How to Interpret Titration Curves find the equivalence








- Slides: 15

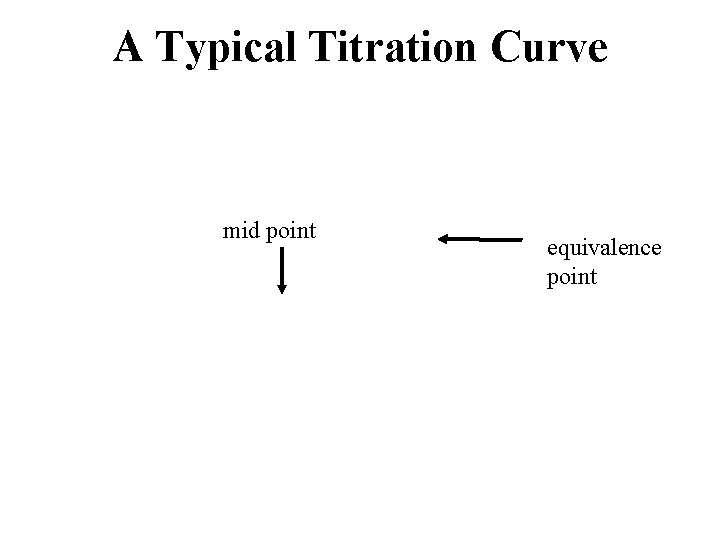
How to Interpret Titration Curves • find the equivalence point – it is the steepest part of the curve where the p. H rises the fastest – the equivalence point can be used to determine the equivalent weight (molar mass) of the acid • find the mid point – located in the center of the buffer region – geometrically halfway between the equivalence point and the beginning of the titration – sometimes it is a little more complicated than this - see the example – the midpoint determines the p. Ka of the acid

How to Interpret Titration Curves • things to do first – graph your data as seen in the next slide – make sure you turn on the major and minor tick marks on both axes • double click on the axis and click on the “patterns” tab) – there is enough precision in the tick marks • you should have at least 1 m. L or smaller for the minor tick mark on the x-axis • you should have at least 0. 2 p. H units or smaller for the minor tick mark on the y-axis

Two Different Methods • there are two methods of analysis that will be shown – geometric method • requires a ruler, a pencil, and the titration graph – 1 st derivative method • requires a spreadsheet and some formula entries • gives you cool graphs with the 1 st derivative pointing to the equivalence and mid points • scores you brownie points with the instructors • pick your method (either will work)

A Typical Titration Curve mid point equivalence point

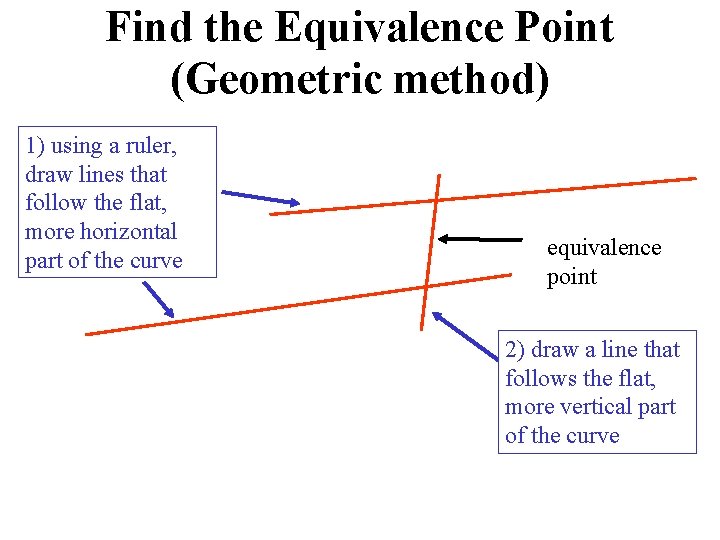
Find the Equivalence Point (Geometric method) 1) using a ruler, draw lines that follow the flat, more horizontal part of the curve equivalence point 2) draw a line that follows the flat, more vertical part of the curve

