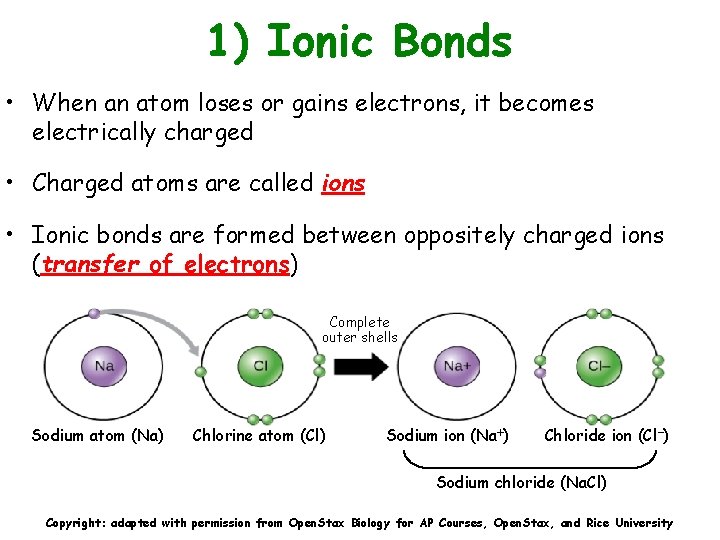
1) Ionic Bonds • When an atom loses or gains electrons, it becomes electrically charged • Charged atoms are called ions • Ionic bonds are formed between oppositely charged ions (transfer of electrons) Complete outer shells Sodium atom (Na) Chlorine atom (Cl) Sodium ion (Na ) Chloride ion (Cl ) Sodium chloride (Na. Cl) Copyright: adapted with permission from Open. Stax Biology for AP Courses, Open. Stax, and Rice University

2) Covalent Bonds • Sharing of electrons between atoms to form covalent bonds. • These bonds are stronger and much more common than ionic bonds in the molecules of living organisms. • Commonly found in carbon-based organic molecules, such as our DNA and proteins • Covalent bonds are also found in inorganic molecules like H 2 O, CO 2, and O 2. One, two, or three pairs of electrons may be shared, making single, double, and triple bonds, respectively. • The more covalent bonds between two atoms, the stronger their connection. Thus, triple bonds are the strongest. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University

The Water Molecule • In a polar covalent bond the electrons are unequally shared by the atoms and are attracted more to one nucleus than the other. • Because of the unequal distribution of electrons between the atoms of different elements, a slightly positive (δ+) or slightly negative (δ–) charge develops. • This partial charge is an important property of water and accounts for many of its characteristics. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University

Water • Water - one of the most important molecules in life. · 70% of the bodies mass is water · 2/3 of total body water is intracellular (55 -66% body weight of men and 10% less for women) · The rest is interstitial fluid of which 25% is in the blood plasma. • p. H - The body tightly controls both the volume and p. H of water. · The bicarbonate system is crucial for blood maintenance · changes of p. H greater than 0. 1 are dangerous and can lead to coma -diabetics

Life on Earth would not be possible without water Its chemical and physical properties actually defy some fundamental laws of physics Almost all biochemical reactions require water!

Water is a polar molecule • When electrons are not shared equally in a covalent bond, the molecule is described as polar. • Water molecules are polar. This means that while water molecules are neutral as a whole, one end of the water molecule tends to have a positive charge while the other has a negative charge. • The oxygen end has a slight negative charge while the hydrogen end has a slight positive charge. • Each end of a water molecule is attracted to the opposite charged end of another water molecule. • Water's polarity is responsible for the "stickiness" or cohesion between the molecules. From the wikimedia free licensed media file repository
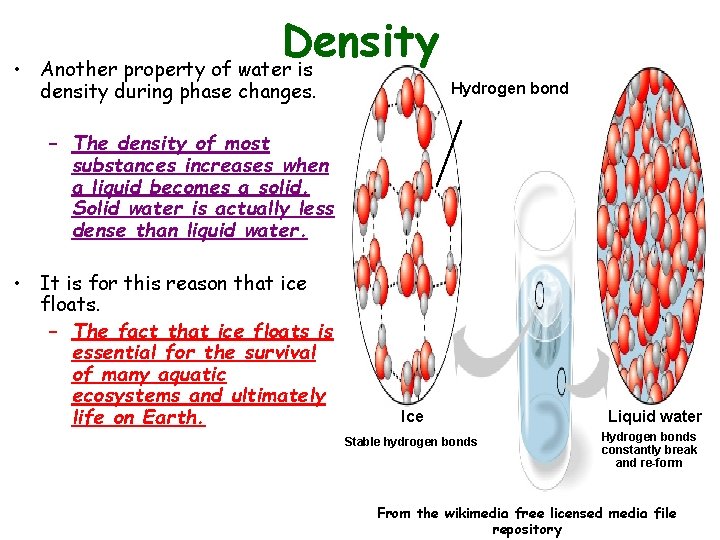
• Density Another property of water is density during phase changes. Hydrogen bond – The density of most substances increases when a liquid becomes a solid. Solid water is actually less dense than liquid water. • It is for this reason that ice floats. – The fact that ice floats is essential for the survival of many aquatic ecosystems and ultimately life on Earth. Ice Stable hydrogen bonds Liquid water Hydrogen bonds constantly break and re-form From the wikimedia free licensed media file repository

• Hydrogen bonding makes ice less dense than liquid water. (a) The lattice structure of ice makes it less dense than the freely flowing molecules of liquid water. (b) enabling it to float on water. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University

Capillary Action & Surface tension • Cohesion of water causes capillary attraction, which is the ability of water to move upward in small spaces. • Cohesion makes it possible for water to move up the fibers of a plant. • – This is how plants get the water they need to survive. – In addition, it moves water upwards in soil. – Allows water to be taken into human cells attached to other molecules Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University credit: modification of work by Pearson-Scott Foresman, donated to the Wikimedia Foundation

