HUMAN ANATOMY PHYSIOLOGY CHAPTER 2 BASIC CHEMISTRY Concepts

HUMAN ANATOMY & PHYSIOLOGY CHAPTER 2 -BASIC CHEMISTRY

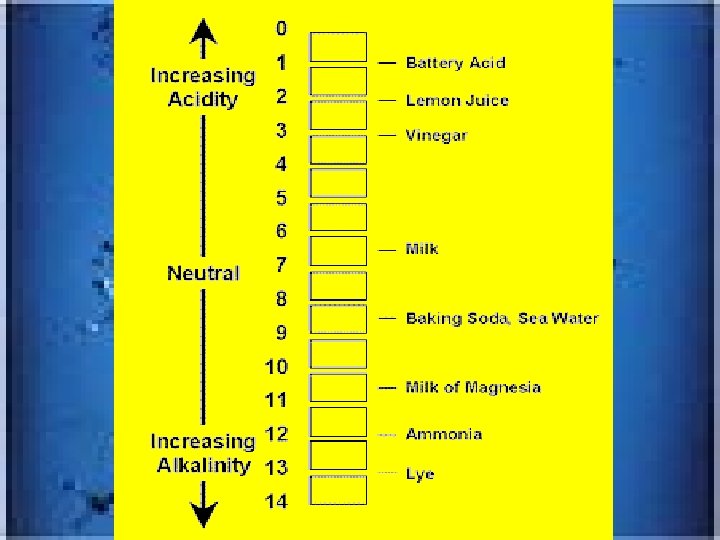
Concepts of Matter & Energy • Matter=”stuff” of the universe existing as __________states on earth/solid has definite volume and shape; liquid only definite volume and gas-has neither • Physical change-does not alter basic nature whereas chemical alters _____________. • ENERGYonly measured by effects on matter________ • ______energy of motion and potential is _____. kinetic Solid liquid and gas composition Ability to do work Stored energy

Energy forms: • _______-stored in chemical bondsinvolved in cellular respiration – _______-comes from movement of charged particles-used in nerve impulses – _____-Directly involved in moving matter-eg. movement of limbs – _______-travels in waves; electromagnetic spectrum-x-rays, infrared, visible light, radio, UV, gamma Chemical energy radiant electrical Mechanical energy
• Energy is usually converted from one form to another-some of initial energy is lost to environment-This is how we maintain body temperature

II. Composition of Matter • 112_______(#s 113 -118 alleged)92 occur in nature- elements are alike in atoms and unique atomic #/represented on periodic table , organized by atomic # and short hand representation is _________-Look at Chart p. 30 • Atoms made of positive protons and neutral ______in nucleus/both heavier w/ mass of 1 AMU, , surrounded by _______electrons(mass =minute fraction of an amu ), in energy levels elements symbol neutrons (-)
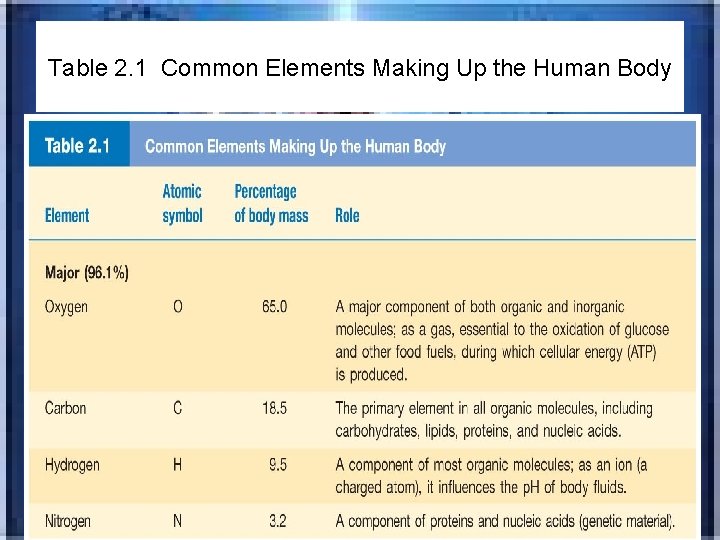
Table 2. 1 Common Elements Making Up the Human Body