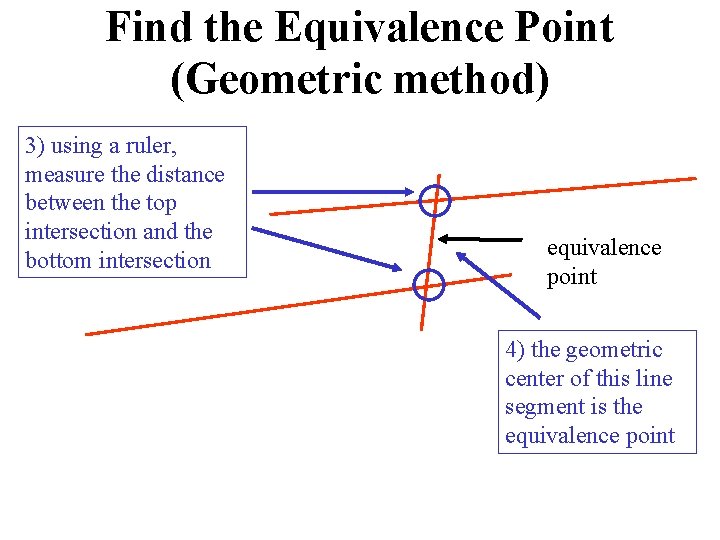
Find the Equivalence Point (Geometric method) 3) using a ruler, measure the distance between the top intersection and the bottom intersection equivalence point 4) the geometric center of this line segment is the equivalence point

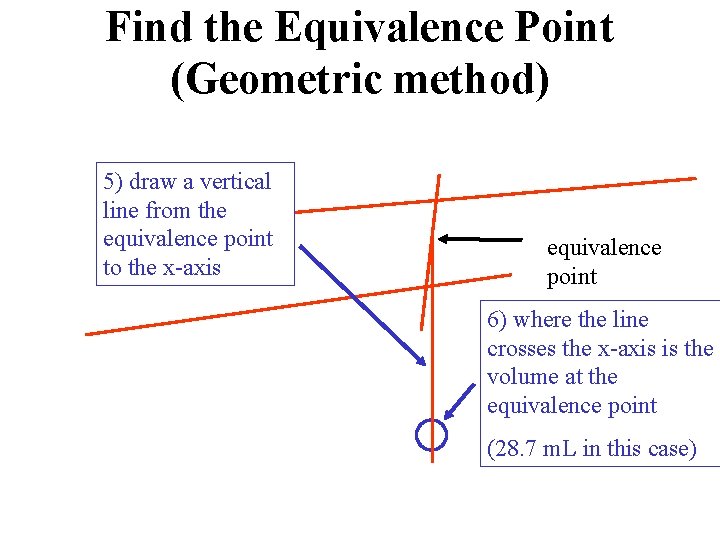
Find the Equivalence Point (Geometric method) 5) draw a vertical line from the equivalence point to the x-axis equivalence point 6) where the line crosses the x-axis is the volume at the equivalence point (28. 7 m. L in this case)

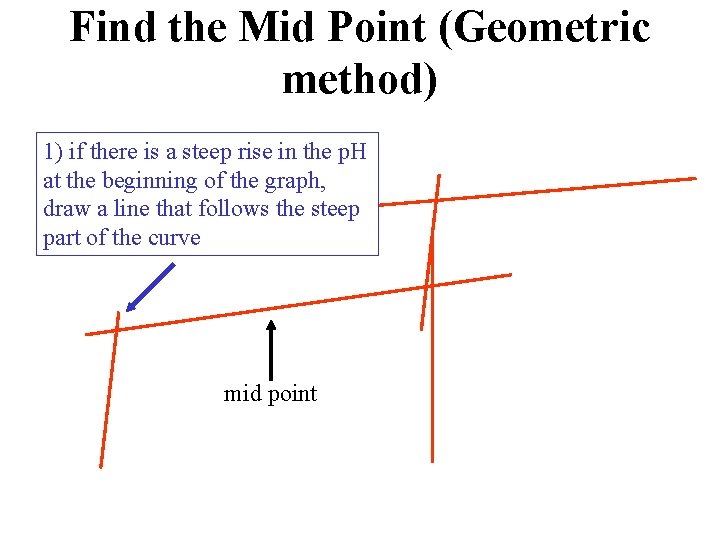
Find the Mid Point (Geometric method) 1) if there is a steep rise in the p. H at the beginning of the graph, draw a line that follows the steep part of the curve mid point

