How To Make Biological Molecules Macromolecules Smaller organic
How To Make Biological Molecules

Macromolecules • Smaller organic molecules join together to form larger molecules – macromolecules • 4 major classes of macromolecules: – carbohydrates – lipids – proteins – nucleic acids
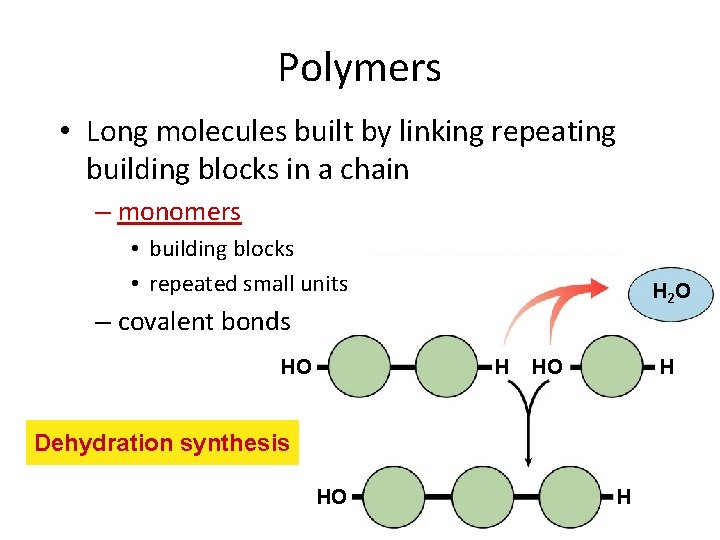
Polymers • Long molecules built by linking repeating building blocks in a chain – monomers • building blocks • repeated small units H 2 O – covalent bonds HO H Dehydration synthesis HO H
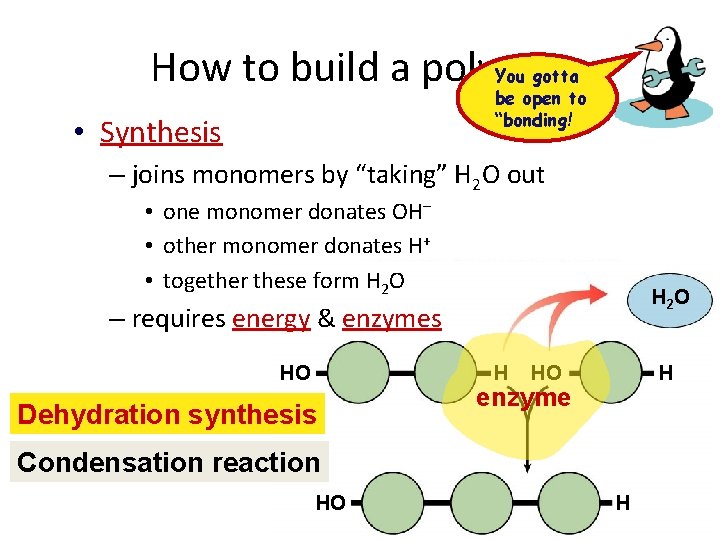
How to build a polymer You gotta be open to “bonding! • Synthesis – joins monomers by “taking” H 2 O out • one monomer donates OH– • other monomer donates H+ • together these form H 2 O H 2 O – requires energy & enzymes HO H Dehydration synthesis HO H enzyme Condensation reaction HO H
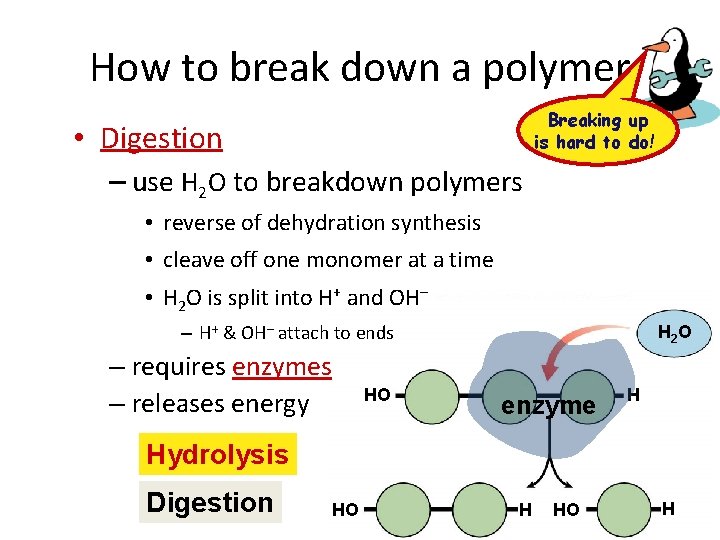
How to break down a polymer Breaking up is hard to do! • Digestion – use H 2 O to breakdown polymers • reverse of dehydration synthesis • cleave off one monomer at a time • H 2 O is split into H+ and OH– – H+ & OH– attach to ends – requires enzymes – releases energy HO H 2 O enzyme H Hydrolysis Digestion HO H

