How to replace bicarbonate deficit in metabolic acidosis

How to replace bicarbonate deficit in metabolic acidosis 가톨릭대학교 여의도성모병원 내과 정성진
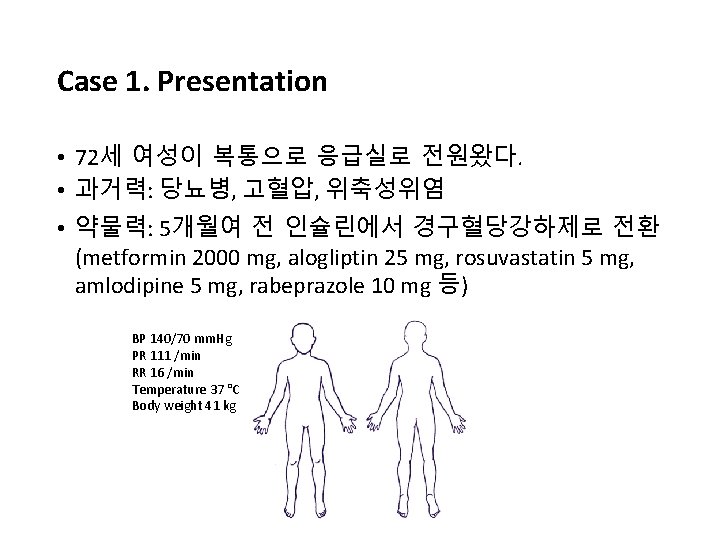
Case 1. Presentation • 72세 여성이 복통으로 응급실로 전원왔다. • 과거력: 당뇨병, 고혈압, 위축성위염 • 약물력: 5개월여 전 인슐린에서 경구혈당강하제로 전환 (metformin 2000 mg, alogliptin 25 mg, rosuvastatin 5 mg, amlodipine 5 mg, rabeprazole 10 mg 등) BP 140/70 mm. Hg PR 111 /min RR 16 /min Temperature 37 °C Body weight 41 kg
Case 1. Imaging studies
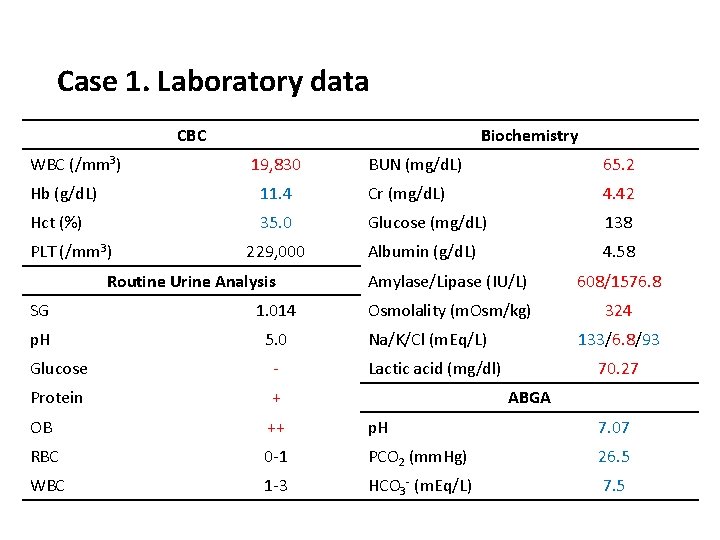
Case 1. Laboratory data CBC WBC (/mm 3) Biochemistry 19, 830 BUN (mg/d. L) 65. 2 Hb (g/d. L) 11. 4 Cr (mg/d. L) 4. 42 Hct (%) 35. 0 Glucose (mg/d. L) 138 Albumin (g/d. L) 4. 58 PLT (/mm 3) 229, 000 Routine Urine Analysis SG 1. 014 p. H 5. 0 Amylase/Lipase (IU/L) 608/1576. 8 Osmolality (m. Osm/kg) 324 Na/K/Cl (m. Eq/L) 133/6. 8/93 Glucose - Lactic acid (mg/dl) 70. 27 Protein + OB ++ p. H 7. 07 RBC 0 -1 PCO 2 (mm. Hg) 26. 5 WBC 1 -3 HCO 3 - (m. Eq/L) 7. 5 ABGA