Table 2. 1 Common Elements Making Up the Human Body
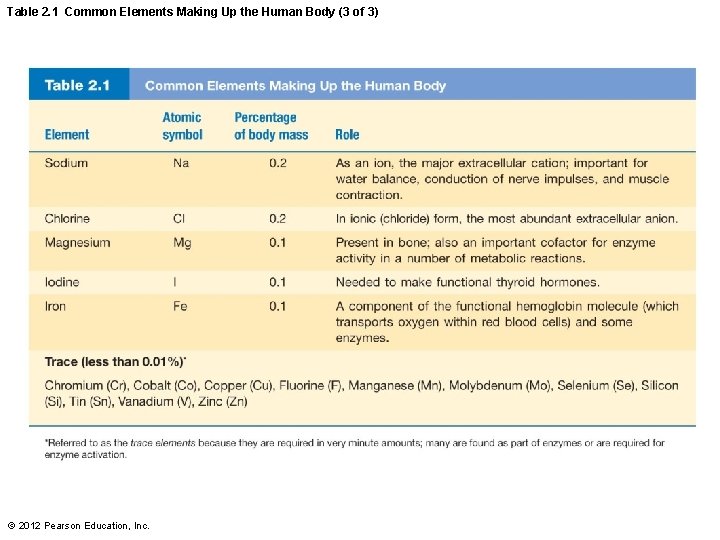
Table 2. 1 Common Elements Making Up the Human Body (3 of 3) © 2012 Pearson Education, Inc.

• Atomic # = # protons • Atomic mass #= # p+n • Atomic mass –average of all isotope masses for an element

• p’s and e-‘s are = to make a neutral atom/losing electrons creates a + charge and gaining e’s creates a (-) charge/+ and- attract , while likes repel • _______-e’s are arranged in energy levels w/in an electron cloud • Orbital model
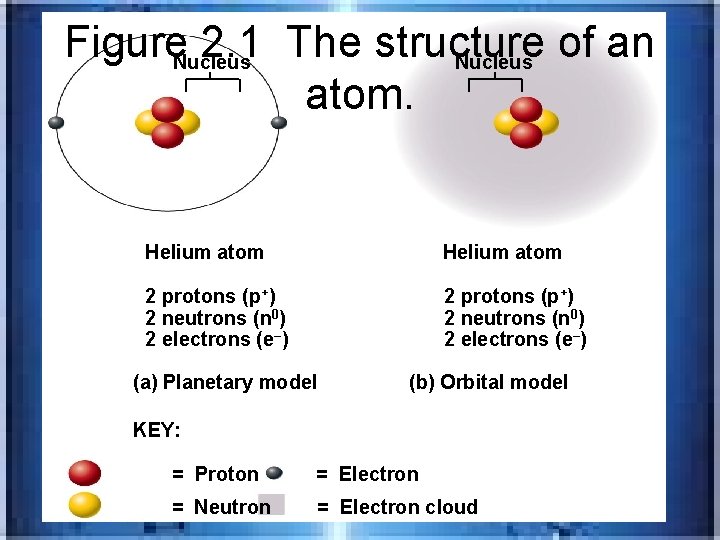
Figure 2. 1 The structure of an Nucleus atom. Helium atom 2 protons (p+) 2 neutrons (n 0) 2 electrons (e–) (a) Planetary model (b) Orbital model KEY: = Proton = Electron = Neutron = Electron cloud

• _______________are atoms that differ in number of neutrons, are identified my mass number, and one medical use is as tracers in medical diagnosis. ---see. pp 10 -11 • ___________=spontaneous atomic decay(alpha, beta, and the more intense gamma) • _________________occur whenever atoms combine with or dissociate from other atoms • ___________-2 or more combined chemically; more specifically a compound radioisotopes molecule Chemical reactions radiation
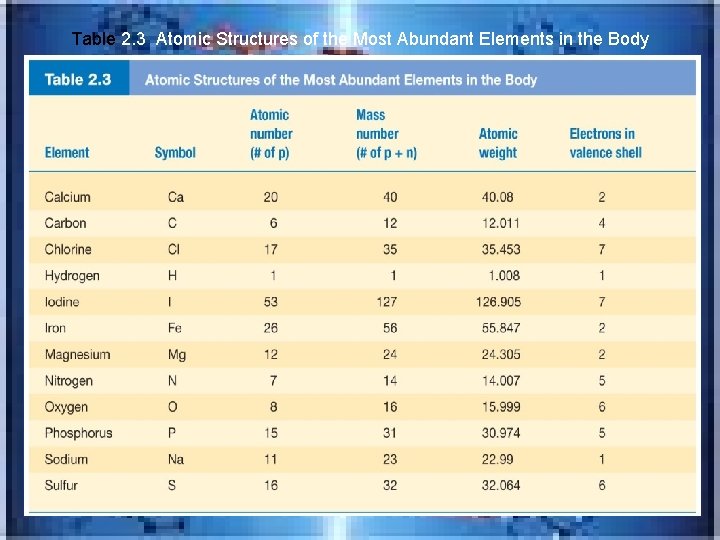
Table 2. 3 Atomic Structures of the Most Abundant Elements in the Body

• 2 bond we see in living things are _________, w/a transfer of electrons and ______________w/a sharing of electrons-this transfer or sharing is occurring with the electron shells(ie. Energy levels) • Outer -___________electrons determine bonding capacity and thus properties--These electrons have more energy , away from the + nucleus • _______-charged atom ionic valence ion covalent

• Polar molecules have unequal sharing of electrons , as seen in water • Water bonds to other water atoms by H- bonds-a weak bond/polar molecules orient themselves towards other polar molecules • These combined H-bonds create a high surface tension

Figure 2. 9 b Hydrogen bonding between polar water molecules.

