HTA Bodies Cooperation on Early Dialogues Recent Advances

HTA Bodies Cooperation on Early Dialogues: Recent Advances François Meyer MD HAS – EUnet. HTA WP 5 Lead Partner ECRD 2018 European network for Health Technology Assessment | JA 3 2016 -2020 | www. eunethta. eu

Early Dialogue / Scientific advice • Definition and aim ‒ Advice given to a developer on the appropriate studies in the development of a product (medicine, device…). ‒ Advice = answers given to questions raised by the developer ‒ Aim = facilitating the availability of the most appropriate data to properly evaluate a drug for Marketing Authorisation and for HTA pricing an reimbursement • Main characteristics ‒ Voluntary process for companies ‒ Can be done at national or European level, with regulatory body or HTA bodies or both in parallel ‒ May be subject to fees or not ‒ Advice given is confidential and non binding European network for Health Technology Assessment | JA 3 2016 -2020 | www. eunethta. eu 2

EUnet. HTA European network for Health Technology Assessement ˗ Voluntary participation, institutions named by Mo. H in all participating countries ˗ 60% of costs covered by Europe, 40% by participating institutions ˗ Three/Four-year project ˗ EUnet. HTA is not a legal entity and has no permanent staff ˗ Some dedicated staff, most contributors participate on top of their national activities 2006 EUnet. HTA Project 2010 EUnet. HTA JA 1 2012 EUnet. HTA JA 2 Strengthening practical Inception application European network for Health Technology Assessment | JA 3 2016 -2020 | www. eunethta. eu Putting into practice 2016 2020 EUnet. HTA Joint Action 3 Turning pilots into standard practice 3
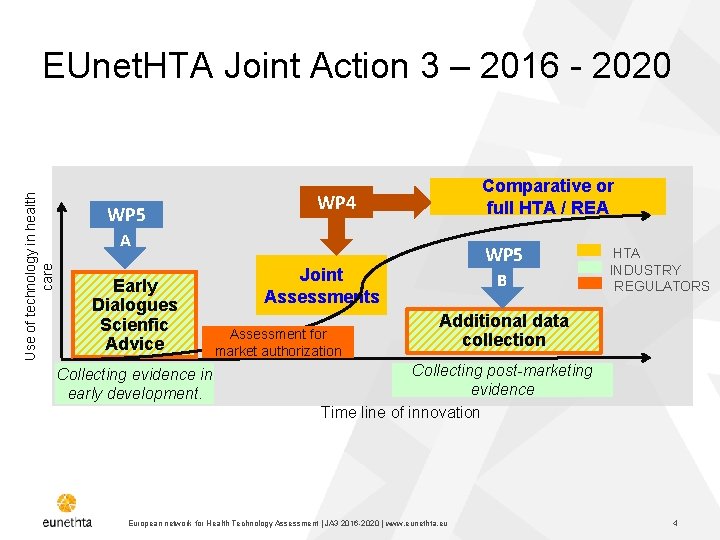
Use of technology in health care EUnet. HTA Joint Action 3 – 2016 - 2020 WP 5 Comparative or full HTA / REA WP 4 A Early Dialogues Scienfic Advice Collecting evidence in early development. WP 5 Joint Assessments Assessment for market authorization B HTA INDUSTRY REGULATORS Additional data collection Collecting post-marketing evidence Time line of innovation European network for Health Technology Assessment | JA 3 2016 -2020 | www. eunethta. eu 4

EUnet. HTA Work Package 5 Life cycle approach to Evidence Generation WP 5 : Lead Partner HAS, Co-lead partner G-BA Objective of EUnet. HTA WP 5 • To help to generate optimal and robust evidence for different stakeholders, bringing benefits for patient access and public health. �Initial evidence generation: Early dialogues WP 5 strand A �Post-launch evidence generation (PLEG) WP 5 Strand B European network for Health Technology Assessment | JA 3 2016 -2020 | www. eunethta. eu 5