When to give bicarbonate to patients with metabolic acidosis Nephrologists Critical care physicians 6% 40% Considering the PCO 2 in making decision to treat >80% 59% Treating lactic acidosis with bicarbonate 86% 66% Treating diabetic ketoacidosis with bicarbonate 60% 28% Calculating the amount of bicarbonate required 75% 33% Not giving bicarbonate unless the p. H is <7. 0 The decisions whether to use base for the treatment of acute severe metabolic acidosis, and under which circumstances, vary among physicians, indicating the need for further studies to develop evidence-based guidelines for therapy. Clin Exp Nephrol 2006; 10: 111 -117

Causes of metabolic acidosis General Mechanism Specific Clinical Example True HCO 3 deficit Kidney Renal tubular acidosis Gastrointestinal Diarrhea H+ gain Exogenous acid NH 4 Cl administration, toxins Abnormal lipid metabolism Diabetic ketoacidosis Abnormal carbohydrate metabolism Lactic acid Normal protein metabolism Uremic acidosis J Am Soc Nephrol 2009; 20: 692– 695

Treatment of metabolic acidosis with alkali • Patients with a normal AG acidosis (hyperchloremic acidosis) or an AG attributable to a nonmetabolizable anion due to advanced kidney failure should receive alkali therapy, either PO (Na. HCO 3 or Shohl’s solution) or IV (Na. HCO 3), in an amount necessary to slowly increase the plasma [HCO 3−] to a target value of 22 mmol/L. • Controversy exists in regard to the use of alkali in patients with a pure AG acidosis owing to accumulation of a metabolizable organic acid anion (ketoacidosis or lactic acidosis). Harrison's Principles of Internal Medicine, 20 th ed. Mc. Graw-Hill Education; 2018

4 Questions regarding bicarbonate replacement 1. What are the deleterious effects of acidemia, and when do they manifest? 2. When is acidemia severe enough to warrant therapy? 3. How much bicarbonate should be given, and how is that amount calculated? 4. What are the deleterious effects of bicarbonate therapy? J Am Soc Nephrol 2009; 20: 692– 695

Complications associated with severe acidemia • ↓ myocardial contractility, cardiac output and BP • ↓ binding of norepinephrine to its receptors • Shifting the oxyhemoglobin curve to the right releasing more O 2 • Protons bind to intracellular & extracellular proteins (albumin & hemoglobin) • Adversely affecting cell functions such as enzymatic reactions, ATP generation, fatty acid biosynthesis, bone formation/resorption J Am Soc Nephrol 2009; 20: 692– 695
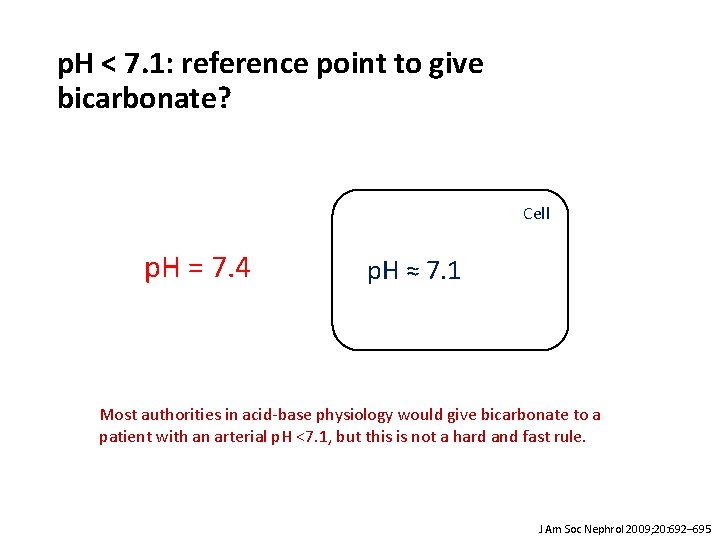
p. H < 7. 1: reference point to give bicarbonate? Cell p. H = 7. 4 p. H ≈ 7. 1 Most authorities in acid-base physiology would give bicarbonate to a patient with an arterial p. H <7. 1, but this is not a hard and fast rule. J Am Soc Nephrol 2009; 20: 692– 695