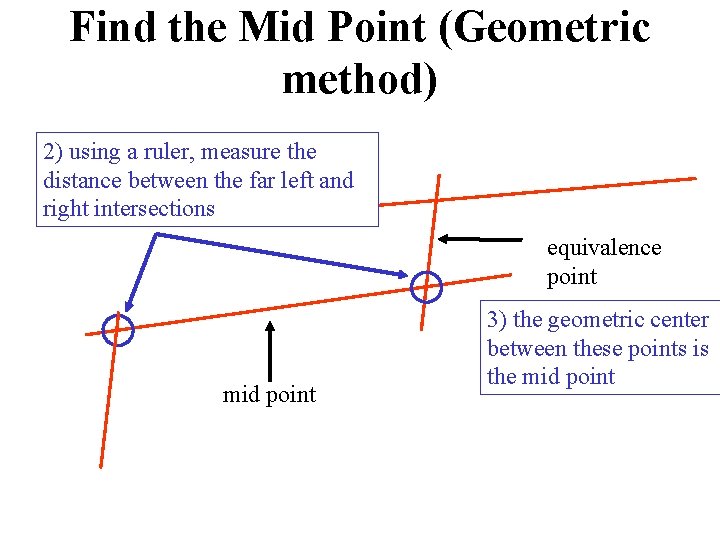
Find the Mid Point (Geometric method) 2) using a ruler, measure the distance between the far left and right intersections equivalence point mid point 3) the geometric center between these points is the mid point

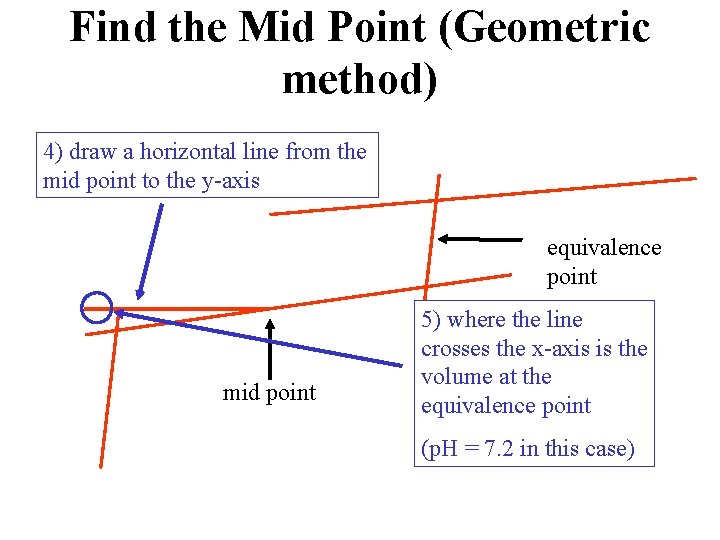
Find the Mid Point (Geometric method) 4) draw a horizontal line from the mid point to the y-axis equivalence point mid point 5) where the line crosses the x-axis is the volume at the equivalence point (p. H = 7. 2 in this case)

How to Interpret Titration Curves • find the equivalence point – make sure you subtract the initial buret volume! – in this case, the initial buret volume was 1. 07 m. L – true equiv. pt. = 28. 7 m. L - 1. 07 m. L = 27. 63 m. L – the 3 is the indicate the limit of the significant figures • calculate the equivalent weight (molar mass) – equiv. wt. = (acid mass)/[(Na. OH conc)(equiv. pt. )] – equiv. wt. = (430. 2 mg)/[(0. 1139 M)(27. 63 m. L)] – equiv. wt. = 136. 699 = 137 g/mol

How to Interpret Titration Curves • find the mid point – mid pt = 7. 2 = p. Ka of the acid

For you Excel Aficionados • equivalence point – use the first derivative d p. H / d Vol – the spike in the graph points to the equiv. pt. • mid point – reverse the axes for the p. H curve • x axis = p. H values; y-axis = Vol values – use the first derivative d Vol / d p. H – the spike in the graph points to the mid point • use extra columns in the spreadsheet to make these calcs – 1 st deriv. (d p. H / d Vol) = (p. H 2 - p. H 1)/(Vol 2 - Vol 1) – 1 st deriv. (d Vol / d p. H) = (Vol 2 - Vol 1)/ (p. H 2 - p. H 1) • or just (the first 1 st deriv)-1

Find the Equivalence Point (derivative method) 1)1)identifyvolume valueatatthethepeak (28. 5 m. L in this case)

Find the Mid Point (derivative method) 1) identify p. H value at the peak (p. H = 7. 3 in this case)