HRA HCRW Approval Natalie Wilson February 2020 Getting

HRA & HCRW Approval Natalie Wilson – February 2020

Getting it right first time…. . Applying for HRA & HCRW Approval www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

Learning objectives Today we will: • discuss when HRA-HCRW is and is not required • learn about the HRA-HCRW Approval process • describe the essential documentation required as part of a submission • learn briefly about the REC review and assessment process and documents • discuss steps to take regarding participating NHS organisations • briefly discuss study amendments • consider individual study queries www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk
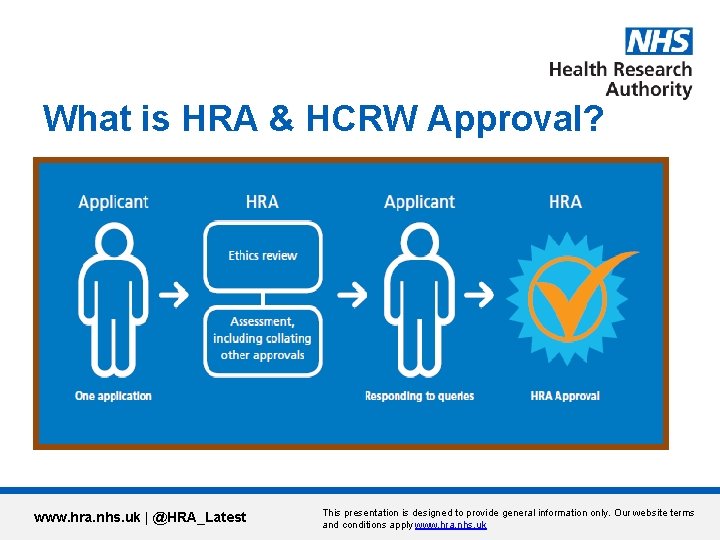
What is HRA & HCRW Approval? www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

When is HRA & HCRW Approval required? HRA & HCRW Approval is required for: • research including NHS patients • research including NHS staff • research where the duty for care for participants is the NHS In addition to the above, those recruited should be from NHS organisation because of their affiliation with the organisation (i. e. a patient at the organisation, member of staff) • It may also be required where the protocol mandates the collection of data from GP records but may not fall under the above. www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

Is HRA & HCRW Approval required in all case? • No, there are instance where HRA & HCRW are not required. This includes: – Where they are no NHS organisations involved that will act as PICs or research sites. – Research that only includes healthy volunteers – NHS staff research where they are recruited due to their affiliation/membership of a group (e. g. Royal College of Surgeons) rather than by virtue of their employment at an NHS site – Where activity at a site is limited to displaying a poster www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

Is HRA & HCRW Approval required in all case? continued • Where an NHS organisation is sponsor, this does not necessarily mean that HRA & HCRW Approval is required. This is the case in the following instances: – Where no NHS organisations are involved (e. g. only involves local authority/care homes – Also previously mentioned examples www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

What does a good submission look like? • Remember – it is a single application to HRA for research ethics review and for HRA assessment • Need to consider requirements for both reviews – Read guidance in IRAS and on HRA website – HRA publish Assessment Standards and Criteria www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

Completing IRAS • • IRAS = Platform Application process is via IRAS www. myresearchproject. org. uk Integrated forms – based on filter questions Guidance available to complete form Electronically submitted for HRA Approval Other regulators – submit as directed – CAG www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

IRAS Project Filter Clicking on the green info icon provides question specific guidance

IRAS Project Filter Research using tissue: check section B 4 & B 5 are fully completed. ALC: If study involves adults (16 yrs plus) lacking capacity, this box should be ticked and associated Section B is completed Student Project?

Completing IRAS questions • Don’t cut and paste • Answer questions • Answer all questions – If not applicable – put not applicable. • Ensure consistency www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

Electronic authorisations • Chief Investigator – Supervisor normally expected to be CI if educational research (exception listed in UK policy framework for H&S) • Sponsor – Each organisation has a list of authorised signatories – speak to your RD office as to who this is. • Other e. g. Academic supervisor www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

Essential applicant/ sponsor check Pre-submission checks IRAS Form Filter questions completed correctly All sections of Form completed Form has been electronically authorised Correct documents submitted with version numbers and dates IRAS reference number on PIS and consent form GDPR transparency wording in PIS IRAS Checklist completed

Supporting documents Essential Documents Other documents examples Protocol Assessments/Tasks Organisation Information Document/ Interview schedules Schedule of Events or So. ECAT (if multicentre/Sponsor and site differ) Evidence of Insurance (non-NHS Sponsors) Questionnaires Funding Letter GP Letters CV for CI *** Student and supervisor CVs if educational*** Participant information Sheets Consent Forms www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