Bicarbonate deficit • HCO 3 - deficit (m. Eq) = 0. 5 x lean body weight (kg) x (desired [HCO 3 -] - measured [HCO 3 -]) • Bicarbonate dose (m. Eq): 0. 5 (L/kg) x body weight (kg) x desired increase in serum HCO 3 - (m. Eq/L) • Deficit of Na. HCO 3 (m. Eq) = volume of distribution (Vd) x target change in [HCO 3 -] • Vd = total body weight (kg) x [0. 4 + (2. 4/[HCO 3 -]) • Bicarbonate deficit = 0. 2 x weight (kg) x base deficit (m. Eq/L) • Bicarbonate deficit = Vd x (desired [HCO 3 -] - measured [HCO 3 -]) • Vd = (0. 4 + 2. 6/HCO 3 -) x lean body weight • Administration: 50% of total deficit over 3 to 4 hours + remainder over 8 -24 hours Manual of Medical Therapeutics, 28 th ed. Little, Brown and Company; 1995 Handbook of Applied Therapeutics, 1 st ed. Lippincott Williams & Wilkins; 2006 The Washington Manual of Critical Care, 1 st ed. Lippincott Williams & Wilkins; 2007 Mayo Clinic Internal Medicine Concise Textbook, 1 st ed. CRC Press; 2007 J Am Soc Nephrol 2009; 20: 692– 695

Volume of distribution of bicarbonate • The volume of distribution of bicarbonate is approximately that of total body water. • In patients with metabolic acidosis, it is said to vary from 50% to 100%, depending on the severity of the academia • This distribution obviously affects the calculated bicarbonate deficit. • Any calculated amount is only approximate. J Am Soc Nephrol 2009; 20: 692– 695

Issue associated with bicarbonate therapy • PCO 2 change after an infusion of sodium bicarbonate p. H PCO 2 (mm. Hg) HCO 3 - (m. Eq/L) 7. 1 13 4 ? Na. HCO 3 infusion 8 α = solubility coefficient for CO 2 = 0. 0306 mmol/L/mm. Hg (for plasma at 37°C) The change in PCO 2 (ΔPCO 2) at the end of the Na. HCO 3 infusion was 6. 7± 1. 8 mm. Hg (range: 4. 4 -10. 3 mm. Hg). Intensive Care Med 2000; 26: 558 -564

Mortality risk associated with bicarbonate therapy • ↓ BP and cardiac output • Sensitizing the heart to abnormal electrical activity and subsequent arrhythmias • Shift in ionized Ca • K shifts out of the cell • Paradoxic intracellular acidosis • Volume expansion and hypernatremia • Fulminant congestive heart failure with flash pulmonary edema • Superoxide formation, ↑ proinflammatory cytokine release or ↑ apoptosis • Rebound alkalemia • ↑ blood acetoacetate, β-hydroxybutyrate and lactate levels (DKA) Am J Kidney Dis 2016; 68: 473 -482 J Am Soc Nephrol 2009; 20: 692– 695