• • 3 reaction types synthesis ----2 H 2 + O 2 2 H 2 O _____------H 2 O 2 H 2 +O 2 Exchange---2 HCl + Zn Zn. Cl 2 +H 2 OR HCl + Na. OH HOH +Na. Cl • Most chemical reactions are reversible, indicated by a double arrow • A+B AB • decomposition
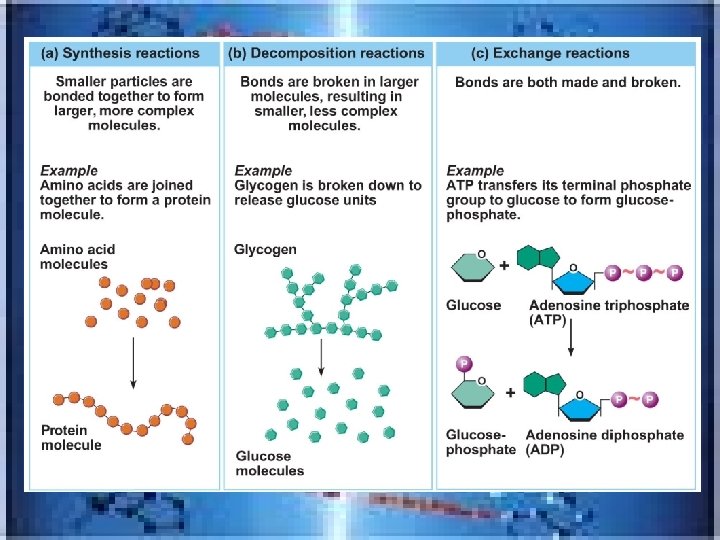


III. Biochemistry: Chemical Composition of Living Matter • _________ contain carbon , but not all are involved in living things(inorganic lack C-----CO, CO 2 and cyanide 3 exemptions-they are inorganic Organic compounds

INORGANICS--_WATER-most abundant compound in the body • high heat capacity(ie. before a temperature change absorbs and releases a lot of heat change thus preventing us from sudden body temperature changes. • ______________-“universal solvents” • Solute dissolves in a _________ to make a solution-Thus water able to transport many materials • 3. chemical reactivity • _____________reaction---water added to the bond. • 4. ______________-protective as in CSF or amniotic fluid solvent Polarity/solvent properties cushioning hydrolysis
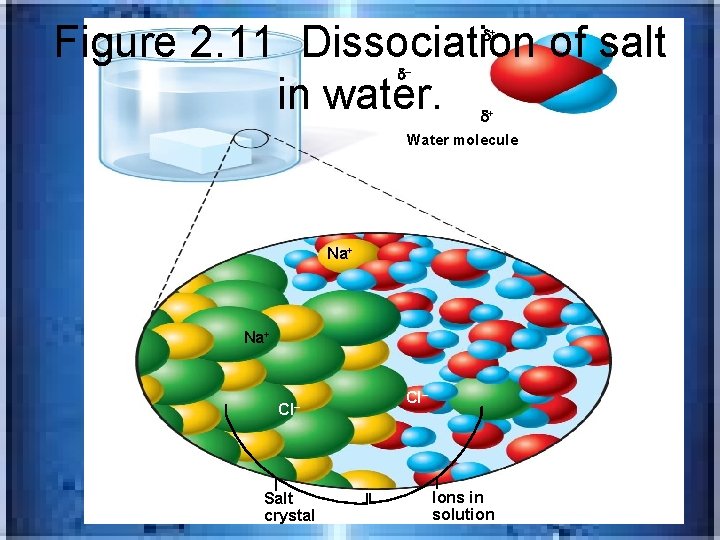
Figure 2. 11 Dissociation of salt in water. H – + O H + Water molecule Na+ Cl– Salt crystal Cl– Ions in solution