Organisation Information Document • One per site type • Provides sites with relevant information to aid the setup of research to support arranging capacity and capability • Can be used as the intended agreement between sponsor and participating NHS sites – Is the Organisation Information Document being used as the contract with the study sites. If not, what will be used instead? www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

Organisation Information Document cont… • Identify whether PI, Local Collaborator or CI are required • Have the appendices been correctly completed (finance, material transfer, data, IP)? • Guidance available • Lets take a closer look… www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

Schedule of Events/So. ECAT • One per site type So. E: • Details research activity occurring at participating NHS organisations • Includes cost attributions So. ECAT: • As above but includes ETCs • Authorised by Aco. RD Specialist

Schedule of Events/So. ECAT cont… • Does the Schedule list all of the activities that will take place at each study visit (either at the NHS site or by NHS staff)? • Are timings for these activities included and a statement re: who will undertake the activity? • Have cost attributions been provided for each activity? (required even if no funding available) • Guidance available and training module

m. NCA/modified agreements • Usually see for top 4 studies in IRAS • m. NCA is generally used where sponsor is NHS • Universities may use modified agreements because: – Inappropriate indemnity and liabilities clauses are used (lack of reciprocal indemnity) – Funding terms and conditions cannot be included – Inability to reflect some co-sponsor arrangements www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

m-NC-PICA • Sub-contract set up between PIC and participating NHS organisation the PIC supports • Required to cover data processing arrangements NHS PIC only studies • If study only involves NHS PICs, the type of contracting arrangements sponsor and the PIC has in place should be confirmed www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

Submitting • Ring Central Booking Service • Answer questions by CBS – Have your application form with you – Questions are based on filter question • Add ref number only (this will not knock off authorisations. www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk
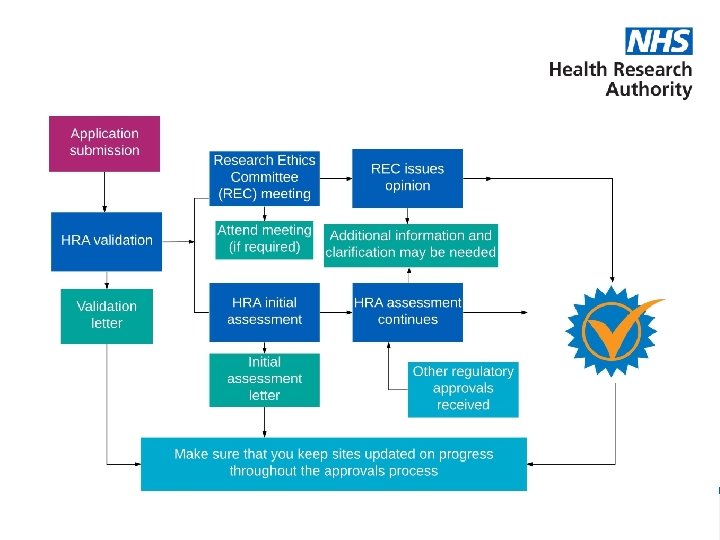

During review… • Strongly encouraged where possible to attend REC meeting in person to answer queries – For PR – be contactable by phone/email during review period • Answer queries from Assessors or REC promptly • Confirm any conditions of the REC FO promptly • Submit any other regulatory approvals – e. g CAG, ARSAC www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

What happens at a Full Research Ethics Committee meeting? • Application sent to the members 2 weeks prior to the meeting date. • Pre-discussion and discussion with the researcher • Ethical Opinion issued within a maximum of 60 days (target 40 days) www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

Committee Members • Volunteers; Mix of lay and expert members. • Up to 15 members per REC (at least 1/3 must be lay). • Quoracy at a meeting is 7 members, to include an Officer, an expert member and a lay member. • Various RECs may have special expertise www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

Decisions at a REC Meeting These include: • • • Favourable opinion with conditions Provisional opinion Unfavourable opinion No Opinion (PR only) www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

HRA & HCRW Initial Assessment Letter • Clarifies: – Site types – Documents being used – Funding Information – Person responsible for activities at sites – HR Good Practice requirements • Applicant provides to sites as part of UK Local Information Pack www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

HRA Approval Letter • Includes information previously detailed in the Initial Assessment Letter • Lists documents approved – including any revised documents www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