Suggested alkali therapy in severe academia (p. H <7. 10) in an adult patient • Estimation of the ‘bicarbonate deficit’ by calculation of the volume of distribution of bicarbonate is often taught but is unnecessary and may result in administration of excessive amounts of alkali. Initial goal ↑ [HCO 3−] to 10– 12 mmol/L and ↑ p. H to ∼ 7. 20 (not to normal) Method IV administration of 50 m. Eq of Na. HCO 3 diluted in 300 m. L of sterile water over 30– 45 min, during the initial 1 -2 hr of therapy (added measure of safety) Harrison's Principles of Internal Medicine, 20 th ed. Mc. Graw-Hill Education; 2018
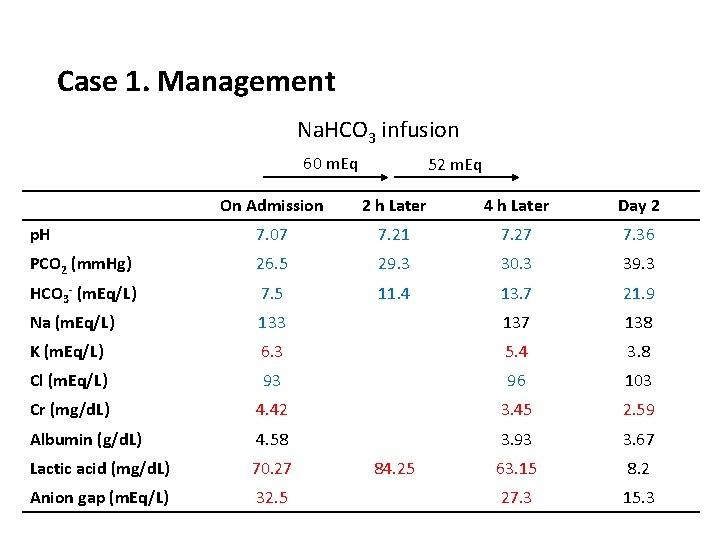
Case 1. Management Na. HCO 3 infusion 60 m. Eq 52 m. Eq On Admission 2 h Later 4 h Later Day 2 p. H 7. 07 7. 21 7. 27 7. 36 PCO 2 (mm. Hg) 26. 5 29. 3 30. 3 39. 3 HCO 3 - (m. Eq/L) 7. 5 11. 4 13. 7 21. 9 Na (m. Eq/L) 133 137 138 K (m. Eq/L) 6. 