Capillary Action & Surface tension • Cohesion allows for the development of surface tension, the capacity of a substance to withstand being ruptured when placed under tension or stress. • This is also why water forms droplets when placed on a dry surface rather than being flattened out by gravity. • Cohesion and surface tension keep the hydrogen bonds of water molecules intact and support the item floating on the top. Water’s cohesive and adhesive properties allow this water strider (Gerris sp. ) to stay afloat. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University (credit: Tim Vickers)
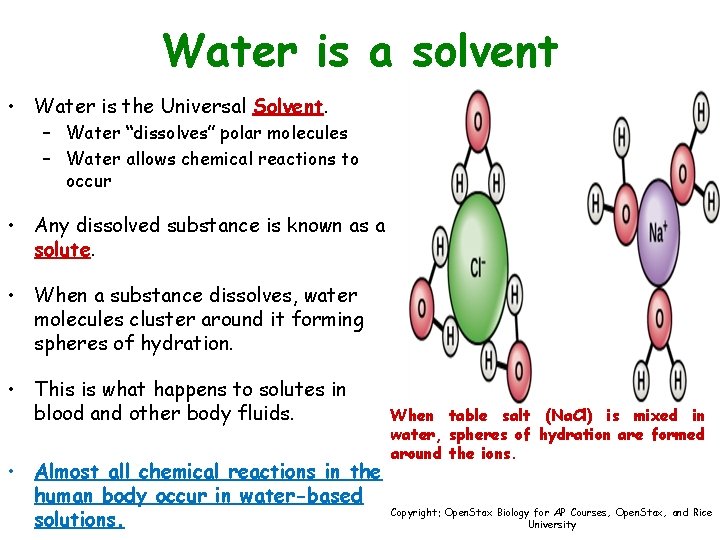
Water is a solvent • Water is the Universal Solvent. – Water “dissolves” polar molecules – Water allows chemical reactions to occur • Any dissolved substance is known as a solute. • When a substance dissolves, water molecules cluster around it forming spheres of hydration. • This is what happens to solutes in blood and other body fluids. • Almost all chemical reactions in the human body occur in water-based solutions. When table salt (Na. Cl) is mixed in water, spheres of hydration are formed around the ions. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University
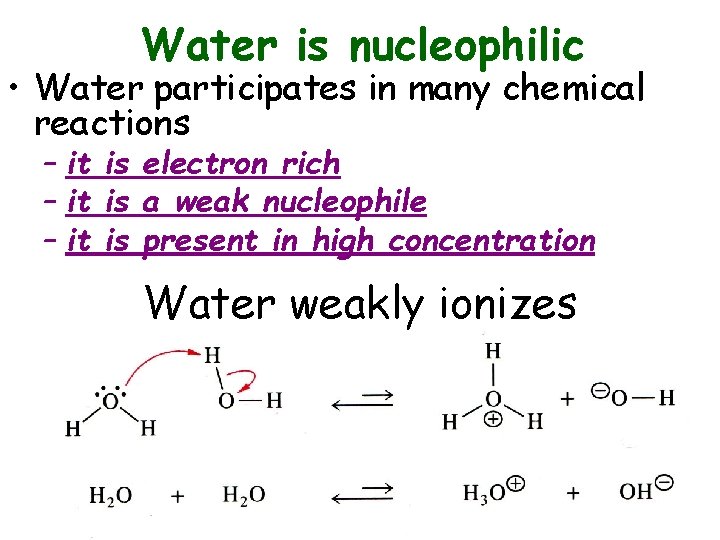
Water is nucleophilic • Water participates in many chemical reactions – it is electron rich – it is a weak nucleophile – it is present in high concentration Water weakly ionizes

p. H and buffers • Measure of the acidity or basicity of an aqueous solution. – Solutions with a p. H less than 7 are said to be acidic – Solutions with a p. H greater than 7 are basic or alkaline. – Pure water has a p. H very close to 7 • • Acid A chemical compound that donates H+ ions to solutions. Base A compound that accepts H+ ions and removes them from solution. • Remember H 3 O+ and OH-? • The H 3 O+ donates H+ to a solution and the OH- removes H+ from a solution • If an equal number of these ions are present in a solution the p. H will not change as it is said to be buffered.
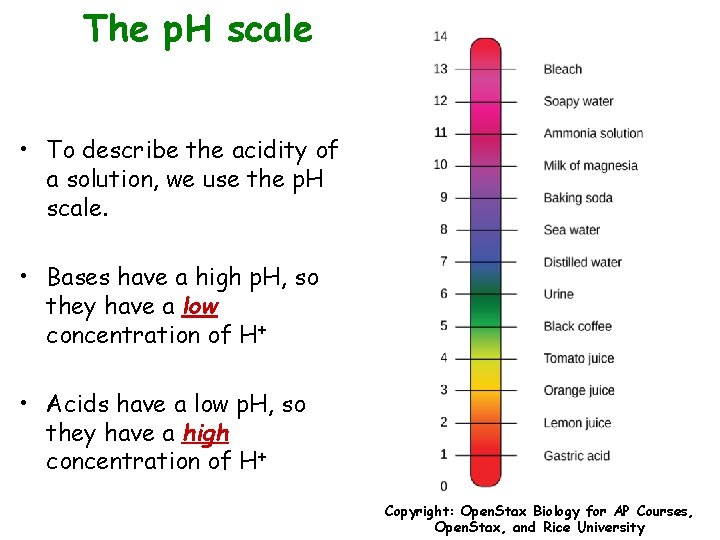
The p. H scale • To describe the acidity of a solution, we use the p. H scale. • Bases have a high p. H, so they have a low concentration of H+ • Acids have a low p. H, so they have a high concentration of H+ Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University

H 20 helps buffer our blood • Water reacts with CO 2 to form an important blood buffer • We breath in and out gaseous CO 2 • In the blood, CO 2 reacts with water to form the buffering compound H 2 CO 3 –carbonic acid • Disturbances in blood buffering system leads to acidosis (p. H below 7. 1) or alkalosis (p. H above 7. 6) • Bleeding in lungs – death! “a few good men” The body’s buffering of blood p. H levels. The blue arrows show the process of raising p. H as more CO 2 is made. The purple arrows indicate the reverse process: the lowering of p. H as more bicarbonate is created. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University

The Biomolecules • These are the molecules of life – Carbohydrates – Lipids – Proteins • Glycoproteins – Nucleotides and Nucleic acids • These molecules are involved in the chemical reactions that allow us to live. • Remember the levels of biological organization? • Understanding the molecules allows you to understand how the cell works – Which is the fundamental unit of life

Carbohydrates • Carbohydrates constitute more than 1/2 of organic molecules • Main role of carbohydrates in nature: · Storage of energy · Structural support · Lipid and protein modification: · membranes asymmetry, recognition by Ig. G/fertilization/virus recognition/cell communication Definition: Carbohydrates, Sugars and Saccharides- are all polyhydroxy · (at least 2 OH) Cn(H 20) n = hydrate of carbon

Basic facts Monosaccharides - Simple sugars • Single polyhydroxyl · Can’t be hydrolyzed to simpler form Trioses - Smallest monosaccharides have three carbon atoms Tetroses (4 C) Pentose (5 C) Hexoses (6 C) Heptoses (7 C) etc… Disaccharide - two sugars linked together. Can be the same molecule or two different sugars. Attached together via a glycosidic linkage Oligosaccharide - 2 to 6 monosaccharides Polysaccharides - straight or branched long chain monosaccharides. Bonded together by glycosidic linkages
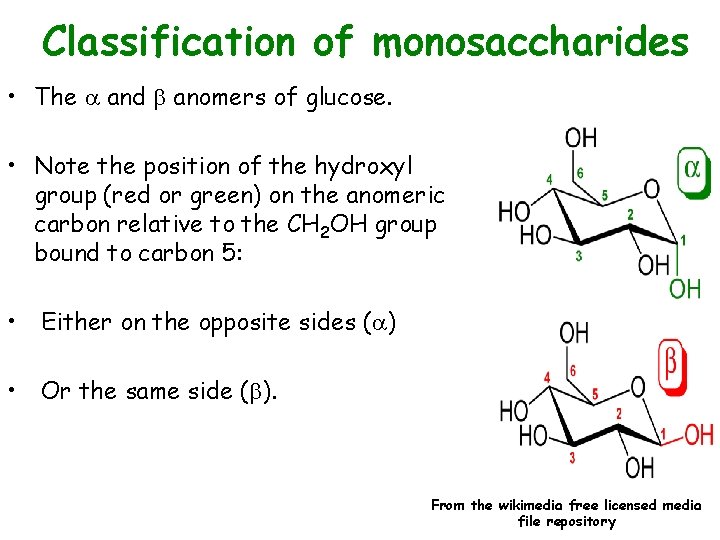
Classification of monosaccharides • The and b anomers of glucose. • Note the position of the hydroxyl group (red or green) on the anomeric carbon relative to the CH 2 OH group bound to carbon 5: • Either on the opposite sides ( ) • Or the same side (b). From the wikimedia free licensed media file repository
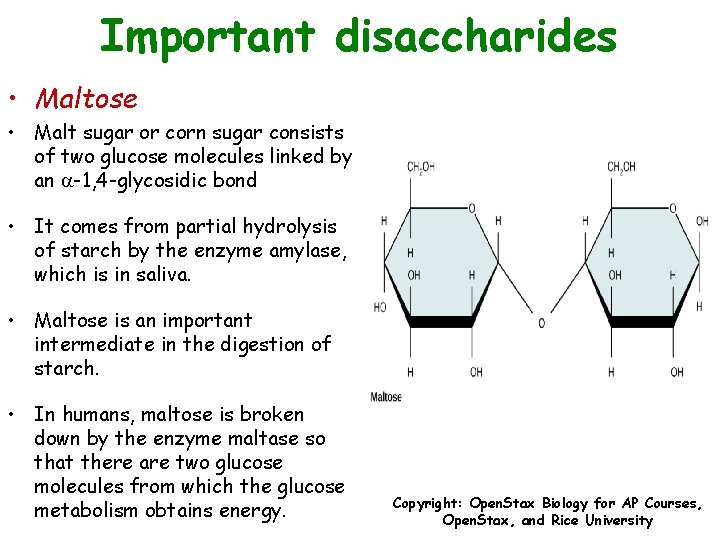
Important disaccharides • Maltose • Malt sugar or corn sugar consists of two glucose molecules linked by an -1, 4 -glycosidic bond • It comes from partial hydrolysis of starch by the enzyme amylase, which is in saliva. • Maltose is an important intermediate in the digestion of starch. • In humans, maltose is broken down by the enzyme maltase so that there are two glucose molecules from which the glucose metabolism obtains energy. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University