INORGANIC COMPOUNDSSALTS • Salt contains cation besides H+ and an anion besides OH-…examples: Na. Cl, KCl, Ca. I 2 • Easily separate into ions____________ • Because ions are charged particles, all salts are ____________-able to conduct a current dissociation electrolytes
• _______-p. H<7 and proton donor • _______-p. H> 7 and proton acceptor • p. H measures H+ concentration • Acid +Base=Salt + water –all neutral NEUTRALIZATION REACTION base acid
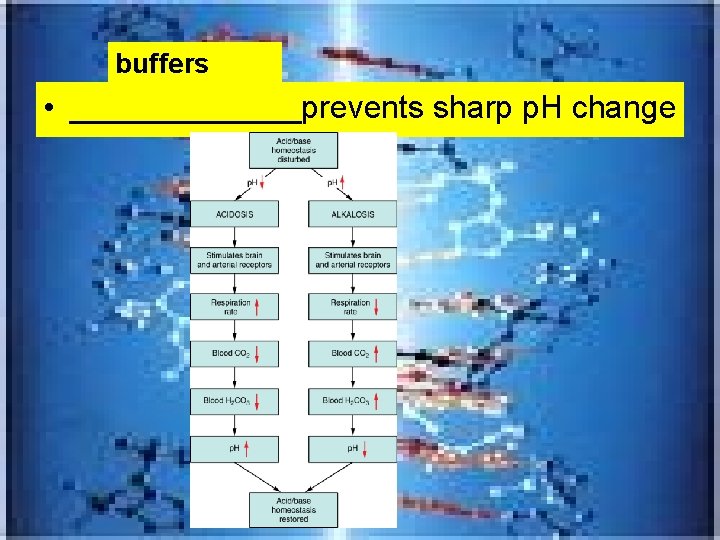
buffers • _______prevents sharp p. H change

FOUR CLASSES of MACROMOLECULES in living things : • _______=chainlike molecules made of many similar repeating units called_________. These are joined by dehydration synthesis-Here a H-atom is removed from one monomer and a hydroxyl group(OH) is removed from the monomer it is being joined with…a water molecule is released • When a polymer needs to be broken down/digested the reverse happens-_______occurs-water molecule added to each bond, releasing monomers polymers monomers hydrolysis

• • • 1)Carbohydrates(this means”hydrated C”)-FIRST SOURCE FOR ENERGY!!!!!inc. sugars, starches/have C, H and O in a 1: 2: 1 ratio-(w/some exceptions) Example: C 6 H 12 O 6= GLUCOSE-(blood sugar) Sugars inc. monosaccharide(Glucose, fructose, galactose , ribose and deoxyiribose), disaccharidedouble sugars-2 monosaccharides joined by dehydration(Sucrose, lactose, maltose)---must be broken down to monosaccharides to be digested) and polysaccharides-long branching chains (starch and glycogen )---ideal for storage---/but lack the sweetness of smaller sugars(starch and glycogen)…. Examples of starches_______________ Pasta, rice, bread, carrots
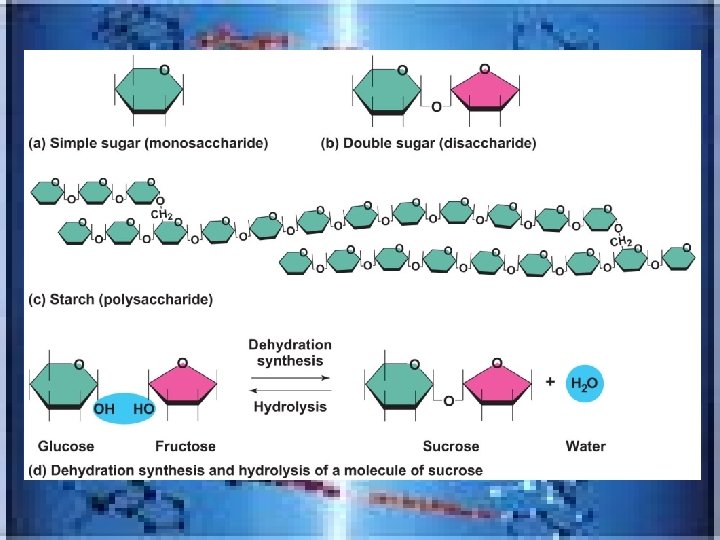

Figure 2. 14 c Carbohydrates.