Any Questions? ? 2007 -2008

CH 2 OH H HO O H OH Carbohydrates energy molecules
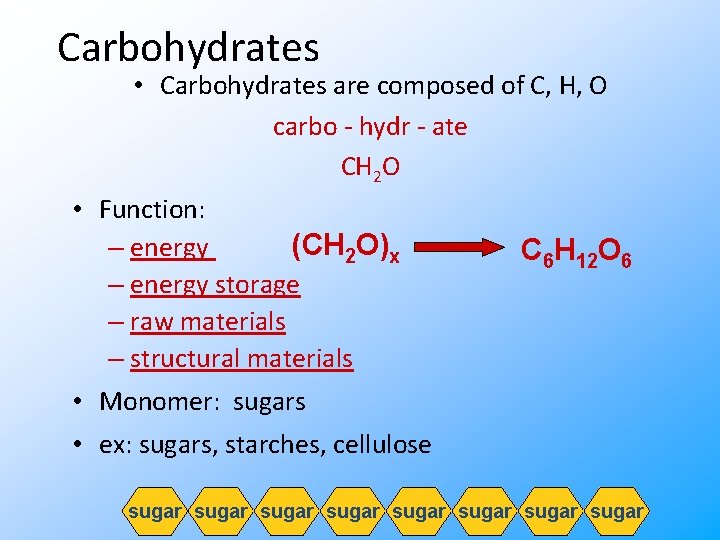
Carbohydrates • Carbohydrates are composed of C, H, O carbo - hydr - ate CH 2 O • Function: (CH 2 O)x – energy storage – raw materials – structural materials • Monomer: sugars C 6 H 12 O 6 • ex: sugars, starches, cellulose sugar sugar
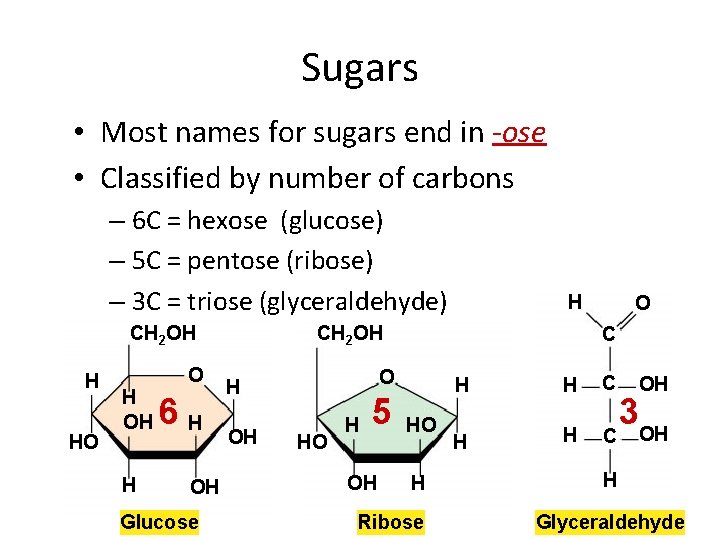
Sugars • Most names for sugars end in -ose • Classified by number of carbons – 6 C = hexose (glucose) – 5 C = pentose (ribose) – 3 C = triose (glyceraldehyde) CH 2 OH H HO O H OH H 6 H OH Glucose H CH 2 OH OH C O H HO H 5 OH O HO H Ribose H H C OH 3 OH H Glyceraldehyde
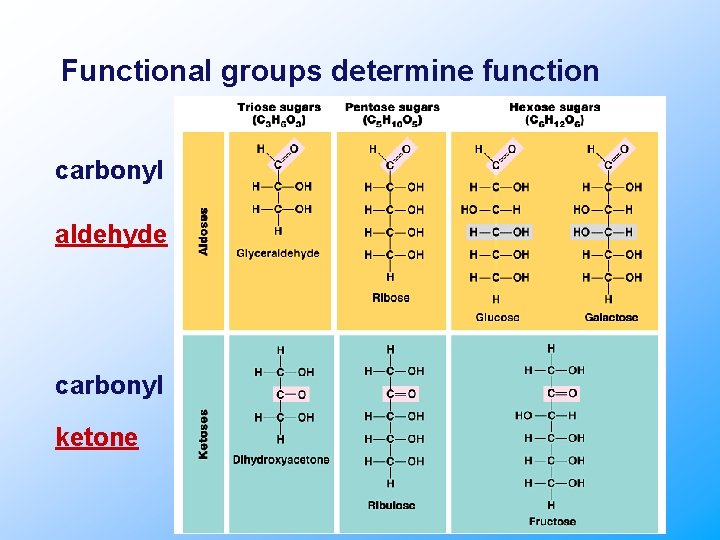
Functional groups determine function carbonyl aldehyde carbonyl ketone
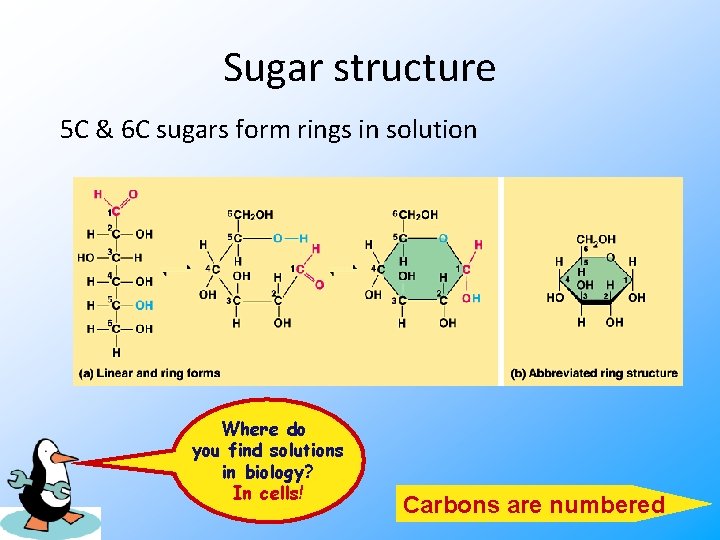
Sugar structure 5 C & 6 C sugars form rings in solution Where do you find solutions in biology? In cells! Carbons are numbered
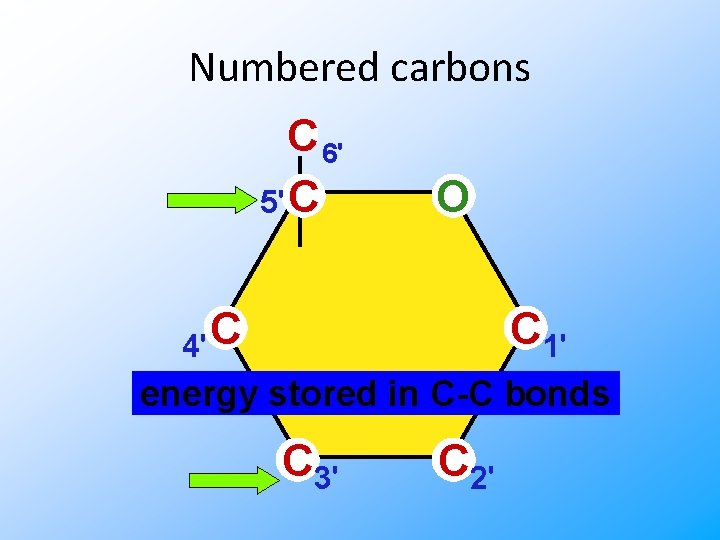
Numbered carbons C 6' 5' C O C 4' C 1' energy stored in C-C bonds C 3' C 2'