Important disaccharides • Sucrose (table sugar) • Sugar beets, fruits and vegetables all contain sucrose. • When sucrose is consumed, it is broken down to glucose and fructose. • The body will use glucose as its main energy source • The excess energy from fructose, if not needed, will be poured into fat synthesis, which is stimulated by the insulin released in response to glucose Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University

Sucrose (table sugar) • Human health • Tooth decay has become a prominent health hazard associated with the consumption of sugars, especially sucrose. – Oral bacteria such as Streptococcus mutans live in dental plaque and metabolize sucrose into lactic acid. – The resultant lactic acid lowers the p. H of the tooth's surface, stripping it of minerals in the process known as tooth decay • Obesity • A United Nations report (2011) stated that obesity may correlate better with sugar consumption than with fat consumption, and that reducing fat consumption while increasing sugar consumption may increase the level of obesity. Year Sex Carbohydrate Fat Protein Obesity 1971 Male 42. 4% 36. 9% 16. 5% 12. 1% 1971 Female 45. 4% 36. 1% 16. 9% 16. 6% 2000 Male 49. 0% 32. 8% 15. 5% 27. 7% 2000 Female 51. 6% 32. 8% 15. 1% 34. 0%
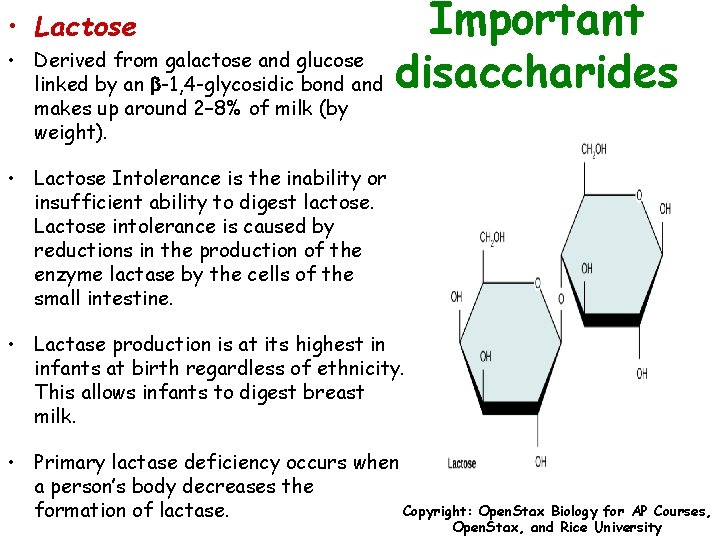
Important Derived from galactose and glucose linked by an b-1, 4 -glycosidic bond and disaccharides • Lactose • makes up around 2– 8% of milk (by weight). • Lactose Intolerance is the inability or insufficient ability to digest lactose. Lactose intolerance is caused by reductions in the production of the enzyme lactase by the cells of the small intestine. • Lactase production is at its highest in infants at birth regardless of ethnicity. This allows infants to digest breast milk. • Primary lactase deficiency occurs when a person’s body decreases the Copyright: Open. Stax Biology for AP Courses, formation of lactase. Open. Stax, and Rice University
Important disaccharides • Lactose • Symptoms of lactose intolerance can be mild to severe, depending on how much lactase your body makes. Symptoms usually begin 30 minutes to 2 hours after you eat or drink milk products. • If you have lactose intolerance, your symptoms may include – – – Bloating. Pain or cramps. Gurgling or rumbling sounds in your belly. Gas. Loose stools or diarrhea. Throwing up. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University

Important disaccharides • Lactose • Primary lactose intolerance appears to have a genetic component with specific populations showing high levels of intolerance. Ethnicity American Indians Asians African Americans Ashkenazi Jews Hispanics Caucasian Americans Northern European Origin % Adults with Primary Lactose Intolerance 80 to 100 % 90 - 95% 60 – 80% 50 – 80% 13% 1 - 2% Lactose Intolerance: Information for Health Care Providers. U. S. Department of Health and Human Services. NIH Publication Number 05 -5305 B. 2006
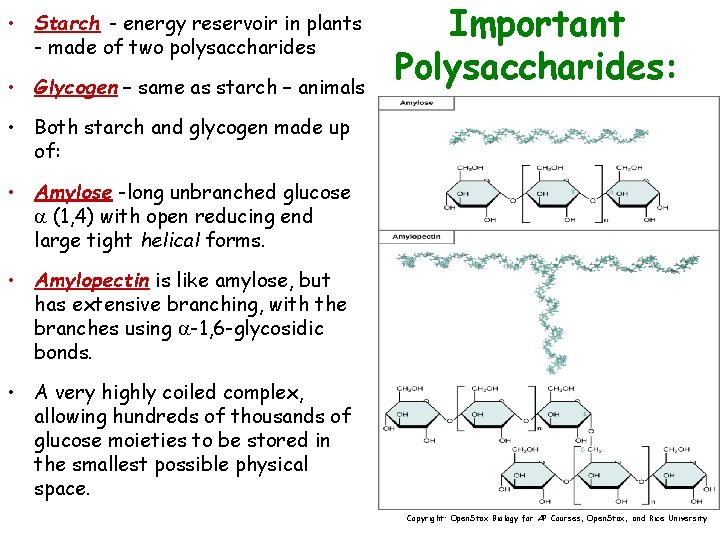
• Starch - energy reservoir in plants - made of two polysaccharides • Glycogen – same as starch – animals Important Polysaccharides: • Both starch and glycogen made up of: • Amylose -long unbranched glucose (1, 4) with open reducing end large tight helical forms. • Amylopectin is like amylose, but has extensive branching, with the branches using -1, 6 -glycosidic bonds. • A very highly coiled complex, allowing hundreds of thousands of glucose moieties to be stored in the smallest possible physical space. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University