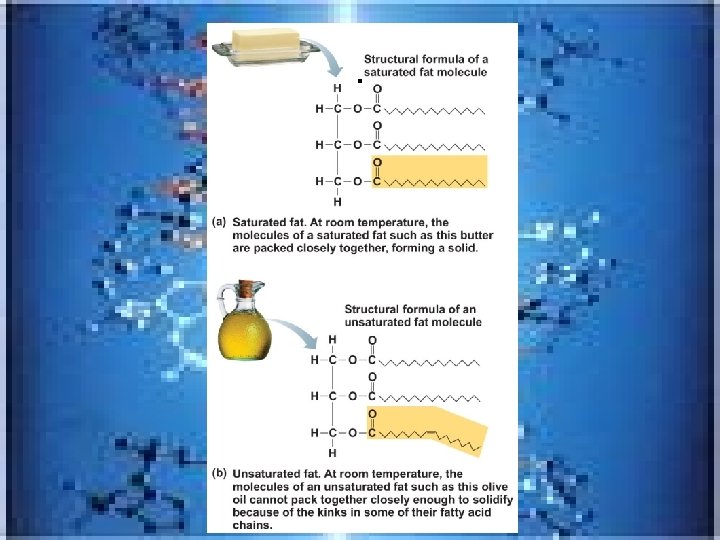
Lipids-made of C, H and O---look at lipid table p. 46 Enter body as fat –marbled meats, egg yolks, milk products, and oils • • ---made up of often fatty acids and glycerol _____________-are solid fats with all C-C bonds filled-no double or triple bonds…. ___________-are liquids and they have at least 1 C-C double bond-either monoor polyunsaturated Saturated fats unsaturated

lipids

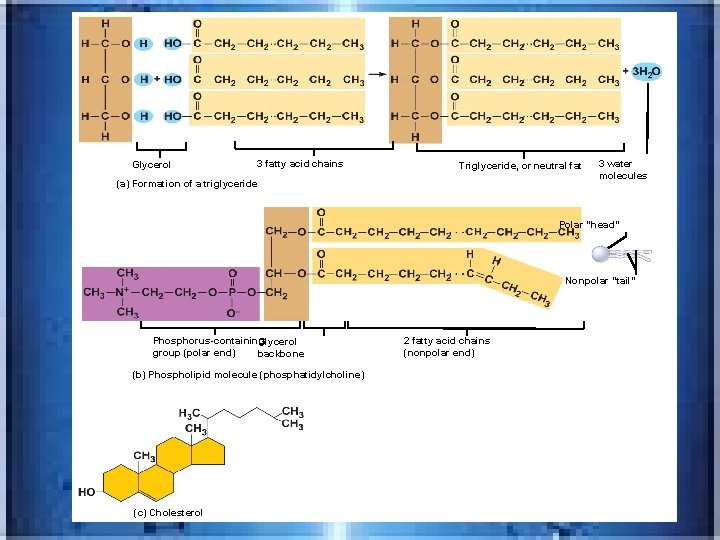
Glycerol 3 fatty acid chains Triglyceride, or neutral fat (a) Formation of a triglyceride 3 water molecules Polar “head” Nonpolar “tail” Phosphorus-containing Glycerol group (polar end) backbone (b) Phospholipid molecule (phosphatidylcholine) (c) Cholesterol 2 fatty acid chains (nonpolar end)
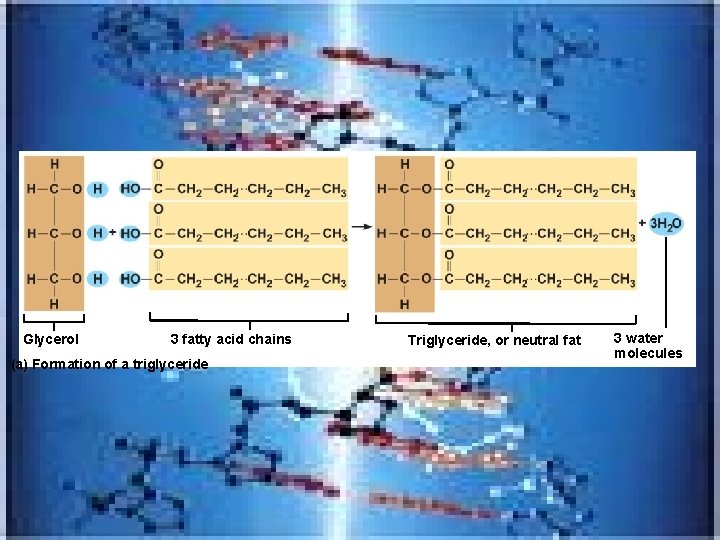
Glycerol 3 fatty acid chains (a) Formation of a triglyceride Triglyceride, or neutral fat 3 water molecules

• Neutral fats_triglycerides • made of fatty acid and glycerol( 3 fatty acids attached to 1 glycerol) • in fat deposits-in subcutaneous tissue and around organs-protect, insulate and major energy source • Trans fats---(common in margarines and baked products) -oils solidifies by adding H atoms---considered bad for the cardiovascular system • Omega -3 fatty acids(in cold water fish)-help your heart and immune system • Phospholipids found in cell membrane