3 5. 4 3. 8 Cl (m. Eq/L) 93 96 103 Cr (mg/d. L) 4. 42 3. 45 2. 59 Albumin (g/d. L) 4. 58 3. 93 3. 67 Lactic acid (mg/d. L) 70. 27 63. 15 8. 2 Anion gap (m. Eq/L) 32. 5 27. 3 15. 3 84. 25
Case 2. Presentation • 39세 여성이 3개월여 전부터 시작된 극도의 피 로감으로 왔다. • ROS: weight loss (+, 5 kg/2 m), fatigue (+), foamy urine (+, for 3 m), nocturia (+), constipation (+) BP 140/90 mm. Hg PR 90 /min Height 163. 1 cm Body weight 49. 65 kg
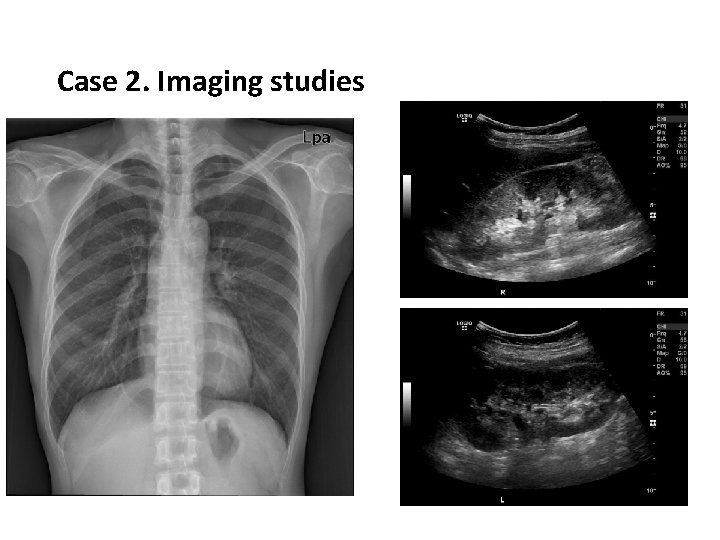
Case 2. Imaging studies
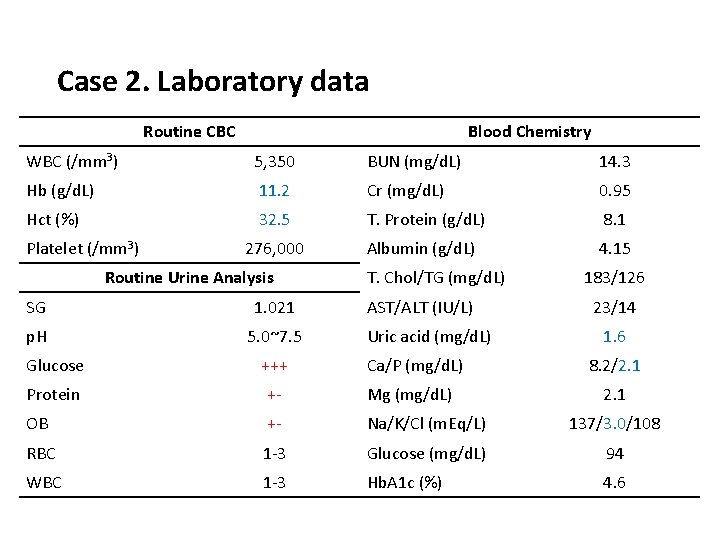
Case 2. Laboratory data Routine CBC Blood Chemistry WBC (/mm 3) 5, 350 BUN (mg/d. L) 14. 3 Hb (g/d. L) 11. 2 Cr (mg/d. L) 0. 95 Hct (%) 32. 5 T. Protein (g/d. L) 8. 1 276, 000 Albumin (g/d. L) 4. 15 Platelet (/mm 3) Routine Urine Analysis SG 1. 021 p. H 5. 0~7. 5 T. Chol/TG (mg/d. L) AST/ALT (IU/L) Uric acid (mg/d. L) 183/126 23/14 1. 6 Glucose +++ Ca/P (mg/d. L) 8. 2/2. 1 Protein +- Mg (mg/d. L) 2. 1 OB +- Na/K/Cl (m. Eq/L) 137/3. 0/108 RBC 1 -3 Glucose (mg/d. L) 94 WBC 1 -3 Hb. A 1 c (%) 4. 6