Glycogen • Mix both of these two types of polysaccharides together and you get glycogen. • In humans, glycogen is made and stored primarily in the cells of the liver and the muscles, and functions as the secondary long-term energy storage. • In the liver cells (hepatocytes), glycogen can compose up to 8% of its fresh weight (100– 120 g in an adult) soon after a meal. • Only the glycogen stored in the liver can be made accessible to other organs. From the wikimedia free licensed media file repository
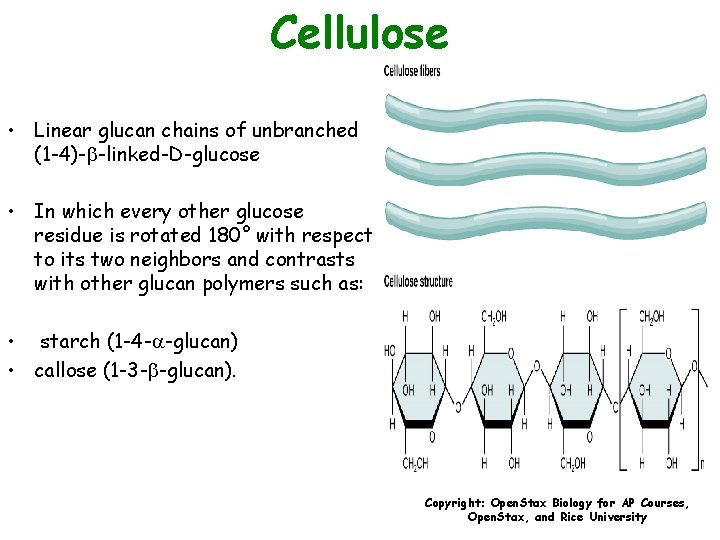
Cellulose • Linear glucan chains of unbranched (1 -4)-b-linked-D-glucose • In which every other glucose residue is rotated 180° with respect to its two neighbors and contrasts with other glucan polymers such as: • starch (1 -4 - -glucan) • callose (1 -3 -b-glucan). Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University
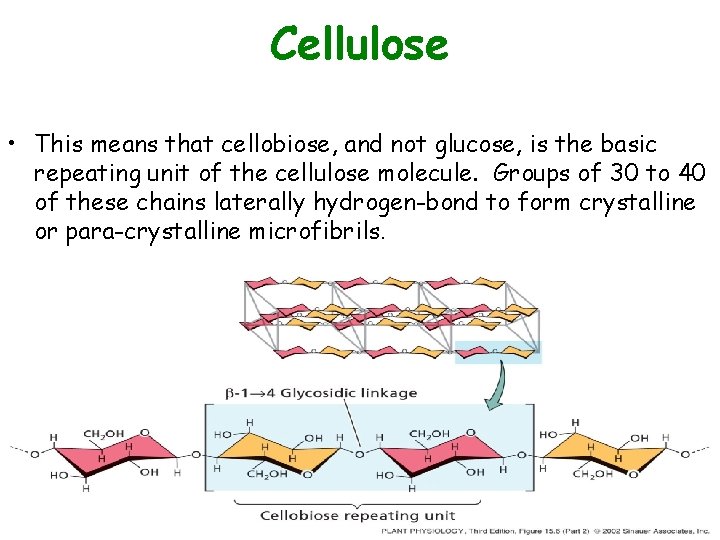
Cellulose • This means that cellobiose, and not glucose, is the basic repeating unit of the cellulose molecule. Groups of 30 to 40 of these chains laterally hydrogen-bond to form crystalline or para-crystalline microfibrils.

Lipids fats oils…. Greasy molecules, mmmmm donuts. Several levels of complexity: • Simple lipids - a lipid that cannot be broken down to smaller constituents by hydrolysis. – Fatty acids, waxes and cholesterol • Complex lipids - a lipid composed of different molecules held together mostly by ester linkages and susceptible to cleavage reactions. – acylglycerols - mono, di and triacyl glycerols ( fatty acids and glycerol) – phospholipids - lipids which are made of fatty acids, glycerol, a phosphoryl group and an alcohol. Many also contain nitrogen
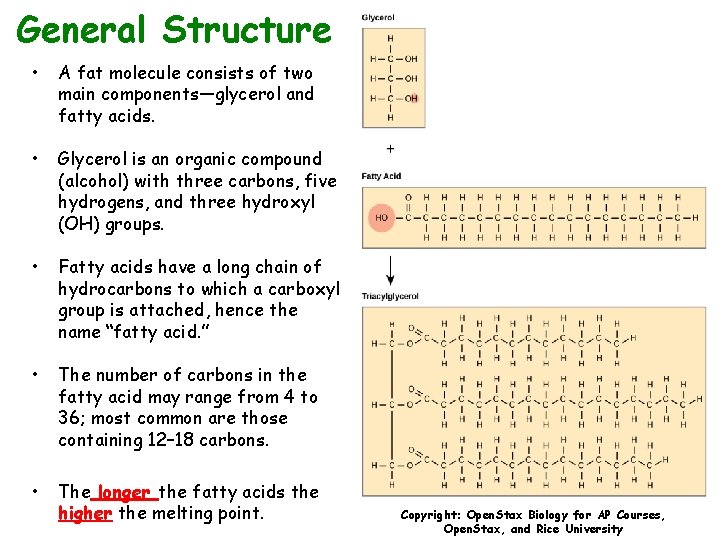
General Structure • A fat molecule consists of two main components—glycerol and fatty acids. • Glycerol is an organic compound (alcohol) with three carbons, five hydrogens, and three hydroxyl (OH) groups. • Fatty acids have a long chain of hydrocarbons to which a carboxyl group is attached, hence the name “fatty acid. ” • The number of carbons in the fatty acid may range from 4 to 36; most common are those containing 12– 18 carbons. • The longer the fatty acids the higher the melting point. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University