Simple & complex sugars CH 2 OH H • Monosaccharides – simple 1 monomer sugars – glucose • Disaccharides – 2 monomers – sucrose • Polysaccharides – large polymers – starch HO O H OH H H OH Glucose H OH
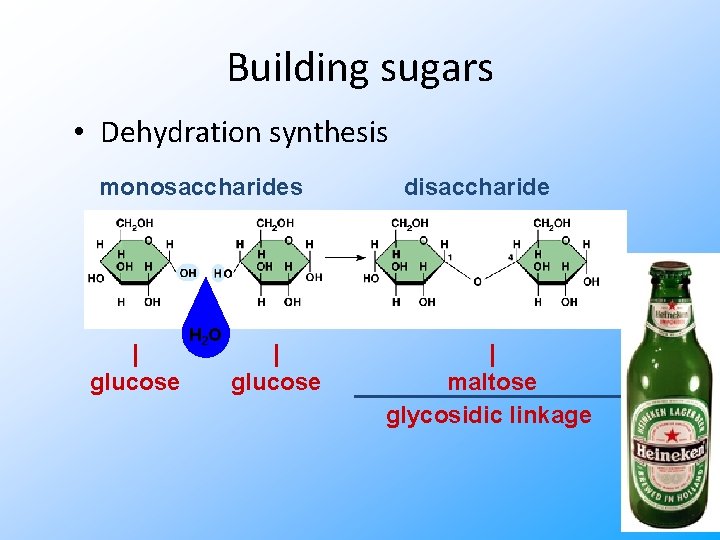
Building sugars • Dehydration synthesis monosaccharides | glucose H 2 O | glucose disaccharide | maltose glycosidic linkage
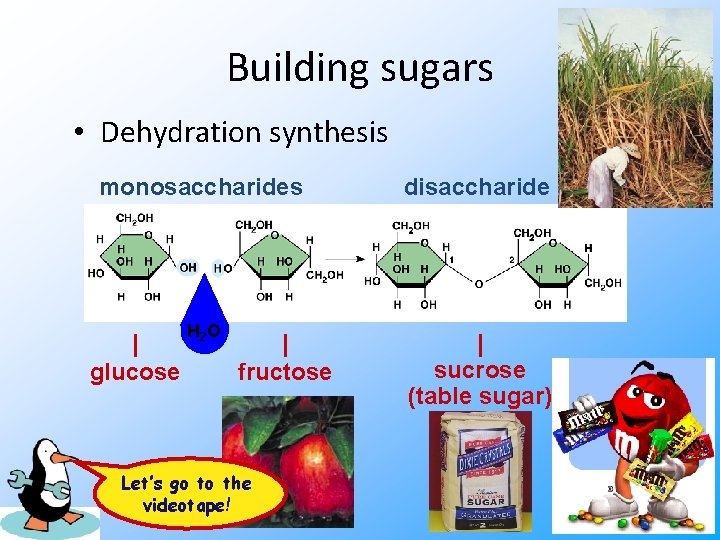
Building sugars • Dehydration synthesis monosaccharides disaccharide H 2 O | sucrose (table sugar) | glucose | fructose Let’s go to the videotape!

Polysaccharides • Polymers of sugars – costs little energy to build – easily reversible = release energy • Function: – energy storage • starch (plants) • glycogen (animals) – in liver & muscles – structure • cellulose (plants) • chitin (arthropods & fungi)