Working with sites www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk
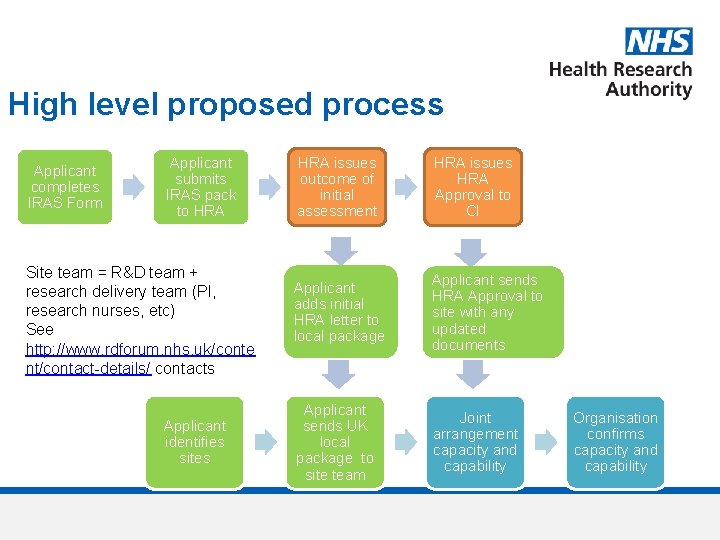
High level proposed process Applicant completes IRAS Form Applicant submits IRAS pack to HRA Site team = R&D team + research delivery team (PI, research nurses, etc) See http: //www. rdforum. nhs. uk/conte nt/contact-details/ contacts Applicant identifies sites HRA issues outcome of initial assessment HRA issues HRA Approval to CI Applicant adds initial HRA letter to local package Applicant sends HRA Approval to site with any updated documents Applicant sends UK local package to site team Joint arrangement capacity and capability Organisation confirms capacity and capability

Amendments www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk
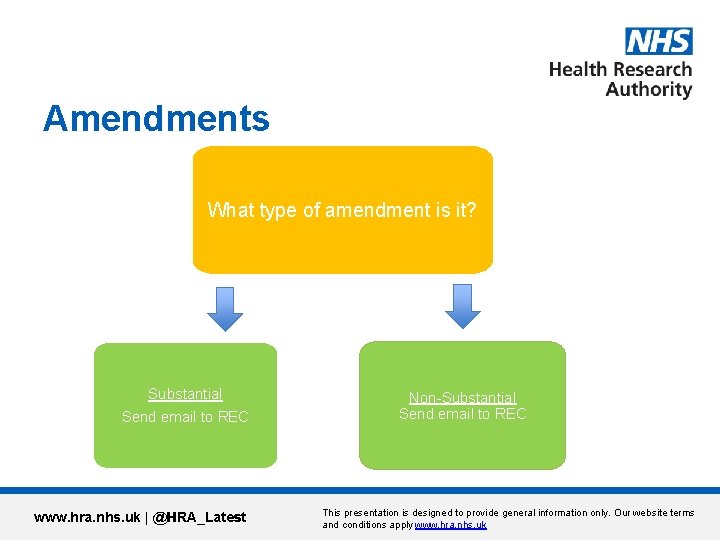
Amendments What type of amendment is it? Substantial Send email to REC www. hra. nhs. uk | @HRA_Latest Non-Substantial Send email to REC This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

Amendments • Ultimately sponsor’s responsibility to classify an amendment as substantial/non-substantial. • Make the changes clear – impact to support departments. Will need to confirm they can continue support. • Further Information and guidance can be found on in IRAS/on our website. www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

Top Tips • Read all communications from HRA carefully • Make sure that all relevant documentation is submitted • Include version control • Discuss relevant information with supervisor – HRA expects student researchers to be fully supported by their supervisors who should understand the process • Complete relevant learning modules available via our website for supervisors and students www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

Discussion Time Things to discuss: • Student specific queries • Any additional requirements Remember, no query/question is a stupid query/question! www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

Any questions www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

Summary Today we have: • discussed when HRA-HCRW is and is not required • learned about the HRA-HCRW Approval process • described the essential documentation required as part of a submission • learned briefly about the REC review and assessment process and documents • discussed steps to take regarding participating NHS organisations • briefly discussed study amendments • considered individual study queries www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk

References • https: //www. hra. nhs. uk/planning-and-improvingresearch/best-practice/nhs-site-set-up-in-england/ • https: //www. myresearchproject. org. uk/help/hlpsitespe cific. aspx • HRA & HCRW Approval • Amendments www. hra. nhs. uk | @HRA_Latest This presentation is designed to provide general information only. Our website terms and conditions apply www. hra. nhs. uk
- Slides: 39