Saturated or not – the power of H • The terms saturated, monounsaturated, and poly-unsaturated refer to the number of hydrogens attached to the hydrocarbon tails of the fatty acids as compared to the number of double bonds between carbon atoms in the tail. • Oils, mostly from plant sources, have some double bonds between some of the carbons in the hydrocarbon tail. • Because some of the carbons share double bonds, they’re not bonded to as many hydrogens as they could if they weren’t double bonded to each other. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University
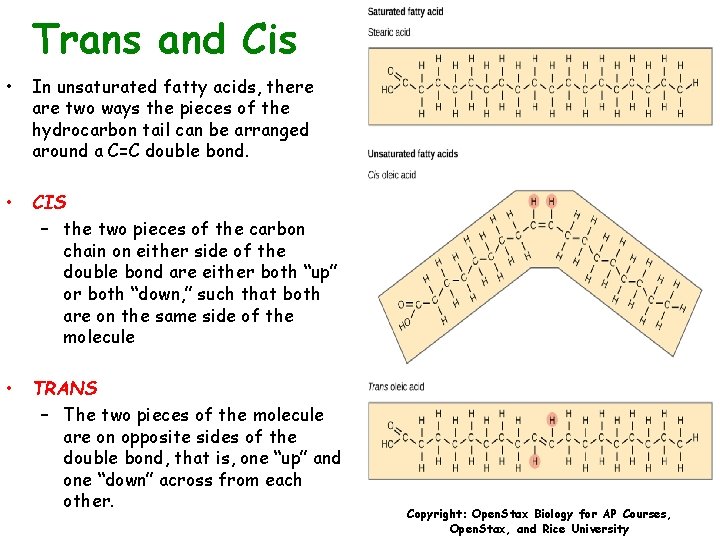
Trans and Cis • In unsaturated fatty acids, there are two ways the pieces of the hydrocarbon tail can be arranged around a C=C double bond. • CIS – the two pieces of the carbon chain on either side of the double bond are either both “up” or both “down, ” such that both are on the same side of the molecule • TRANS – The two pieces of the molecule are on opposite sides of the double bond, that is, one “up” and one “down” across from each other. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University
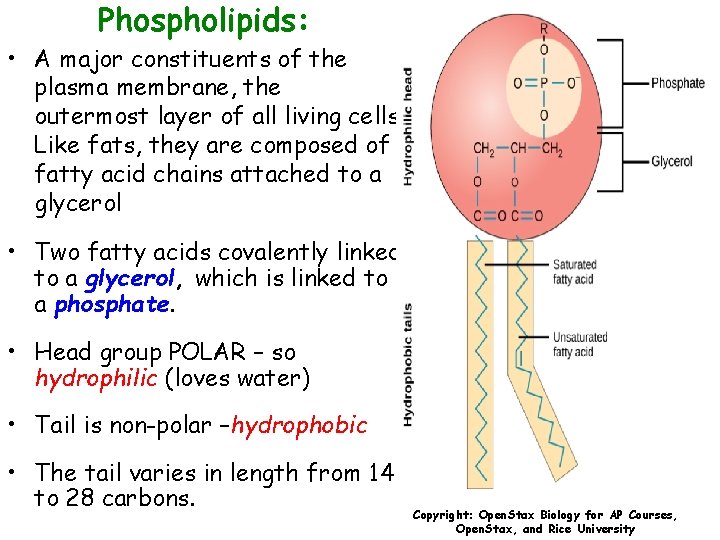
Phospholipids: • A major constituents of the plasma membrane, the outermost layer of all living cells. Like fats, they are composed of fatty acid chains attached to a glycerol • Two fatty acids covalently linked to a glycerol, which is linked to a phosphate. • Head group POLAR – so hydrophilic (loves water) • Tail is non-polar –hydrophobic • The tail varies in length from 14 to 28 carbons. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University
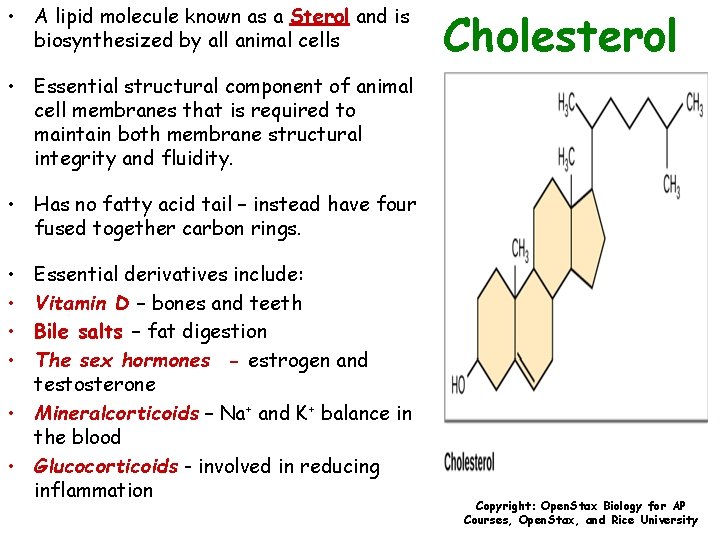
• A lipid molecule known as a Sterol and is biosynthesized by all animal cells Cholesterol • Essential structural component of animal cell membranes that is required to maintain both membrane structural integrity and fluidity. • Has no fatty acid tail – instead have four fused together carbon rings. • • Essential derivatives include: Vitamin D – bones and teeth Bile salts – fat digestion The sex hormones - estrogen and testosterone • Mineralcorticoids – Na+ and K+ balance in the blood • Glucocorticoids - involved in reducing inflammation Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University

Proteins: Peptides & Primary Structure Protein Functions: – Enzymes -Catalyze a thermodynamically favorable reaction – Storage/transport - binding proteins fatty acids w/albumin – no catalytic activity but do form chemical bonds with ligands – Structure - Several levels – cytoskeleton, collagen, bone. . . – Receptors – Growth factors – Antibodies
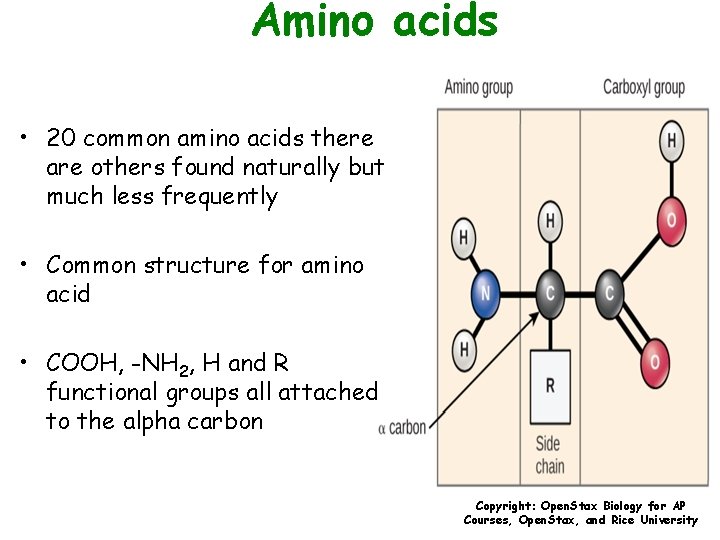
Amino acids • 20 common amino acids there are others found naturally but much less frequently • Common structure for amino acid • COOH, -NH 2, H and R functional groups all attached to the alpha carbon Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University
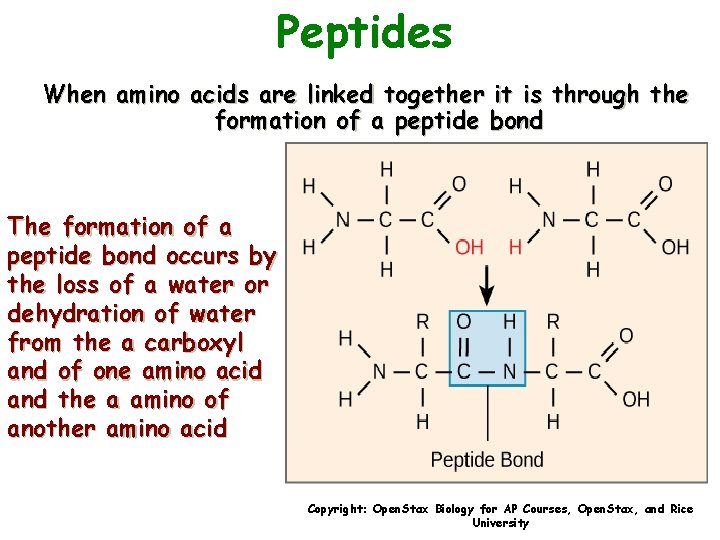
Peptides When amino acids are linked together it is through the formation of a peptide bond The formation of a peptide bond occurs by the loss of a water or dehydration of water from the a carboxyl and of one amino acid and the a amino of another amino acid Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University