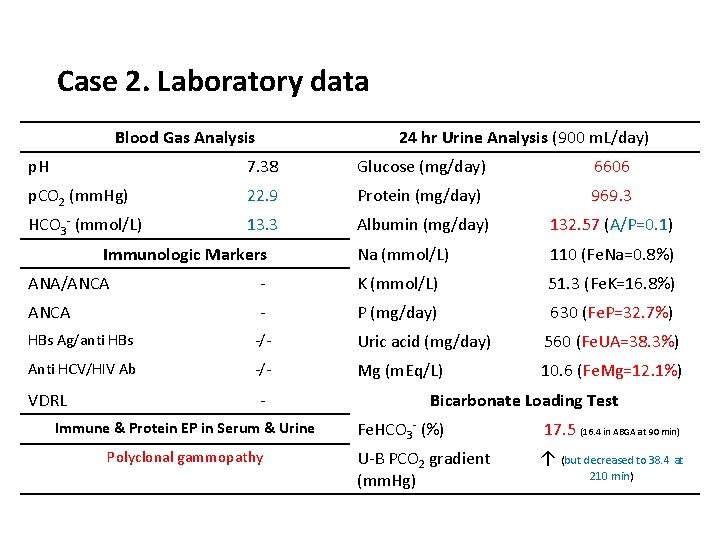
Case 2. Laboratory data Blood Gas Analysis 24 hr Urine Analysis (900 m. L/day) p. H 7. 38 Glucose (mg/day) 6606 p. CO 2 (mm. Hg) 22. 9 Protein (mg/day) 969. 3 HCO 3 - (mmol/L) 13. 3 Albumin (mg/day) 132. 57 (A/P=0. 1) Na (mmol/L) 110 (Fe. Na=0. 8%) Immunologic Markers ANA/ANCA - K (mmol/L) 51. 3 (Fe. K=16. 8%) ANCA - P (mg/day) 630 (Fe. P=32. 7%) HBs Ag/anti HBs -/- Uric acid (mg/day) 560 (Fe. UA=38. 3%) Anti HCV/HIV Ab -/- Mg (m. Eq/L) 10. 6 (Fe. Mg=12. 1%) VDRL - Immune & Protein EP in Serum & Urine Polyclonal gammopathy Bicarbonate Loading Test Fe. HCO 3 - (%) 17. 5 (16. 4 in ABGA at 90 min) U-B PCO 2 gradient (mm. Hg) ↑ (but decreased to 38. 4 at 210 min)
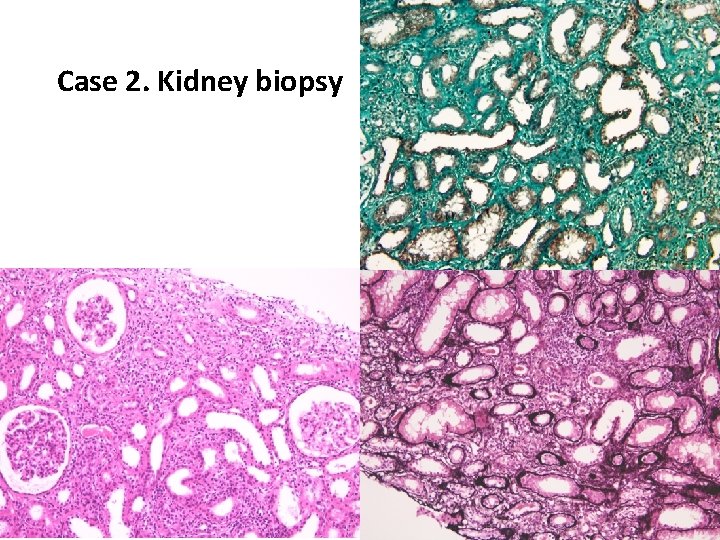
Case 2. Kidney biopsy