Involvement of HTA bodies (HTAB) in Early Dialogues (ED) / Scientific Advice (SA) • SA by one single HTAB ˗ Started in 2009 ˗ NICE, G-BA, AIFA, HAS… ˗ HTAB only or in parallel with national regulatory agency • SA by multiple HTABs: ˗ Started in 2012: EUnet. HTA: 13 Early Dialogues ˗ Dedicated project: SEED Shaping European Early Dialogues: 14 HTABs coordinated by HAS. 11 EDs, 4 in parallel with EMA ˗ EMA initiative Parallel Scientific Advice started in 2011 European network for Health Technology Assessment | JA 3 2016 -2020 | www. eunethta. eu

Parallel Scientific Advice / Early Dialogues: situation before starting Eunet. HTA JA 3 EMA-HTA Parallel Scientific Advice Choice of HTA bodies Preference expressed by (HTAB) involved company EMA-SEED/EUnet. HTA Parallel Early Dialogues Decided by SEED partners Recruitment of participating HTABs Company seeks HTA availability SEED Coordinator (HAS) Coordination role among HTABs HTA coordinator One HTA list of issues Yes One HTA list of issues Exchanges between HTABs Exchange of pre-meeting HTA positions more intensive Final outcome Individual written HTA answers Compilation of HTA answers with an effort to reach consensus when possible European network for Health Technology Assessment | JA 3 2016 -2020 | www. eunethta. eu 7

ry t s u a Ind 7 m r Pha ws 201 Vie HTA/regulatory scientific advice Experience and ways moving forward from sponsor’s perspective Presented at “Industry stakeholder platform on research and development support” 25/04/2017, EMA, London

Industry views – April 2017 • Call for a single European HTA/reg SA process ‒ Parallel Advice between HTA and regulators needed : single global development plan ‒ But: separate advice from regulatory and HTA and national HTA advice should be possible as well, processes are complementary • Further improvements needed (based on EMA PSA and EUnet. HTA/SEED) ‒ Simplify logistics: single point of contact/project management ‒ More consistent & predictable HTA engagement, with dedicated resources and capacity building across HTAs ‒ Clear, aligned and written output from HTA advice European network for Health Technology Assessment | JA 3 2016 -2020 | www. eunethta. eu 9

Industry views – April 2017 (Cont. ) • General process ‒ Advice should be possible at all stages of product lifecycle - Early stage Pre Phase III Post-launch period ‒ Process should be open to all development products, - including new value added elements for products with well-known active substance. No selection or prioritization. ‒ Concise process • The output can inform a future joint European assessment of relative efficacy at time of launch European network for Health Technology Assessment | JA 3 2016 -2020 | www. eunethta. eu 10

List of EUnet. HTA actions for ED/SA (1) • Provide a strong process for Parallel consultations with EMA ‒ Parallel consultations launched in July 2017 • While maintaining multi-HTA Early Dialogues ‒ Available since January 2017 • Improvement of logistics: ‒ Central EUnet. HTA ED secretariat at HAS (Lead Partner) EUnet. HTA-HAS@has-sante. fr • “More consistent & predictable HTA engagement” ‒ Set up of a Working Party European network for Health Technology Assessment | JA 3 2016 -2020 | www. eunethta. eu 11

List of EUnet. HTA actions for ED/SA (2) • “Clear, aligned and written output from HTA advice” ‒ Increase cooperation and coordination by appointing a rapporteur and a scientific coordinator for each ED/SA • No selection criteria, all products to be accepted? ‒ EUnet. HTA budget limits: Selection necessary for EUnet. HTA involvement ‒ Parallel consultation possible for products not selected by EUnet. HTA Working Party : participation of HTA bodies on a voluntary basis (with no use of EUnet. HTA budget) • Life cycle approach ‒ Activities on Post-Launch Evidence Generation (PLEG) European network for Health Technology Assessment | JA 3 2016 -2020 | www. eunethta. eu 12

Patients involvement • Past experience: First tested in SEED project (2013 -2015) ‒ Strong support of EURORDIS for patient recruitment and preparation of their intervention • Several approaches tested in the frame of limited resources ‒ Interview of Individual Patients in all cases ‒ Interview and more active participation of Patients representatives ‒ Participation in the face-to-face meeting with the company not systematic (may become systematic soon) • Dedicated task force at EUnet. HTA level ‒ Global harmonised policy for all Work Packages European network for Health Technology Assessment | JA 3 2016 -2020 | www. eunethta. eu 13

