How to Measure Pressure Barometer measures atmospheric pressure

How to Measure Pressure Barometer – measures atmospheric pressure vacuum Patm PHg Aneroid Barometer Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
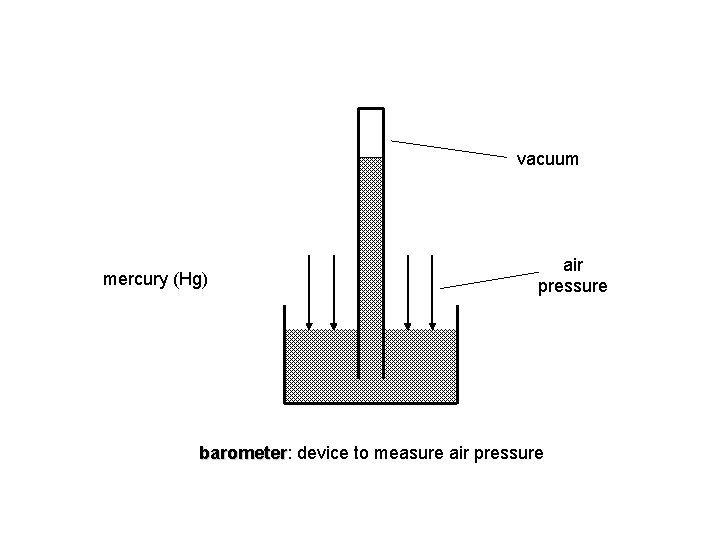
vacuum mercury (Hg) air pressure barometer: barometer device to measure air pressure

Barometer Empty space (a vacuum) Hg Weight of the mercury in the column Weight of the atmosphere (atmospheric pressure) Zumdahl, De. Coste, World of Chemistry 2002, page 401
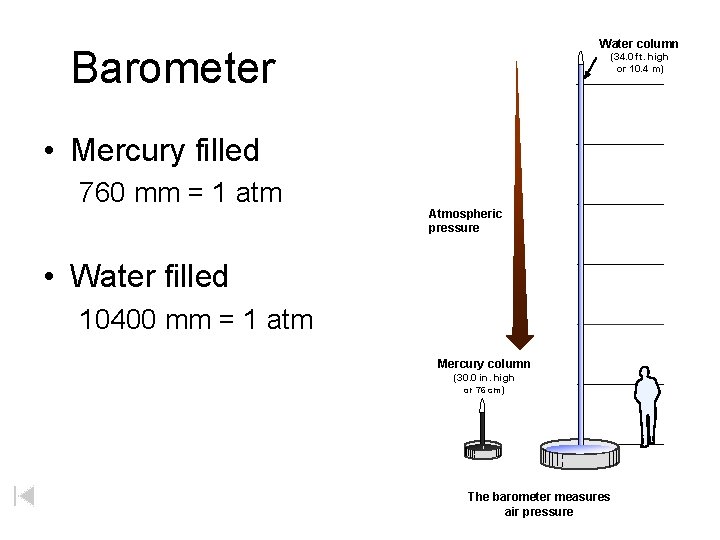
Water column Barometer (34. 0 ft. high or 10. 4 m) • Mercury filled 760 mm = 1 atm Atmospheric pressure • Water filled 10400 mm = 1 atm Mercury column (30. 0 in. high or 76 cm) The barometer measures air pressure
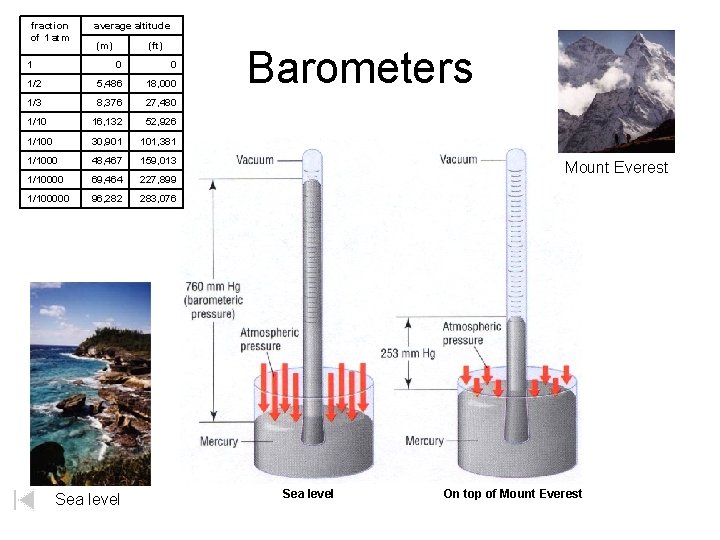