• _______fat soluble-inc. cholesterol and hormones, such as sex hormones-cholesterol basis of all body steroids • Cholesterol is the single most important steroid-from animal products and made by the liver---found in cell membranes , raw material of vitamin D, steroid hormones, and bile salts • _______a breakdown product of cholesterol; released by liver into digestive tract-aid in fat digestion and absorption steroids Bile salts
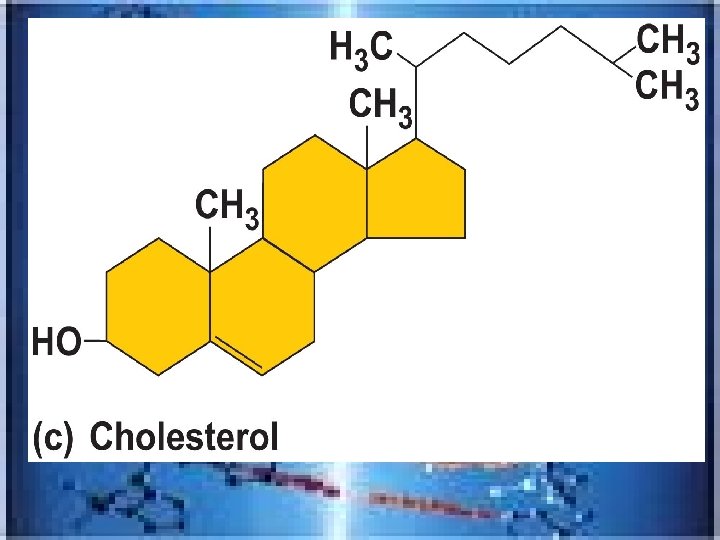

• __________-cortisol; aldersterone • LIPIDS ALSO Includes vitamins A(for vision)E(wound healing, fertility, antioxidant), K(for clotting, Prostaglandins-from fatty acidmembranes(used in labor, blood pressure, movement in digestive tract and in inflammation), lipoproteins(transport fatty acids and cholesterol in bloodstream-HDL and LDL) corticosteroids

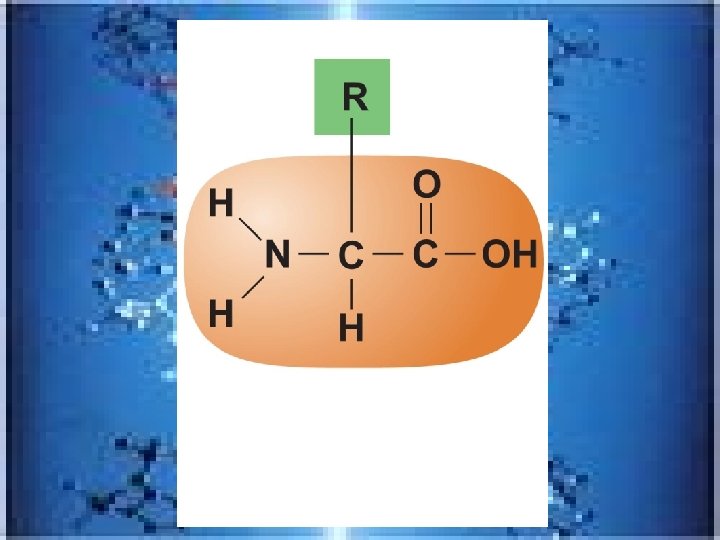
PROTEINS • made up of monomer of amino acids; >50% organic matter in the body, contain C, H, O, and N, ~ 20 amino acids, polypeptide is another word for protein (containing fewer than 50 amino acids )and peptide bonds join amino acids-made of an amine (NH 2) grp and an acid(COOH) grp, differ in R-grp – Fibrous(or structural) strandlike /Proteins are in most body structures; provide strengtheg. collagen in bones, cartilage and tendons…. . also KERATIN—protein of hair , nails, &skin-providing toughness – Globular proteins-mobile and mostly spherical and DO things---FUNCTIONAL PROTEINS---examples: enzymes-regulate reactions, antibodies---H-bonds and van der waals force help them-like hemoglogin, keep their shape