Early Dialogue Working Party (EDWP) Gathers HTA bodies with important experience and cover various countries EDWP members are HTA bodies experienced in collaborative EDs: ˗ with adequate expertise, availability and budget ˗ with high commitment to participate in EDs Currently ˗ France (HAS) ˗ Germany (G-BA) ˗ UK (NICE) ˗ Italy (AIFA, RER as alternate) ˗ NL (ZIN) and BE (RIZIV INAMI) – Shared seat ˗ Hungary (NIPN) Possible extension to Scandinavian countries (NOMA and TLV), Spain European network for Health Technology Assessment | JA 3 2016 -2020 | www. eunethta. eu 14

Selection criteria for EDWP involvement • Primary criteria ‒ A new mode of action for the indication ‒ AND targeting a life-threatening or chronically debilitating disease ‒ AND responding to unmet need (no treatment or only unsatisfactory treatment available) • Secondary criteria ‒ Selected EDs should represent a wide array of topics, therapeutic areas etc. (e. g. orphan, ATMPs, antibiotics, oncology) European network for Health Technology Assessment | JA 3 2016 -2020 | www. eunethta. eu 15
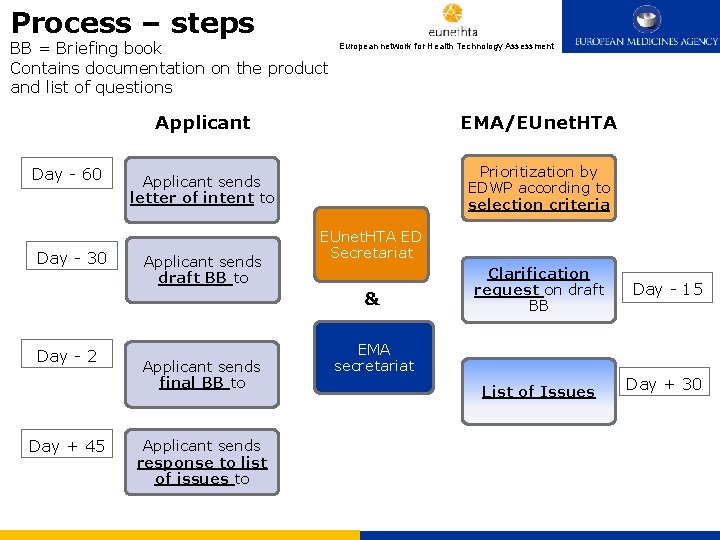
Process – steps BB = Briefing book Contains documentation on the product and list of questions Day - 60 Day - 30 European network for Health Technology Assessment Applicant EMA/EUnet. HTA Applicant sends letter of intent to Prioritization by EDWP according to selection criteria Applicant sends draft BB to EUnet. HTA ED Secretariat & Day - 2 Day + 45 Applicant sends final BB to Applicant sends response to list of issues to Clarification request on draft BB Day - 15 List of Issues Day + 30 EMA secretariat
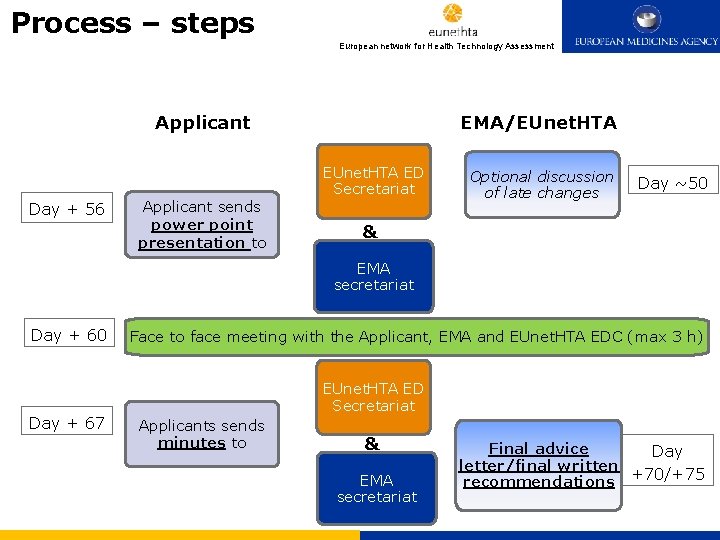
Process – steps 3. Process European network for Health Technology Assessment EMA/EUnet. HTA Applicant Day + 56 Applicant sends power point presentation to EUnet. HTA ED Secretariat Optional discussion of late changes Day ~50 & EMA secretariat Day + 60 Day + 67 Face to face meeting with the Applicant, EMA and EUnet. HTA EDC (max 3 h) EUnet. HTA ED Secretariat Applicants sends minutes to & EMA secretariat Final advice Day letter/final written recommendations +70/+75
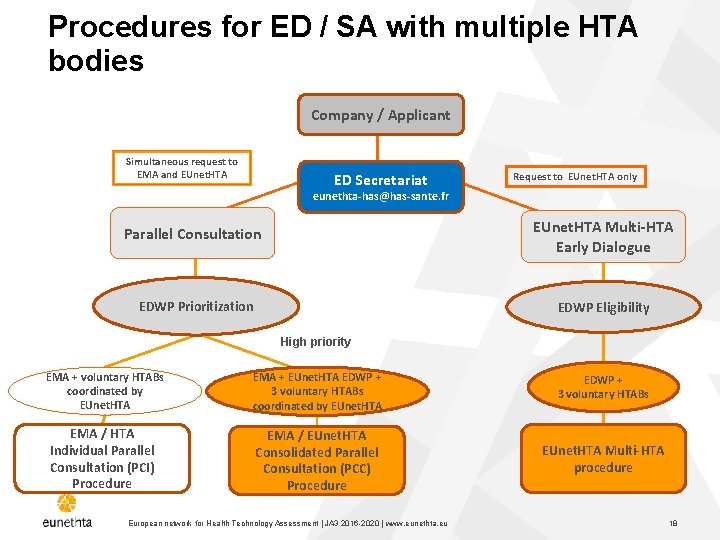
Procedures for ED / SA with multiple HTA bodies Company / Applicant Simultaneous request to EMA and EUnet. HTA ED Secretariat Request to EUnet. HTA only eunethta-has@has-sante. fr Parallel Consultation EUnet. HTA Multi-HTA Early Dialogue EDWP Prioritization EDWP Eligibility High priority EMA + voluntary HTABs coordinated by EUnet. HTA EMA + EUnet. HTA EDWP + 3 voluntary HTABs coordinated by EUnet. HTA EDWP + 3 voluntary HTABs EMA / HTA Individual Parallel Consultation (PCI) Procedure EMA / EUnet. HTA Consolidated Parallel Consultation (PCC) Procedure EUnet. HTA Multi-HTA procedure European network for Health Technology Assessment | JA 3 2016 -2020 | www. eunethta. eu 18