fraction of 1 atm 1 average altitude (m) (ft) 0 0 1/2 5, 486 18, 000 1/3 8, 376 27, 480 1/10 16, 132 52, 926 1/100 30, 901 101, 381 1/1000 48, 467 159, 013 1/10000 69, 464 227, 899 1/100000 96, 282 283, 076 Sea level Barometers Mount Everest Sea level On top of Mount Everest
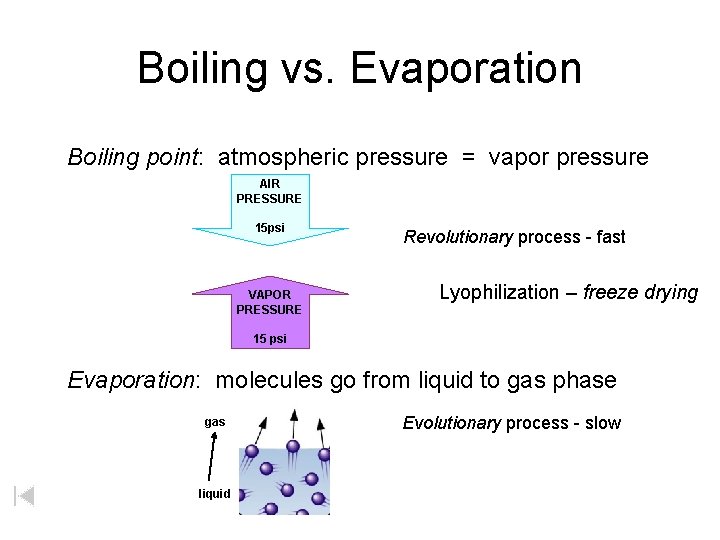
Boiling vs. Evaporation Boiling point: atmospheric pressure = vapor pressure AIR PRESSURE 15 psi VAPOR PRESSURE Revolutionary process - fast Lyophilization – freeze drying 15 psi Evaporation: molecules go from liquid to gas phase gas liquid Evolutionary process - slow
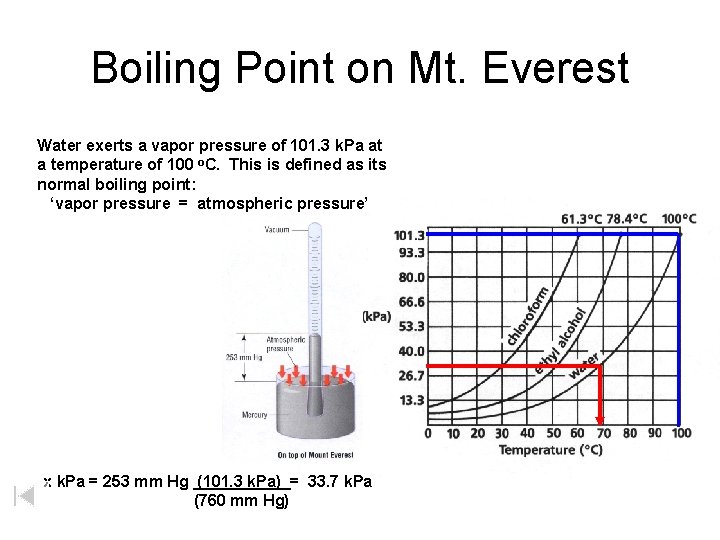
Boiling Point on Mt. Everest Water exerts a vapor pressure of 101. 3 k. Pa at a temperature of 100 o. C. This is defined as its normal boiling point: ‘vapor pressure = atmospheric pressure’ x k. Pa = 253 mm Hg (101. 3 k. Pa) = 33. 7 k. Pa (760 mm Hg)
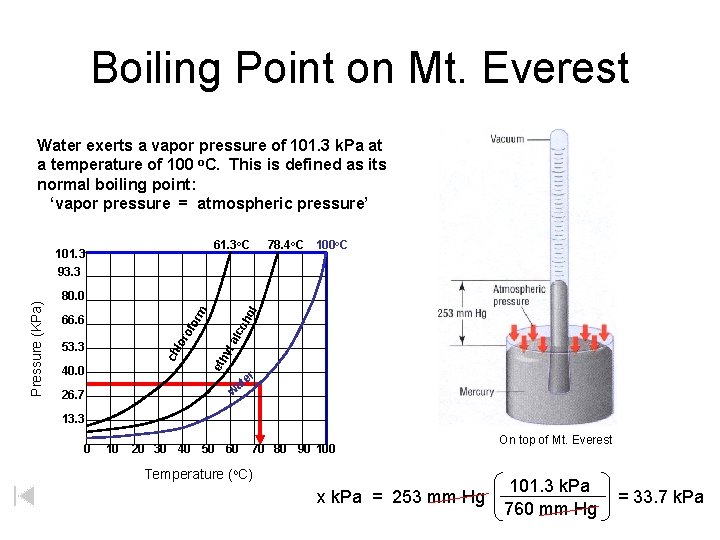
