HPV Zoster TB Etc Med Ch 401 Lecture

HPV, Zoster, TB, Etc. Med. Ch 401 Lecture 9 2 Jun 06 KL Vadheim Lecture 9 1

Human Papillomavirus • • • Papillomaviridae; ds DNA genome ~100 different types Produces warts on various body parts 30 -40 types infect genital tissue Low risk: types 6 and 11, e. g. – genital warts • High risk: ~ 15 types – 16, 18, 31, 45 cause most cancers 2 Jun 06 KL Vadheim Lecture 9 2

HPV infections • • • Most common STD worldwide ~20 million infected in U. S. ~5. 5 million new infections annually Often asymptomatic Most infections spontaneously resolve Infects cervix, vagina, vulva, anus, penis 2 Jun 06 KL Vadheim Lecture 9 3

High Risk HPVs • Cause intraepithelial neoplasias • Can progress to cancer if undetected/untreated • HPV viral sequences (oncogenes) integrated in cellular DNA • Viral E 6 protein binds/degrades p 53, a tumor suppressor gene 2 Jun 06 KL Vadheim Lecture 9 4

HPV Vaccines Under Review • Merck - Gardasil – Quadravalent (6, 11, 16, 18) – Recombinant – Recommended for approval by VRPAC • GSK – Bivalent (16 and 18) – Phase III trials ongoing 2 Jun 06 KL Vadheim Lecture 9 5

Merck’s Gardasil • Targeted for women 9 -26 years of age • 100% effective in preventing type 16 and 18 infection (~70% cervical CA) • 99% effective in preventing type 6 and 11 infection (90% of genital warts) • Complement to - not a replacement for Pap smears • $300 - $500 per vaccination 2 Jun 06 KL Vadheim Lecture 9 6

Cervical Cancer in U. S. • >9, 000 women diagnosed annually • ~3, 700 deaths • Risk factors for development of cervical CA – high-risk type HPV infection – smoking – having many children – long-term oral contraceptive use – HIV infection 2 Jun 06 KL Vadheim Lecture 9 7

Zostavax • Merck’s shingles vaccine • Licensed May 25, 2006 • Reduce risk of herpes zoster in people >60 – 50% efficacy in all people >60 – 64% efficacy in people 60 -69 • Three-year efficacy trial 2 Jun 06 KL Vadheim Lecture 9 8

Merck’s Zostavax & Varivax • Oka/Merck strain of live, attenuated VZV • Initially obtained from child with natural varicella infection • Attenuation – Human embryonic lung cells – Embryonic guinea pig cells – Human diploid cells (WI-38) • Lyophilized • Subcutaneous administration 2 Jun 06 KL Vadheim Lecture 9 9
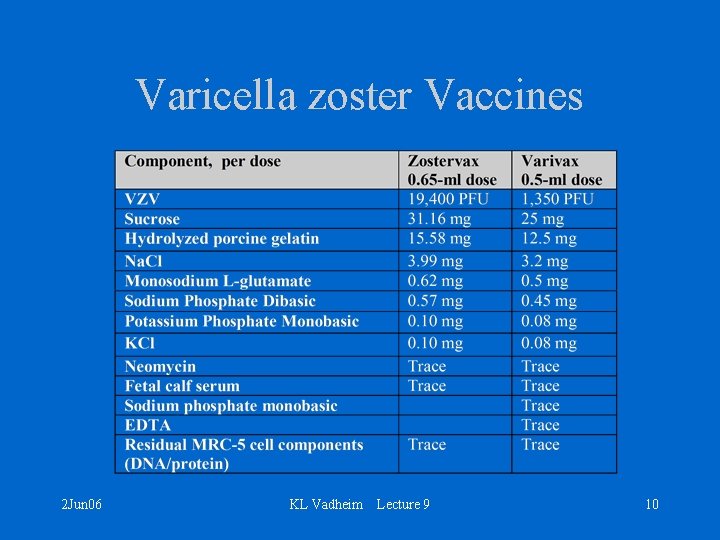
Varicella zoster Vaccines 2 Jun 06 KL Vadheim Lecture 9 10

Tuberculosis • • 2 billion people are infected worldwide 2 million deaths annually worldwide 1 in 10 infected will develop active TB 98% of deaths are in the developing world, affecting primarily young adults • 8. 8 million new TB cases in 2003 – 80% in 22 countries 2 Jun 06 KL Vadheim Lecture 9 11

Pandemic TB • Global incidence is growing at 1% per year • 25% of all cases are in Africa • 50% of new cases are in six Asian countries – Bangladesh – China – India – Indonesia – Pakistan – The Philippines 2 Jun 06 KL Vadheim Lecture 9 12

TB Transmission • Airborne • No animal vector known 2 Jun 06 KL Vadheim Lecture 9 13

Multi-Drug Resistance in TB • Curable, but 5, 000 people die daily • MDR-TB present in nearly all 109 countries surveyed by WHO • 425, 000 new MDR-TB cases every year – highest rates in former USSR and China – up to 14% of all new cases are not responding to standard drug treatment 2 Jun 06 KL Vadheim Lecture 9 14