Proteins: Three-dimensional structure • Background on protein composition: • Two general classes of proteins · Fibrous - long rod-shaped, insoluble proteins. These proteins are strong (high tensile strength). · Globular - compact spherical shaped proteins usually water-soluble. Most hydrophobic amino acids found in the interior away from the water. Nearly all enzymes are globular… · Proteins can be simple - no added groups or modifications, just amino acids • Or proteins can be conjugated. Additional groups covalently bound to the amino acids. The naked protein is called the apoprotein and the added group is the prosthetic group. Together the protein and prosthetic group is called the holoprotein. Ex. Hemoglobin
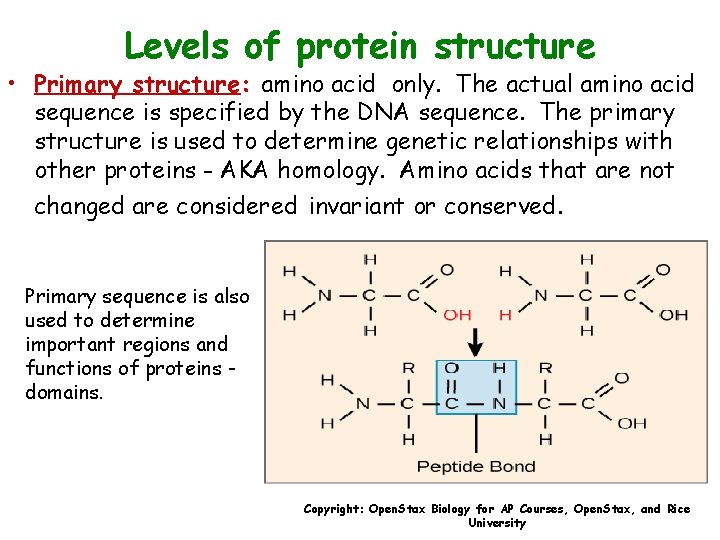
Levels of protein structure • Primary structure: amino acid only. The actual amino acid sequence is specified by the DNA sequence. The primary structure is used to determine genetic relationships with other proteins - AKA homology. Amino acids that are not changed are considered invariant or conserved. Primary sequence is also used to determine important regions and functions of proteins domains. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University
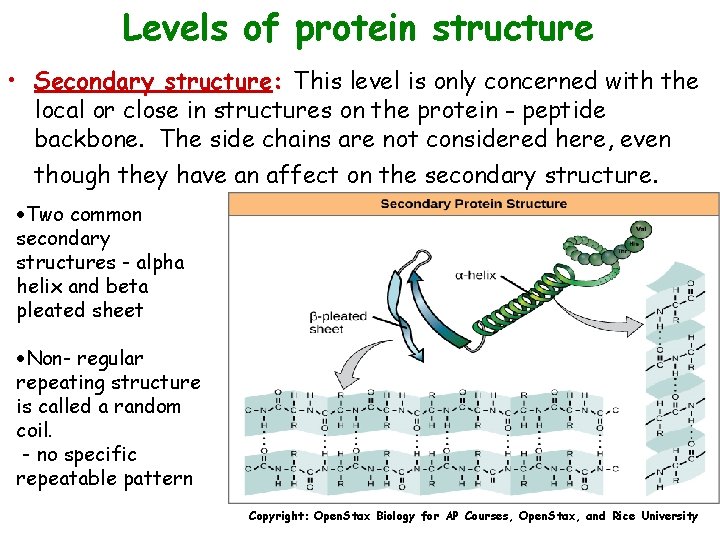
Levels of protein structure • Secondary structure: This level is only concerned with the local or close in structures on the protein - peptide backbone. The side chains are not considered here, even though they have an affect on the secondary structure. ·Two common secondary structures - alpha helix and beta pleated sheet ·Non- regular repeating structure is called a random coil. - no specific repeatable pattern Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University
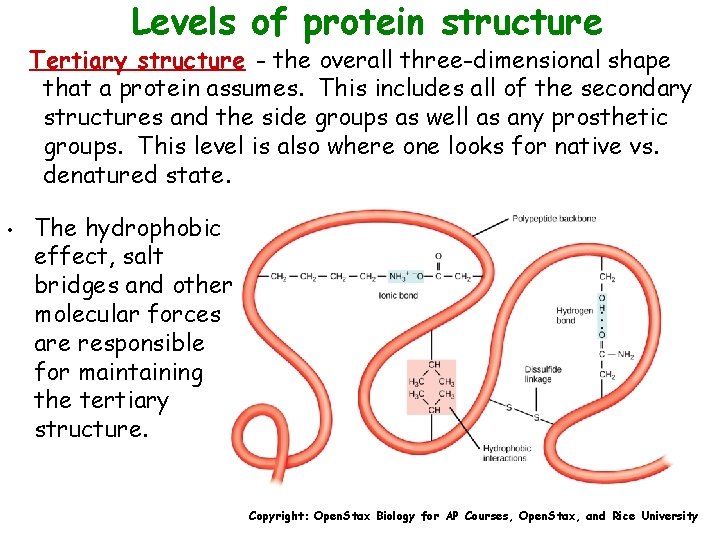
Levels of protein structure Tertiary structure - the overall three-dimensional shape that a protein assumes. This includes all of the secondary structures and the side groups as well as any prosthetic groups. This level is also where one looks for native vs. denatured state. • The hydrophobic effect, salt bridges and other molecular forces are responsible for maintaining the tertiary structure. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University
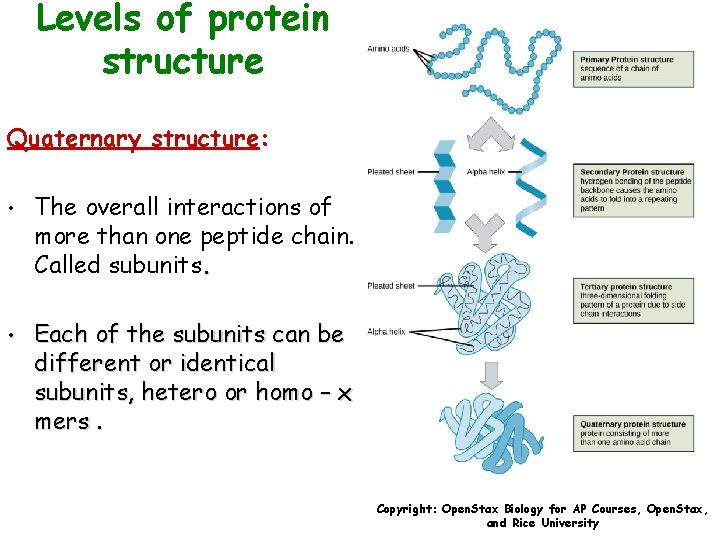
Levels of protein structure Quaternary structure: • The overall interactions of more than one peptide chain. Called subunits. • Each of the subunits can be different or identical subunits, hetero or homo – x mers. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University

Glycoproteins • Proteins that contain oligosaccharide chains covalently attached to polypeptide side-chains. • The carbohydrate is attached to the protein in a cotranslational or posttranslational modification. This process is known as glycosylation. – In proteins that have segments extending extracellularly, the extracellular segments are often glycosylated. • Glycoproteins are often important integral membrane proteins, where they play a role in cell-cell interactions. • Glycoproteins also occur in the cytosol, but their functions and the pathways producing these modifications in this compartment are less well-understood