WP 5 A - Early Dialogues – Status since JA 3 start 20 Letters of Intent • • 3 x withdrawn • • • 1 ophthalmology 1 oncology 1 vaccine 8 Oncology 2 Neurology 2 Immuno-inflammation 1 Ophthalmology 1 Vaccine 1 Metabolic disorder 10 x EDWP 1 infectious disease 3 multi-HTA EDs + 7 PCC 2 Hematology 7 x individual PCI (~2 -3 HTABs) • • 4 oncology 1 neurology 1 Immunoinflammation 1 infectious disease • • • 4 oncology 1 neurology 1 metabolic disorder 2 Hematology 1 infectious disease 1 immuno-inflammation European network for Health Technology Assessment | JA 3 2016 -2020 | www. eunethta. eu

Collaboration on Post Launch Evidence Generation (PLEG) • PLEGs can be a topic for questions during an Early Dialogue / Parallel Consultations Work package 5 Strand B actions • Collaborative actions with the EMA on Registries ‒ Qualification of registries: 2 pilots, one on a rare disease • Drug specific pilots on collaboration between HTA bodies for the requests of PLEG ‒ Two pilots being launched, one on an orphan drug ‒ Collaboration on - analysing evidence gaps defining research questions Data or results pooling once data collection processes are put in place European network for Health Technology Assessment | JA 3 2016 -2020 | www. eunethta. eu 20

Thank you Any questions? European network for Health Technology Assessment | JA 3 2016 -2020 | www. eunethta. eu
- Slides: 21