Figure 2. 17 Amino acid structures. Amine group Acid group (a) Generalized structure of all amino acids (b) Glycine (the simplest amino acid) (c) Aspartic acid (an acidic amino acid) (d) Lysine (a basic amino acid (e) Cysteine (a sulfurcontaining amino acid)
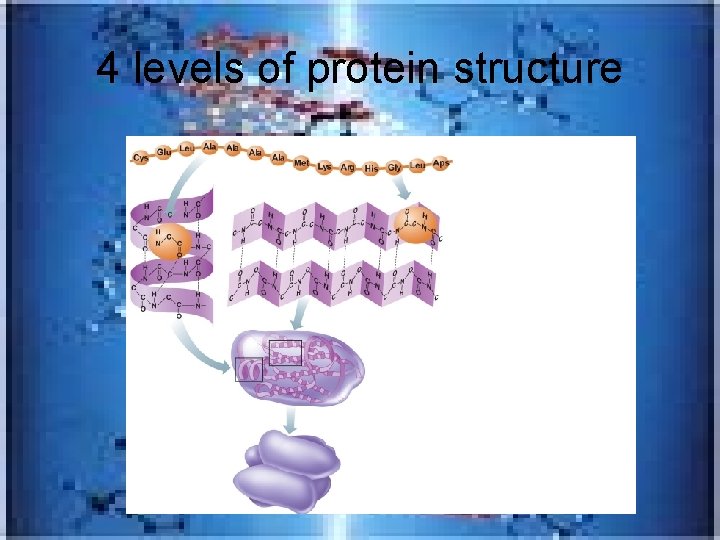
4 levels of protein structure
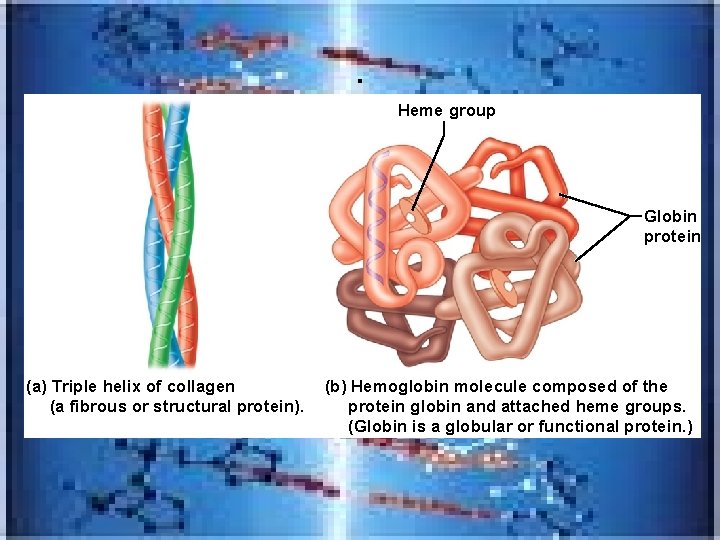
. Heme group Globin protein (a) Triple helix of collagen (a fibrous or structural protein). (b) Hemoglobin molecule composed of the protein globin and attached heme groups. (Globin is a globular or functional protein. )


– less stable than fibrous structures/If that structure is compromised by heat or extremes of p. H, they are_________-no longer perform their physiological functions. (because their structure dictates their function) denatured

– Draw an enzyme (biological catalyst) – substrate complex and label active site – increases reaction rate/controls most physiological reactions
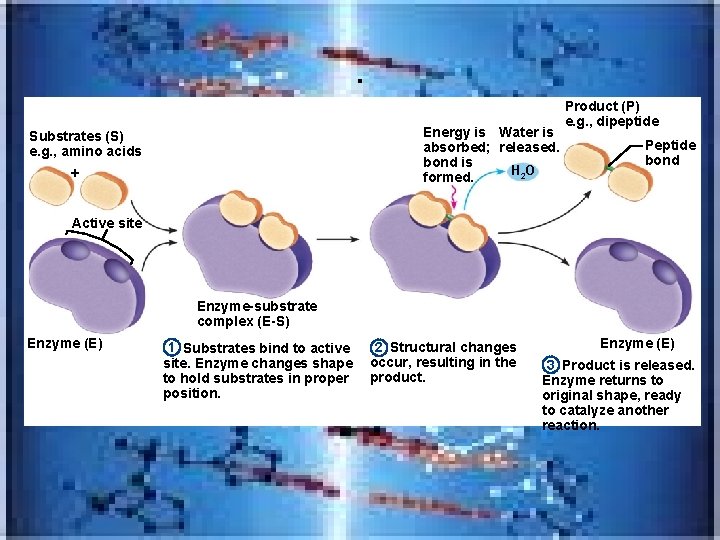
. Energy is Water is absorbed; released. bond is H 2 O formed. Substrates (S) e. g. , amino acids + Product (P) e. g. , dipeptide Peptide bond Active site Enzyme-substrate complex (E-S) Enzyme (E) 1 Substrates bind to active site. Enzyme changes shape to hold substrates in proper position. 2 Structural changes occur, resulting in the product. Enzyme (E) 3 Product is released. Enzyme returns to original shape, ready to catalyze another reaction.

