HRA Transformation Background The HRA has been following

HRA Transformation

Background • The HRA has been following a Transformation journey since its establishment in April 2012 • The Transformation Programme has been continually updated and reconfigured to address the needs of the sector and through implementing associated changes to organisational structure, systems, workforce, culture and processes in response to the strategy • The programme remains connected to the Target Operating Model (TOM) that was shared with the Board in March 2018
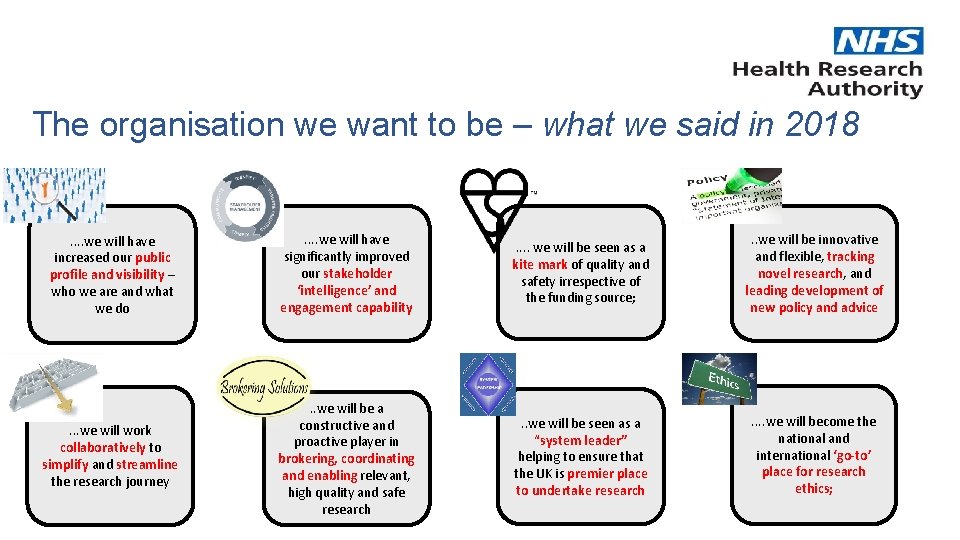
The organisation we want to be – what we said in 2018 . . we will have increased our public profile and visibility – who we are and what we do . . we will have significantly improved our stakeholder ‘intelligence’ and engagement capability . . we will be seen as a kite mark of quality and safety irrespective of the funding source; . . we will be innovative and flexible, tracking novel research, and leading development of new policy and advice . . . we will work collaboratively to simplify and streamline the research journey . . we will be a constructive and proactive player in brokering, coordinating and enabling relevant, high quality and safe research . . we will be seen as a “system leader” helping to ensure that the UK is premier place to undertake research . . we will become the national and international ‘go-to’ place for research ethics;
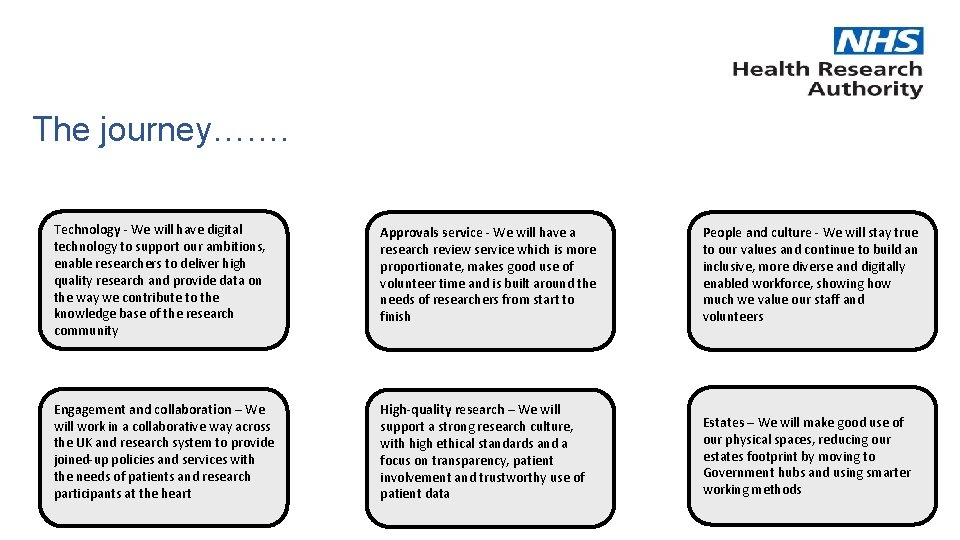
The journey……. Technology - We will have digital technology to support our ambitions, enable researchers to deliver high quality research and provide data on the way we contribute to the knowledge base of the research community Approvals service - We will have a research review service which is more proportionate, makes