Examples of glycoproteins found in the human body • Mucins • secreted in the mucus of the respiratory and digestive tracts. • The sugars attached to mucins give them considerable water-holding capacity and also make them resistant to proteolysis by digestive enzymes. • In the immune system • white blood cell recognition molecules such as antibodies which interact directly with antigens • Molecules of the major histocompatibility complex (or MHC), which are expressed on the surface of cells and interact with T cells as part of the adaptive immune response. From the wikimedia free licensed media file repository
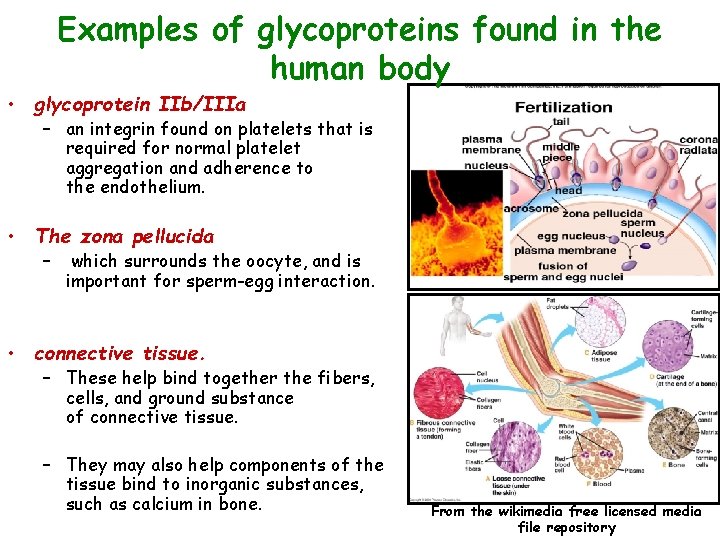
Examples of glycoproteins found in the human body • glycoprotein IIb/IIIa – an integrin found on platelets that is required for normal platelet aggregation and adherence to the endothelium. • The zona pellucida – which surrounds the oocyte, and is important for sperm-egg interaction. • connective tissue. – These help bind together the fibers, cells, and ground substance of connective tissue. – They may also help components of the tissue bind to inorganic substances, such as calcium in bone. From the wikimedia free licensed media file repository
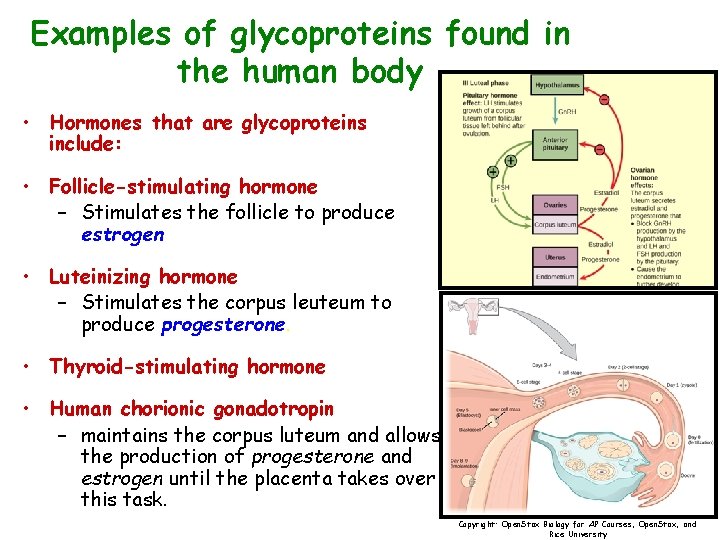
Examples of glycoproteins found in the human body • Hormones that are glycoproteins include: • Follicle-stimulating hormone – Stimulates the follicle to produce estrogen • Luteinizing hormone – Stimulates the corpus leuteum to produce progesterone. • Thyroid-stimulating hormone • Human chorionic gonadotropin – maintains the corpus luteum and allows the production of progesterone and estrogen until the placenta takes over this task. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University
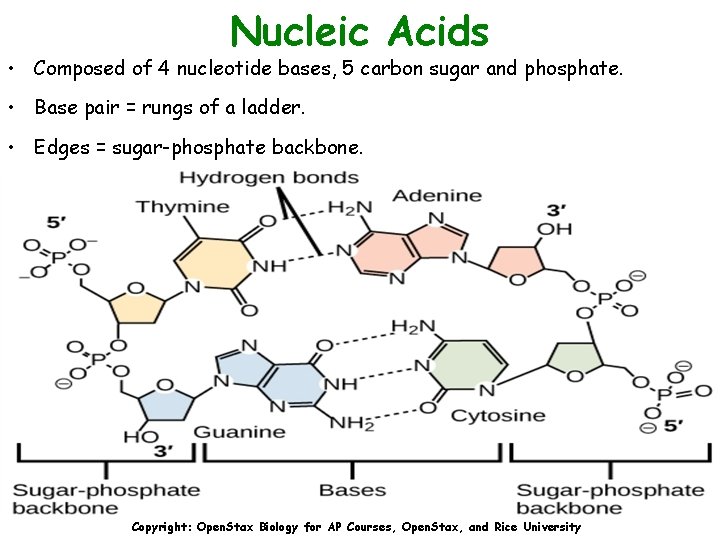
Nucleic Acids • Composed of 4 nucleotide bases, 5 carbon sugar and phosphate. • Base pair = rungs of a ladder. • Edges = sugar-phosphate backbone. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University
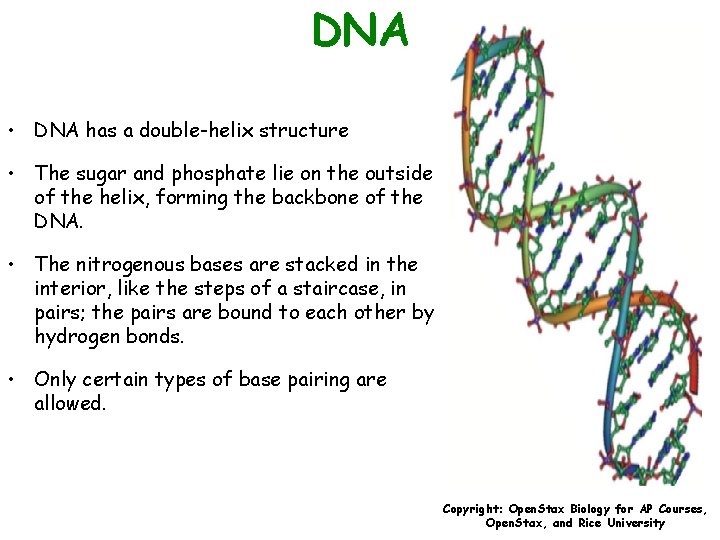
DNA • DNA has a double-helix structure • The sugar and phosphate lie on the outside of the helix, forming the backbone of the DNA. • The nitrogenous bases are stacked in the interior, like the steps of a staircase, in pairs; the pairs are bound to each other by hydrogen bonds. • Only certain types of base pairing are allowed. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University