– Enzymes include_________that add water , OXIDASES-add O 2, etc – Most enzymes are produced in inactive form and must be activated/some inactivated immediately after they catalyze reaction—example in blood clotting process hydrolases
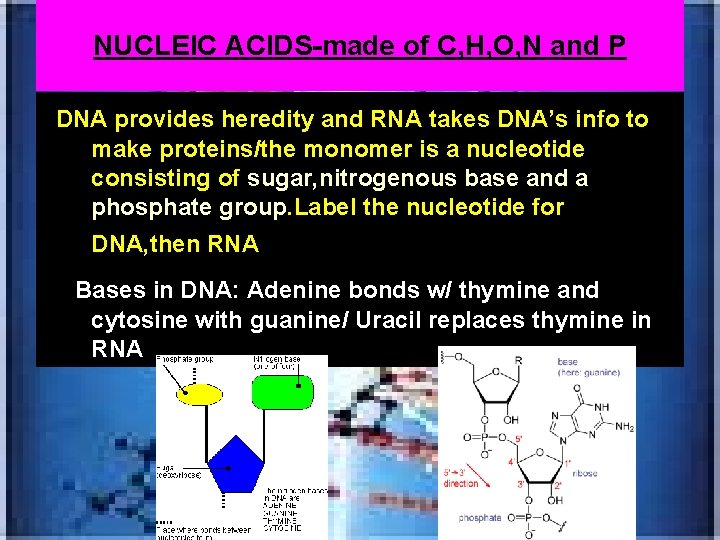
NUCLEIC ACIDS-made of C, H, O, N and P *DNA provides heredity and RNA takes DNA’s info to make proteins/the monomer is a nucleotide consisting of sugar, nitrogenous base and a phosphate group. Label the nucleotide for DNA, then RNA: ---Bases in DNA: Adenine bonds w/ thymine and cytosine with guanine/ Uracil replaces thymine in RNA
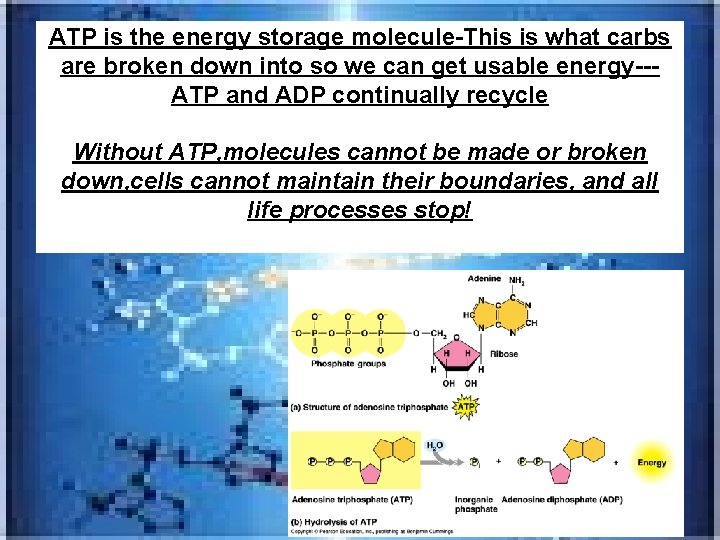
ATP is the energy storage molecule-This is what carbs are broken down into so we can get usable energy--ATP and ADP continually recycle Without ATP, molecules cannot be made or broken down, cells cannot maintain their boundaries, and all life processes stop!

Adenine High energy bonds Ribose Phosphates (a) Adenosine triphosphate (ATP) Adenosine diphosphate (ADP) (b) Hydrolysis of ATP
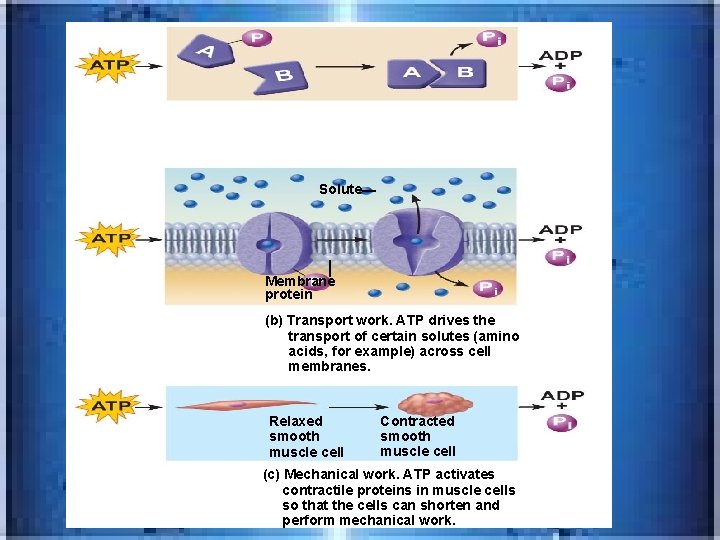
Solute Membrane protein (b) Transport work. ATP drives the transport of certain solutes (amino acids, for example) across cell membranes. Relaxed smooth muscle cell Contracted smooth muscle cell (c) Mechanical work. ATP activates contractile proteins in muscle cells so that the cells can shorten and perform mechanical work.
- Slides: 57