http discover edventures comimagestermlibiisotopesupport gif http www unit
http: //discover. edventures. com/images/termlib/i/isotope/support. gif

http: //www. unit 5. org/christjs/site%20 map. htm
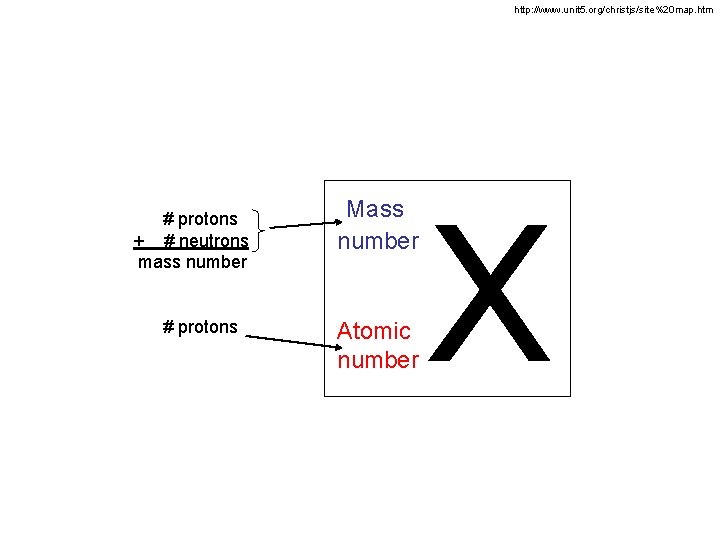
http: //www. unit 5. org/christjs/site%20 map. htm # protons + # neutrons mass number # protons Mass number Atomic number X

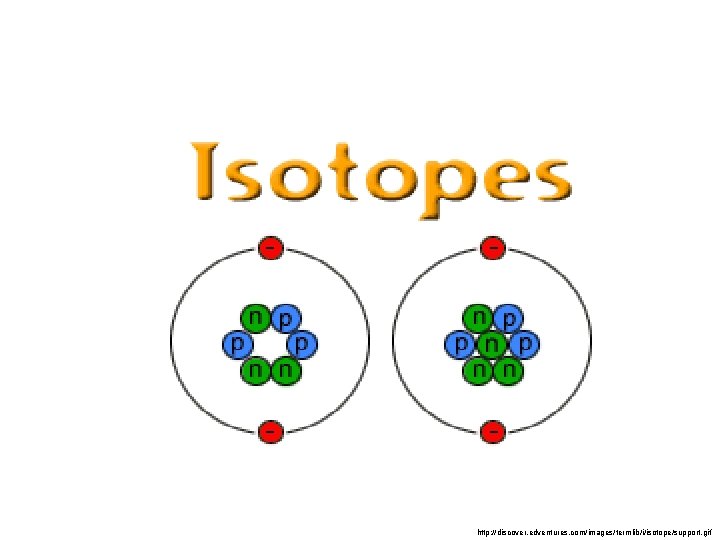
Isotopes • Atoms of the same element can have different numbers of neutrons ØThis gives them different mass numbers. ØThey have the same atomic numbers. ØThey look, act and react the same. C-12 vs. C-14


Naming Isotopes • Put the mass number after the name of the element • Examples: • Carbon-12 and Carbon-14 • Uranium-235 and Uranium-238


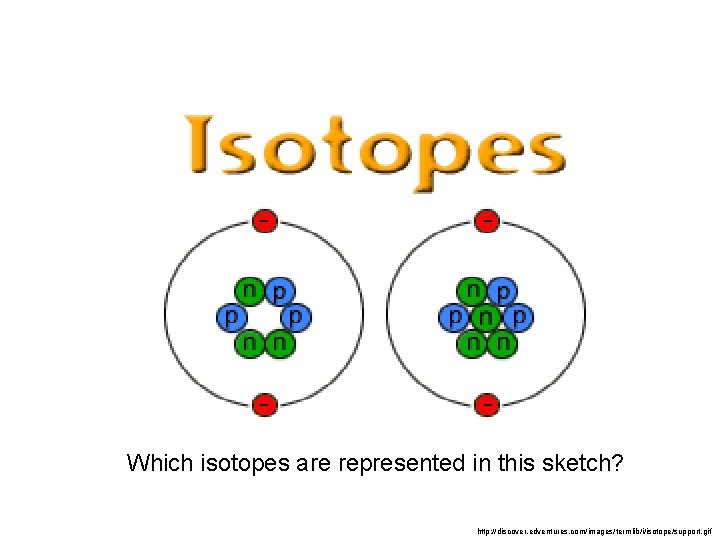
Which isotopes are represented in this sketch? http: //discover. edventures. com/images/termlib/i/isotope/support. gif

Calculations of Average Atomic Mass Why is the mass number on a periodic table never a nice whole number?

Mass Number vs Average. Atomic Mass • The Mass Number is specific to one isotope. • The Average Atomic Mass is the calculated mass based on the abundance of each naturally occurring isotope of that element. • The Average Atomic Mass is shown on Periodic Tables. It is the most accurate number that should be used in calculations.

Average Atomic Mass • How heavy is an atom of oxygen? • There are different kinds of oxygen atoms. üoxygen-16, üoxygen-17 and üoxygen-18 • We are more concerned with average atomic mass, based on the abundance of each element in nature. • We don’t use grams because the numbers would be too small. California WEB http: //www. unit 5. org/christjs/site%20 map. htm

http: //www. unit 5. org/christjs/site%20 map. htm Atomic Mass Unit (amu or u) • This unit is used instead of grams. • It is 1/12 of the mass of a carbon-12 atom.

Your book shows this a different way • Experts show that chlorine is a mixture of 75. 77% Cl-35 and 24. 33% of Cl-37. If the precise molar mass of Cl-35 is 34. 968 852 g and Cl – 37 is 36. 965 903 grams what is the average molar mass of the chlorine atoms in the mixture?

• Assume 100 moles of atoms are used in mixture • Calculate the number of moles of each isotope • Calculate the mass of each isotope • Add the masses together to get the total mass of the mixture

• # moles of Cl-35 = 0. 7577 x 100 mol = 75. 77 mol of Cl-35 – 75. 77 mol X 34. 968 852 g/mol = 26. 4959 g of Cl-35 • # moles of Cl-37 = 0. 2423 x 100 mol = 24. 23 mol – 24. 23 mol X 36. 965 903 g/mol = 8. 9568 g of Cl-37 Total mass of mixture = mass of Cl-35 + Cl-37 = 35. 453 g
- Slides: 16