Treatment of renal tubular acidosis (RTA) Distal RTA Proximal RTA Hyperkalemia RTA Daily loss of HCO 3 -: 1 -2 m. Eq/kg/d Goal: increasing serum HCO 3 Goal: normalizing serum K concentration as close to normal concentration ( improvement as possible of metabolic acidosis) Amount of supplemental sodium or potassium bicarbonate: 1 -2 m. Eg/kg/d (Children: 4 -8 m. Eq/kg/d) 1. Oral HCO 3 -: 10 -15 m. Eq/kg/d with K supplementation 2. Phosphate supplementation: often required in Fanconi syndrome Am J Kidney Dis 2016; 68: 488 -498
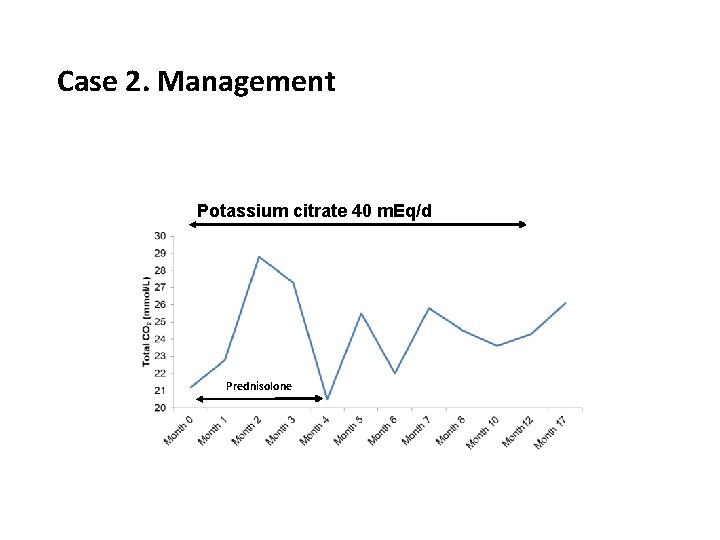
Case 2. Management Potassium citrate 40 m. Eq/d Prednisolone
Case 3. Presentation • 62세 여성이 신장 기능 저하를 걱정하여 왔다. • 과거력: 고혈압 • 수술력: 슬관절치환(좌측), 백내장 • 약물력: valsartan 160, thiazide 12. 5, amlodipine 5 mg, febuxostat 40 mg, kremezin, kayexalate, ferrous sulfate BP 166/80 mm. Hg PR 95 /min Body weight 50 kg
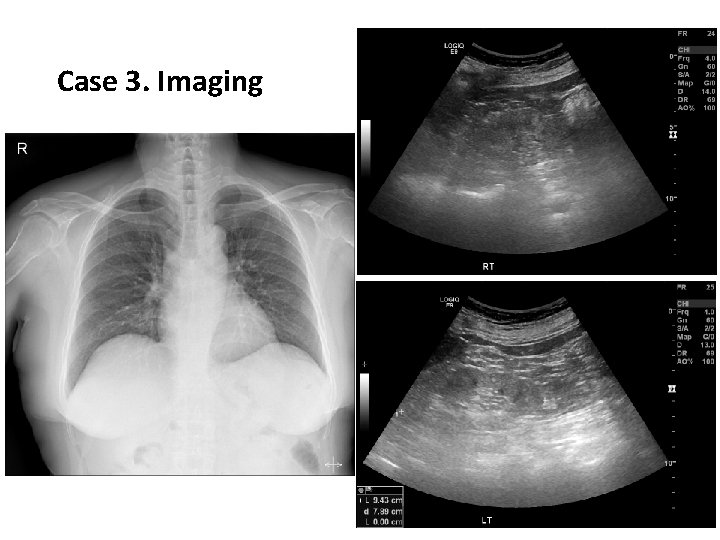
Case 3. Imaging
Case 3. Laboratory data Routine CBC WBC (/mm 3) Blood Chemistry 11, 430 BUN (mg/d. L) 64. 5 Hb (g/d. L) 10. 9 Cr (mg/d. L) 2. 11 Hct (%) 32. 7 e. GFR (m. L/min) 25. 27 280, 000 Glucose (mg/d. L) 99 Platelet (/mm 3) Routine Urine Analysis SG 1. 014 p. H 5. 0 Protein + 24 h Urine Albumin (g/d. L) 4. 34 AST/ALT (IU/L) 22/16 Uric acid (mg/d. L) 5. 6 Ca/P (mg/d. L) 8. 7/4. 4 Mg (mg/d. L) 2. 1 Urine volume (m. L) 2100 Na/K/Cl (m. Eq/L) Protein (mg/day) 596. 4 Total CO 2 (mmol/L) 137/5. 0/112 19. 0