TB control strategies • U. S. – Test and treat – Mantoux test (PPD skin reactivity test) • Europe – BCG vaccination • attenuated TB strain – Dubious efficacy – Recent studies place efficacy near zero 2 Jun 06 KL Vadheim Lecture 9 15

New TB control strategies (WHO) • Government commitment to TB control • Diagnosis through bacteriology and an effective lab network • Standardized short-course chemotherapy with full patient support • Uninterrupted supply of quality-assured drugs • Documenting patient outcomes 2 Jun 06 KL Vadheim Lecture 9 16

Malaria • • • >1 million deaths annually 300 -500 million acute illnesses each year Endemic in >100 countries >80% of deaths in sub-Saharan Africa Most deaths in infants and young children – 3, 000 children die of malaria every day 2 Jun 06 KL Vadheim Lecture 9 17

Malaria transmission • Single-celled parasite carried by Anopheles mosquito • Many animal hosts in addition to humans • Complex life cycle makes control difficult 2 Jun 06 KL Vadheim Lecture 9 18

Malaria Control • Insecticide-laced mosquito nets – reduces childhood deaths ~20% • Chloroquine – effective against P. vivax, ~30% of cases • Sulphadoxin-Pyrimethamine (SP) – P. falciparum • Drug resistance a problem • Artemisinin-based Combination Therapy (ACT) 2 Jun 06 KL Vadheim Lecture 9 19

Malaria Vaccine Development • PATH – Malaria Vaccine Initiative • GSK’s candidate vaccine ~50% efficacy against most lethal forms of disease 2 Jun 06 KL Vadheim Lecture 9 20

Specific Immune Globulins • • 2 Jun 06 Botulinum antitoxin Botulism IGIV Cytomegalovirus IG IV Hepatitis B IGIV Rabies IG (Human) RSV IGIV (Human) Tetanus IG (Human) KL Vadheim Lecture 9 21

Non-specific IGIV • Immune Globulin (Human) • Immune Globulin IV (Human) • Immune Globulin Subcutaneous (Human) – can be self-administered 2 Jun 06 KL Vadheim Lecture 9 22

Non-specific IGIVs • Highly purified Ig. G preparation • Made from donated human plasma • Used to treat people with insufficient antibody production • Usually given every 3 -4 weeks • Treatment may be life-long 2 Jun 06 KL Vadheim Lecture 9 23
2 Jun 06 KL Vadheim Lecture 9 24

Complaints • Call or write the manufacturer • Provide as much information as possible • Expect an investigation and written response 2 Jun 06 KL Vadheim Lecture 9 25

Recalls and Withdrawals • www. fda. gov/cber/recalls 2 Jun 06 KL Vadheim Lecture 9 26

Adverse Events (AEs) • Adverse event - any untoward medical occurrence in a patient administered a medicinal product and which does not necessarily have to have a causal relationship with this treatment • Adverse Drug Reaction - all noxious and unintended responses to a medicinal product • Adverse Reaction - implies causal relationship; aka side effect 2 Jun 06 KL Vadheim Lecture 9 27

AEs • Local, systemic, allergic • Serious AE – results in death – is life-threatening – requires hospitalization – results in persistenr or significant disability – is a congenital anomaly/birth defect • Unexpected v. expected (labeled) 2 Jun 06 KL Vadheim Lecture 9 28

Pharmacovigilence • All scientific and data gathering activities relating to the detection, assessment and understanding of AEs • Involves identification and evaluation of safety signals 2 Jun 06 KL Vadheim Lecture 9 29

Safety signals • A concern about an excess of AEs compared to what is expected with that product • Single event • Group of events that indicate the need for further investigations - e. g. , intussesception with Rota. Shield 2 Jun 06 KL Vadheim Lecture 9 30

Safety Signals • • New, serious AEs Increase in severity of labeled AE Increase in frequency of serious AE New product-product, product-diet supplement interactions • Identification of previously unrecognized atrisk populations • Misuse of a product 2 Jun 06 KL Vadheim Lecture 9 31

Good Case Reports • Complete description of event • Product therapy details - dose, lot #, schedule, dates, dietary supplements or OTC meds taken, etc. • Patient characteristics • Documentation of diagnosis of the event(s) • Clinical course and patient outcomes • Relevant therapeutic measure and lab data during and after therapy • Any other relevant information 2 Jun 06 KL Vadheim Lecture 9 32

Pharmacovigilence • ICH Guidance for Industry: Good Pharmacovigilance Practices and Pharmacoepidemiologic Assessment, April 2005 • ICH Guidance for Industry: Pharmacovigilance Planning, April 2005 • Guidance for Industry: How to Complete the VAERS Form, Sept. 1998 • www. fda. gov/cber/guidelines 2 Jun 06 KL Vadheim Lecture 9 33
- Slides: 33