good use of volunteer time and is built around the needs of researchers from start to finish People and culture - We will stay true to our values and continue to build an inclusive, more diverse and digitally enabled workforce, showing how much we value our staff and volunteers Engagement and collaboration – We will work in a collaborative way across the UK and research system to provide joined-up policies and services with the needs of patients and research participants at the heart High-quality research – We will support a strong research culture, with high ethical standards and a focus on transparency, patient involvement and trustworthy use of patient data Estates – We will make good use of our physical spaces, reducing our estates footprint by moving to Government hubs and using smarter working methods
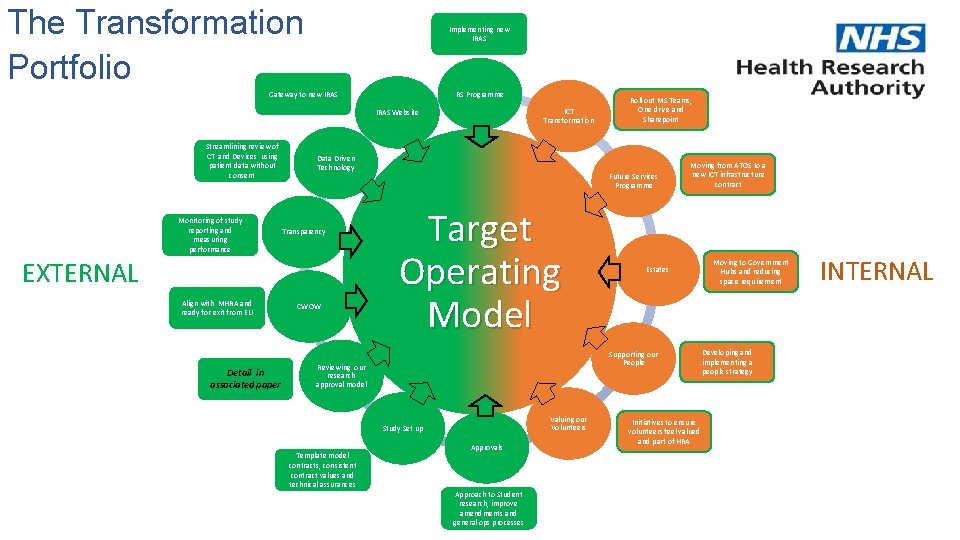
The Transformation Portfolio Implementing new IRAS RS Programme Gateway to new IRAS ICT Transformation IRAS Website Streamlining review of CT and Devices using patient data without consent Monitoring of study reporting and measuring performance Data Driven Technology Transparency EXTERNAL Align with MHRA and ready for exit from EU Detail in associated paper CWOW Future Services Programme Target Operating Model Moving from ATOS to a new ICT infrastructure contract Estates Supporting our People Reviewing our research approval model Valuing our Volunteers Study Set up Template model contracts, consistent contract values and technical assurances Roll out MS Teams, One drive and Sharepoint Approvals Approach to Student research, improve amendments and general ops processes Initiatives to ensure volunteers feel valued and part of HRA Moving to Government Hubs and reducing space requirement Developing and implementing a people strategy INTERNAL