Linear vs. branched polysaccharides slow release starch (plant) energy storage What does branching do? glycogen (animal) fast release Let’s go to the videotape!
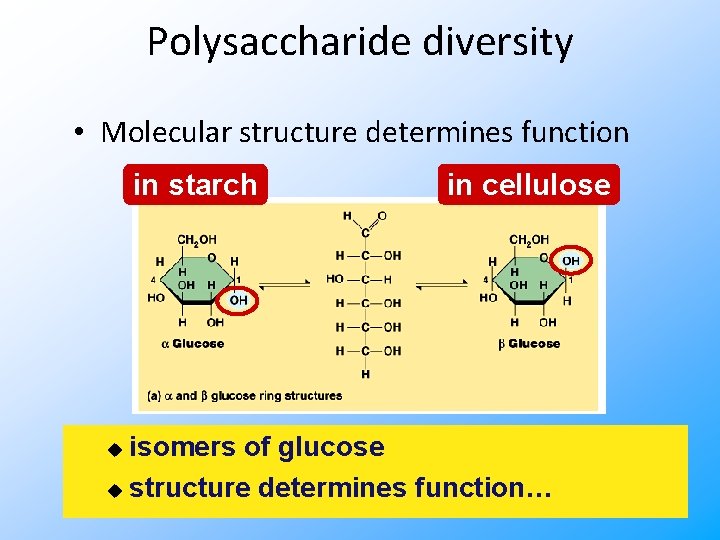
Polysaccharide diversity • Molecular structure determines function in starch in cellulose isomers of glucose u structure determines function… u
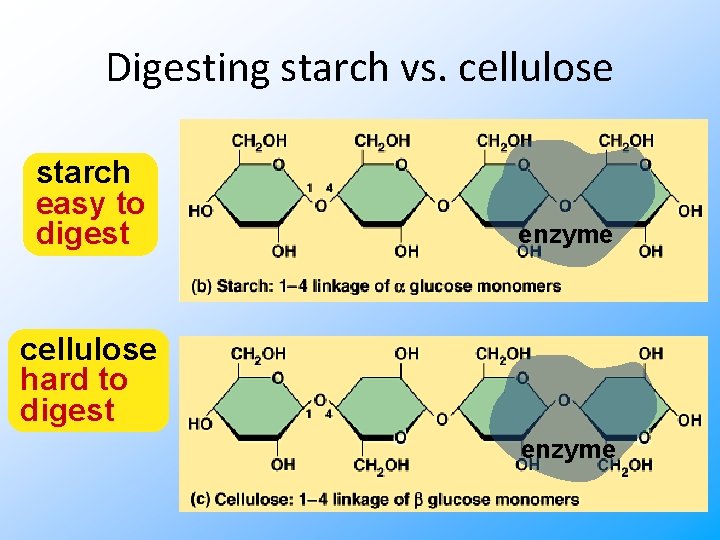
Digesting starch vs. cellulose starch easy to digest enzyme cellulose hard to digest enzyme

Cellulose • Most abundant organic compound on Earth – herbivores have evolved a mechanism to digest cellulose – most carnivores have not • that’s why they eat meat to get their energy & nutrients • cellulose = undigestible roughage But it tastes like hay! Who can live on this stuff? !

Chitin, a different structural polysaccharide H OH CH 2 OH O OH H H H NH C O CH 3 (a) The structure of the chitin monomer. (b) Chitin forms the exoskeleton of arthropods. This cicada is molting, shedding its old exoskeleton and emerging in adult form. It is also found in Fungal Cell Walls. (c) Chitin is used to make a strong and flexible surgical thread that decomposes after the wound or incision heals.

Cow can digest cellulose well; no need to eat other sugars Gorilla can’t digest cellulose well; must add another sugar source, like fruit to diet
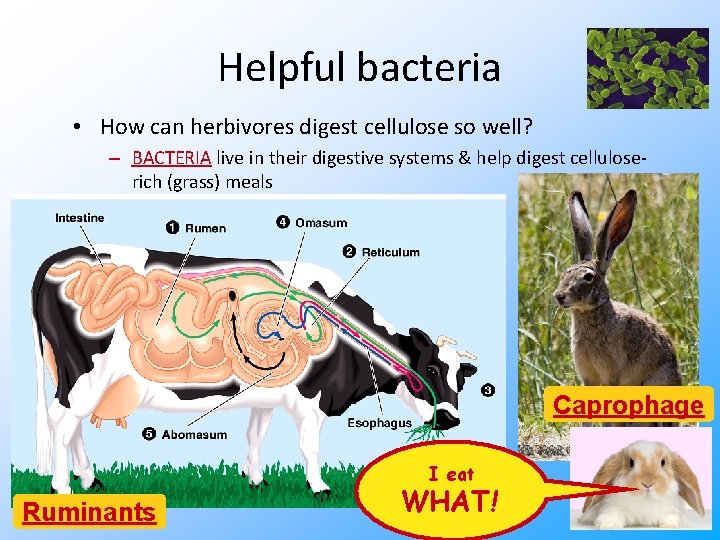
Helpful bacteria • How can herbivores digest cellulose so well? – BACTERIA live in their digestive systems & help digest celluloserich (grass) meals Caprophage Ruminants Tell Ime about eat the rabbits, WHAT! again, George!