Boiling Point on Mt. Everest Water exerts a vapor pressure of 101. 3 k. Pa at a temperature of 100 o. C. This is defined as its normal boiling point: ‘vapor pressure = atmospheric pressure’ 61. 3 o. C 78. 4 o. C 100 o. C 40. 0 oh alc 53. 3 eth yl lor of or 66. 6 ol m 80. 0 ch Pressure (KPa) 101. 3 93. 3 er at w 26. 7 13. 3 0 10 20 30 40 50 60 70 80 90 100 Temperature (o. C) x k. Pa = 253 mm Hg On top of Mt. Everest 101. 3 k. Pa 760 mm Hg = 33. 7 k. Pa
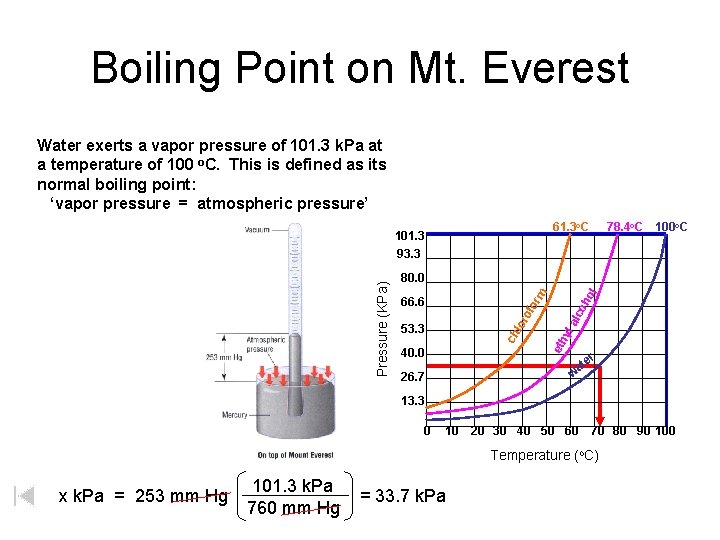
Boiling Point on Mt. Everest Water exerts a vapor pressure of 101. 3 k. Pa at a temperature of 100 o. C. This is defined as its normal boiling point: ‘vapor pressure = atmospheric pressure’ 61. 3 o. C 78. 4 o. C 100 o. C 40. 0 oh alc 53. 3 eth yl lor of or 66. 6 ol m 80. 0 ch Pressure (KPa) 101. 3 93. 3 er at w 26. 7 13. 3 0 10 20 30 40 50 60 70 80 90 100 Temperature (o. C) x k. Pa = 253 mm Hg 101. 3 k. Pa 760 mm Hg = 33. 7 k. Pa
- Slides: 9