Delivery • This is a comprehensive programme of work and needs to be delivered in line with our capacity, capability and financial constraints • This is a 3 year + programme - the timeline to achieve it extends to September 2022 and possibly beyond • A fundamental principal will be co-production • The focus must be on identifying, quantifying and delivering benefits • Covid-19 has created an environment which will enable us to accelerate already planned transformational changes

User input and the development of ‘New IRAS’
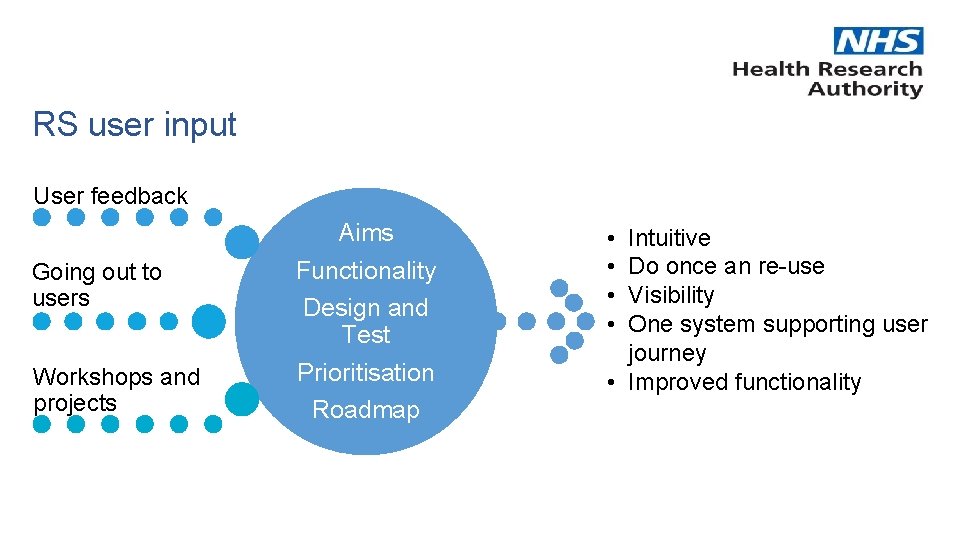
RS user input User feedback Going out to users Workshops and projects Aims Functionality Design and Test Prioritisation Roadmap • • Intuitive Do once an re-use Visibility One system supporting user journey • Improved functionality

The original ambitions – from 2017 • Allow researchers to complete applications proportionate to their studies using a clear, intuitive system, supported by clear guidance and contextualised help; • Support the validation and verification of the information completed in forms and documents to promote more efficient and effective processing • Enable the transfer of data and applications to other HRA systems and external organisations through flexible, real-time integration where possible, and through sharing via secure and open portals • Enable researchers to understand the progress of their applications to HRA and other organisations • Allow HRA personnel to create or modify applications, questions, guidance and integration • Enable HRA to be flexible and agile to future business needs These still stand! Ongoing work develops this eg transparency programme.
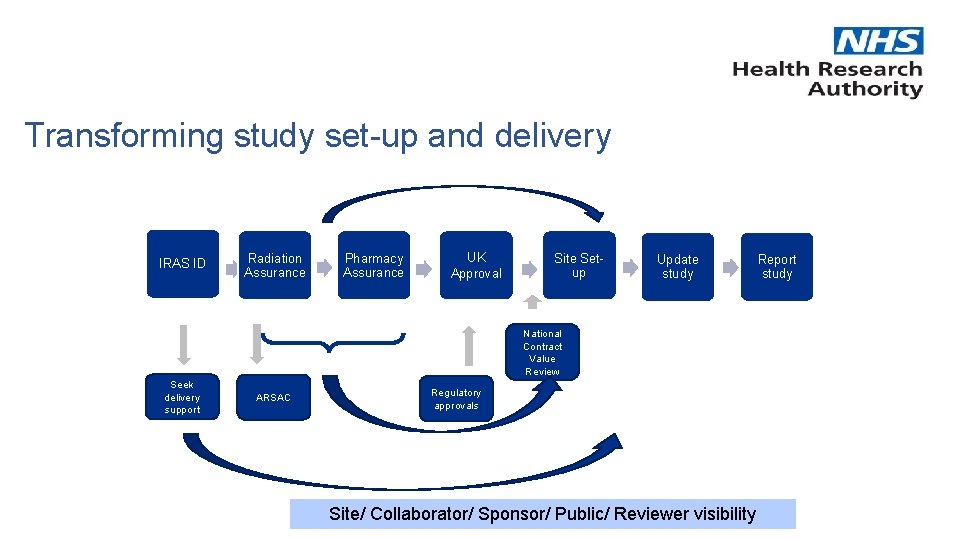
Transforming study set-up and delivery IRAS ID Radiation Assurance Pharmacy Assurance UK Approval Site Setup Update study National Contract Value Review Seek delivery support ARSAC Regulatory approvals Site/ Collaborator/ Sponsor/ Public/ Reviewer visibility Report study
- Slides: 10