Recommendations for treating the metabolic acidosis of chronic kidney disease (CKD) • Calculate bicarbonate requirement • 50% body weight (kg) x (desired serum bicarbonate concentration – actual serum bicarbonate concentration) • Administer sufficient base to increase serum bicarbonate close to mean reference value of 24 m. Eq/L • Administer dose over 3 -4 days while monitoring serum bicarbonate • When serum bicarbonate concentration reaches target (~24 m. Eq/L), reduce dose of base with goal of maintaining serum bicarbonate at ≤ 24 m. Eq/L Case 3 1. 2. 3. 4. Apparent space of distribution of bicarbonate: 25 L To increase serum bicarbonate to 24 m. Eq/L: requiring 5 m. Eq/L x 25 L = 125 m. Eq of base Administer Na. HCO 3 5 tablets (2500 mg, 30 m. Eq) a day for 4 days Reduce to Na. HCO 3 2 -3 tablets a day to approximate (acid production – acid excretion) Am J Kidney Dis. 2016; 67: 307 -317
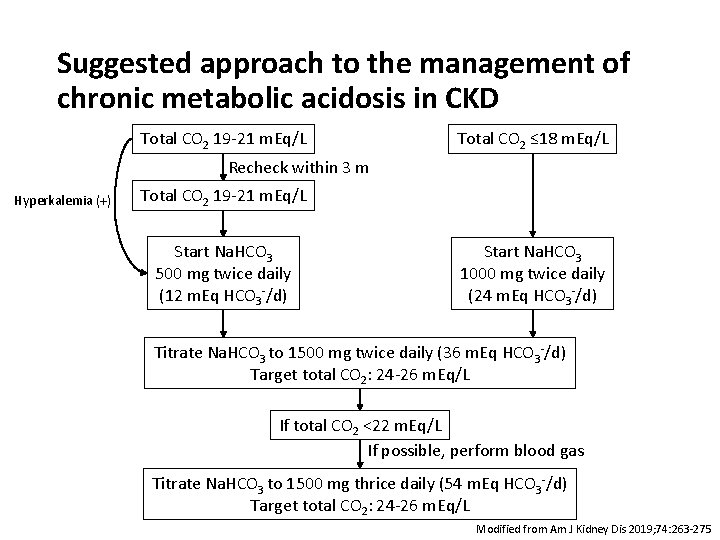
Suggested approach to the management of chronic metabolic acidosis in CKD Total CO 2 19 -21 m. Eq/L Total CO 2 ≤ 18 m. Eq/L Recheck within 3 m Hyperkalemia (+) Total CO 2 19 -21 m. Eq/L Start Na. HCO 3 500 mg twice daily (12 m. Eq HCO 3 -/d) Start Na. HCO 3 1000 mg twice daily (24 m. Eq HCO 3 -/d) Titrate Na. HCO 3 to 1500 mg twice daily (36 m. Eq HCO 3 -/d) Target total CO 2: 24 -26 m. Eq/L If total CO 2 <22 m. Eq/L If possible, perform blood gas Titrate Na. HCO 3 to 1500 mg thrice daily (54 m. Eq HCO 3 -/d) Target total CO 2: 24 -26 m. Eq/L Modified from Am J Kidney Dis 2019; 74: 263 -275

Case 3. Management Na. HCO 3 PO 12 m. Eq/day 6 m. Eq/day 18 m. Eq/day 12 m. Eq/day On Presentation 2 w Later Day 60 Day 120 Na (m. Eq/L) 140 137 140 141 K (m. Eq/L) 5. 0 4. 1 4. 6 Cl (m. Eq/L) 112 112 111 Total CO 2 (mmol/L) 19. 0 19. 1 20. 0 23. 0

Summary • Acute and severe acidemia: IV administration of 50 m. Eq of Na. HCO 3 diluted in 300 m. L of sterile water over 30– 45 min, during the initial 1– 2 h of therapy (despite the controversy). • RTA: replacement with either citrate or Na. HCO 3 tablets with the goal of correcting the serum [HCO 3 –] to normal (with potassium supplementation) • Chronic metabolic acidosis of CKD: oral alkali replacement to maintain the [HCO 3−] to approximately 24 -26 mmol/L
- Slides: 30