EAT Let’s build X some Carbohydrates! 2006 -2007

Review Questions
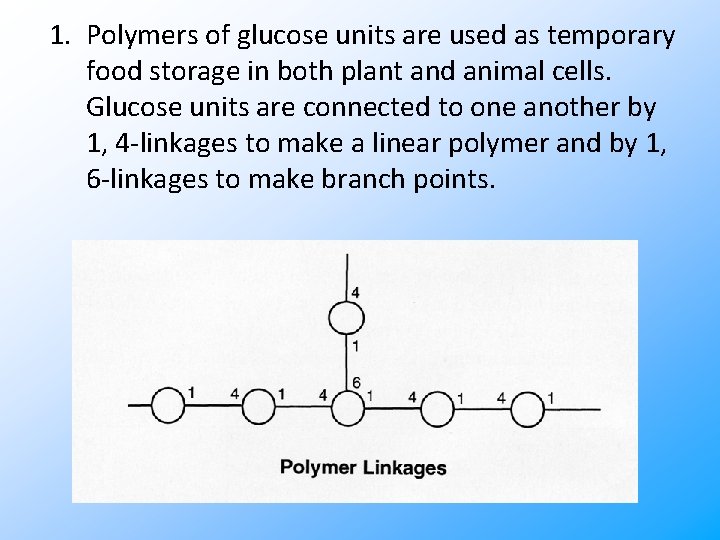
1. Polymers of glucose units are used as temporary food storage in both plant and animal cells. Glucose units are connected to one another by 1, 4 -linkages to make a linear polymer and by 1, 6 -linkages to make branch points.
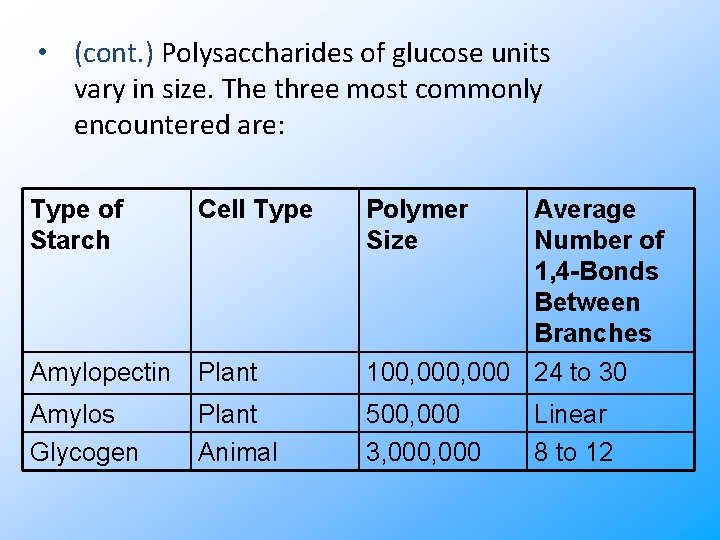
• (cont. ) Polysaccharides of glucose units vary in size. The three most commonly encountered are: Type of Starch Cell Type Polymer Size Amylopectin Plant Average Number of 1, 4 -Bonds Between Branches 100, 000 24 to 30 Amylos Glycogen Plant Animal 500, 000 3, 000 Linear 8 to 12

• (cont. ) When each polymer bond is made, a water molecule is released and becomes part of the cell water. How many water molecules were released during formation of each of the Glycogen? A. B. C. D. E. 1, 000, 000 2, 666 3, 000 3, 300, 000

2. Which of the following is a polymer? A. Simple sugar (aka monosaccharide) B. Carbon atoms C. Glucose D. Cellulose E. deoxyribose

3. Starch and Glycogen both I. Serve as energy storage for organisms II. Provide structure and support III. Are structural isomers of glucose A. I only B. II only C. I and II only D. I and III only E. I, II, and III
- Slides: 30