DNA • Only certain types of base pairing are allowed. • This means A can pair with T, and G can pair with C – known as the base complementary rule. • If the sequence of one strand is AATTGGCC, the complementary strand would have the sequence TTAACCGG. • During DNA replication, each strand is copied, resulting in a daughter DNA double helix containing one parental DNA strand a newly synthesized strand. Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University
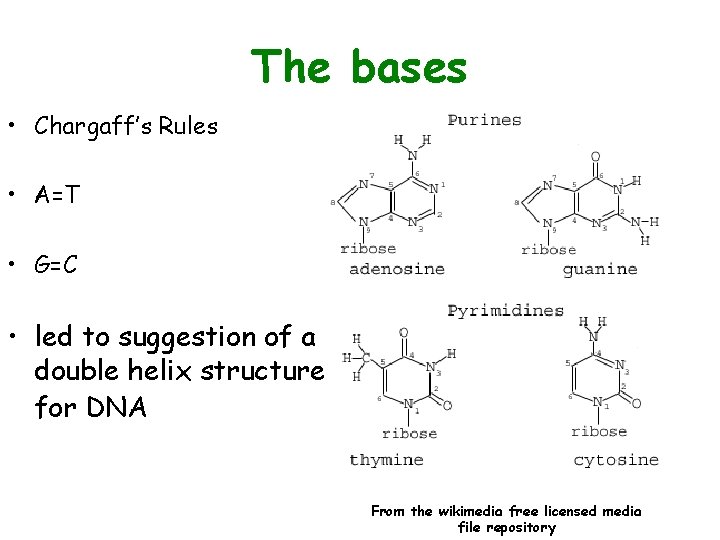
The bases • Chargaff’s Rules • A=T • G=C • led to suggestion of a double helix structure for DNA From the wikimedia free licensed media file repository
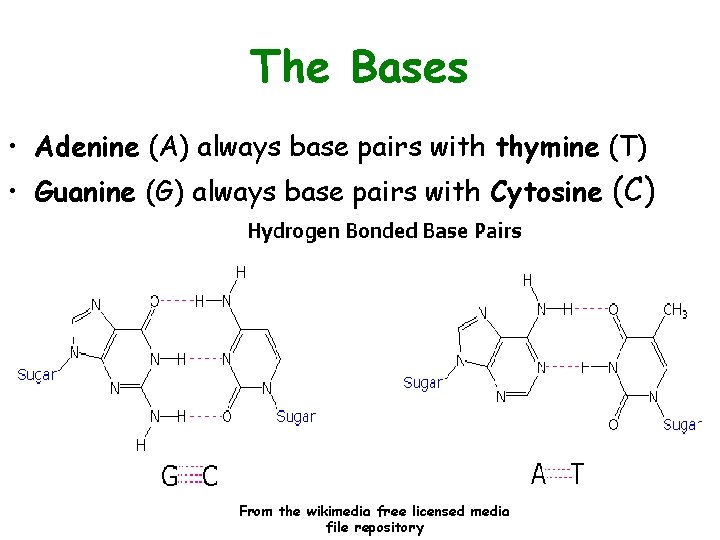
The Bases • Adenine (A) always base pairs with thymine (T) • Guanine (G) always base pairs with Cytosine (C) From the wikimedia free licensed media file repository
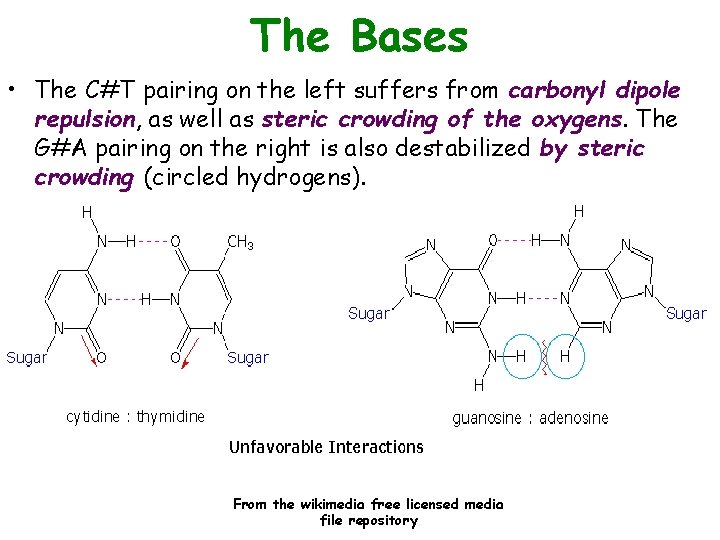
The Bases • The C#T pairing on the left suffers from carbonyl dipole repulsion, as well as steric crowding of the oxygens. The G#A pairing on the right is also destabilized by steric crowding (circled hydrogens). From the wikimedia free licensed media file repository
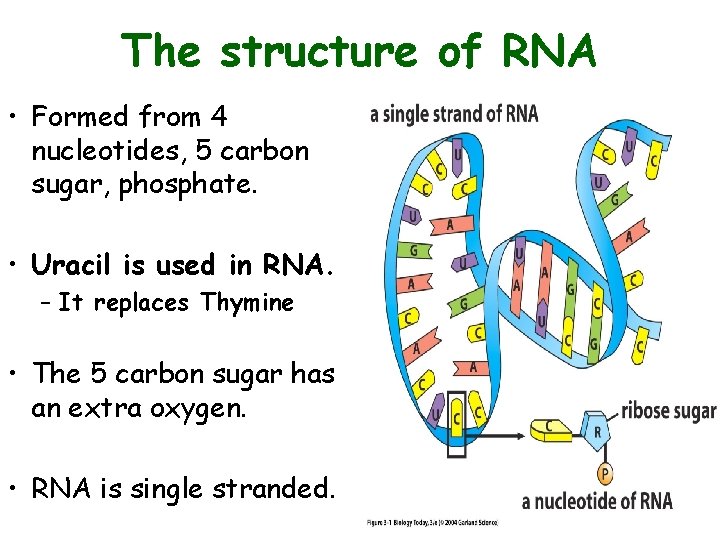
The structure of RNA • Formed from 4 nucleotides, 5 carbon sugar, phosphate. • Uracil is used in RNA. – It replaces Thymine • The 5 carbon sugar has an extra oxygen. • RNA is single stranded.
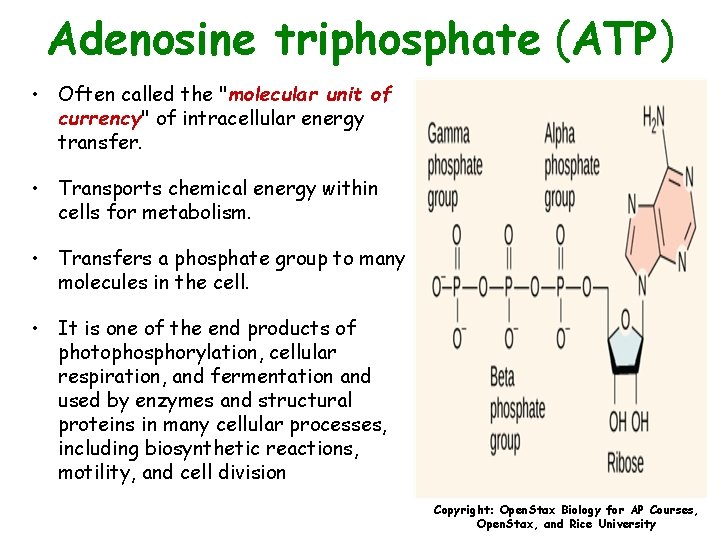
Adenosine triphosphate (ATP) • Often called the "molecular unit of currency" of intracellular energy transfer. • Transports chemical energy within cells for metabolism. • Transfers a phosphate group to many molecules in the cell. • It is one of the end products of photophosphorylation, cellular respiration, and fermentation and used by enzymes and structural proteins in many cellular processes, including biosynthetic reactions, motility, and cell division Copyright: Open. Stax Biology for AP Courses, Open. Stax, and Rice University

The End! Any Questions?
- Slides: 62