How to establish a dosage regimen for a

How to establish a dosage regimen for a sustainable use of antibiotics in veterinary medicine Pierre-Louis Toutain, Ecole Nationale Vétérinaire INRA & National veterinary School of Toulouse, France Wuhan 09/10/2015 1

EMEA "Points to consider" July 2000 • Inadequate dosing of antibiotics is probably an important reason for misuse and subsequent risk of resistance 2

EMA/CVMP 2015 3

EMA guideline 2015 4

The dosage regimen 5

The three endpoints to consider in veterinary medicine • Efficacy in animal • No promotion of resistance in animal (target pathogen) • No promotion of resistance in man 6

What are the elements of a dosage regimen • The dose • The dosing interval • The treatment duration –When to start –When to finish 7
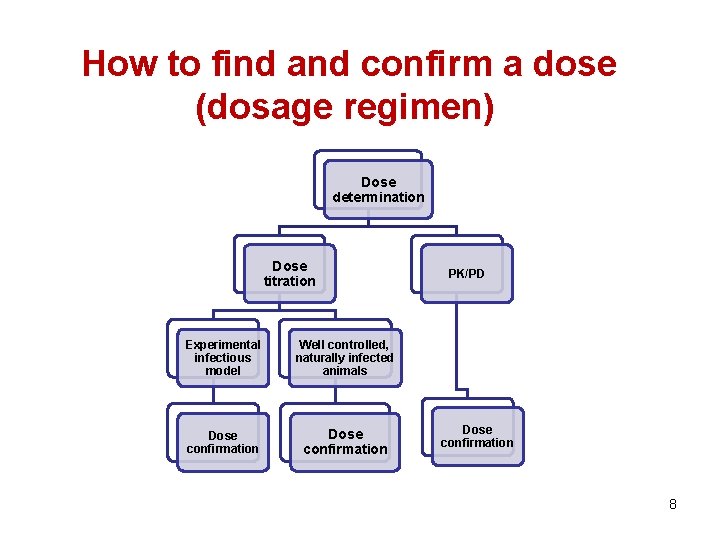
How to find and confirm a dose (dosage regimen) Dose determination Dose titration Experimental infectious model Well controlled, naturally infected animals Dose confirmation PK/PD Dose confirmation 8

The dose-titration ECVPT Toulouse 2009 9
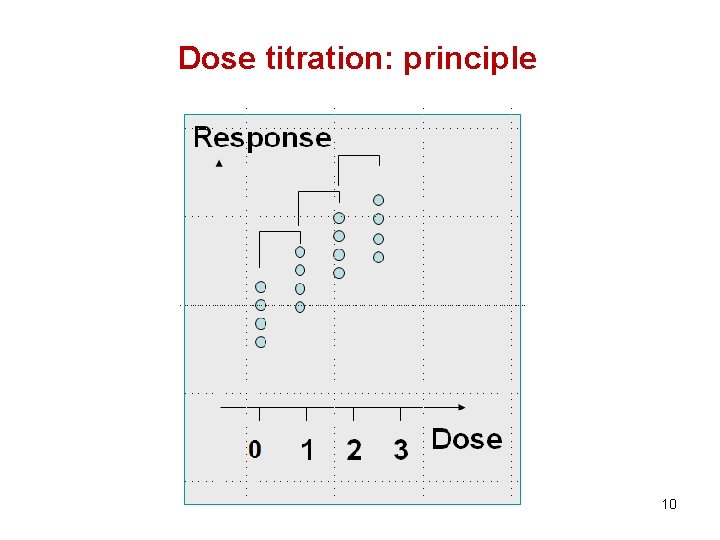
Dose titration: principle 10

Dose titration require: an infectious model • Where possible, experimentally induced infections should be used in the dose-determination studies – If no experimental model is available and study conditions are well controlled, naturally infected animals can be used 11

Endpoints • Efficacy evaluation should be based on clinical and bacteriological response as determined by appropriate clinical and bacteriological assessment 12

Dose titration: design • Usually three levels of dosage of the veterinary medicinal product should be tested, preferably using the final formulation. – Control group (dose=0) compulsory 13
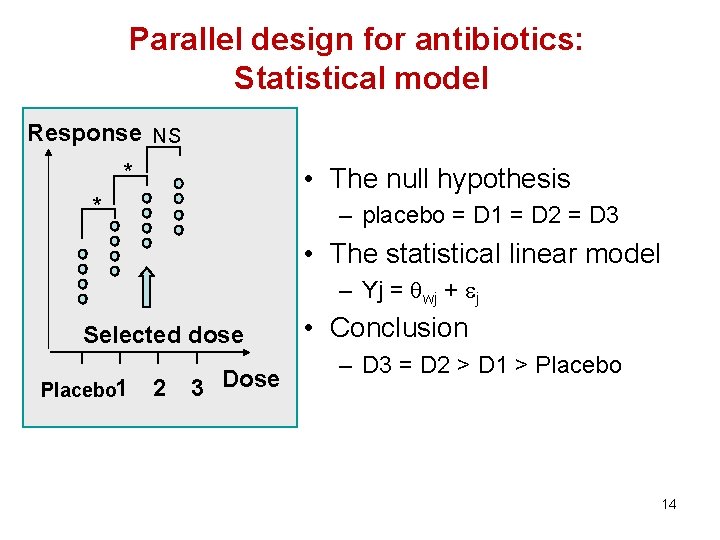
Parallel design for antibiotics: Statistical model Response NS * • The null hypothesis * – placebo = D 1 = D 2 = D 3 • The statistical linear model – Yj = wj + j Selected dose Placebo 1 2 3 Dose • Conclusion – D 3 = D 2 > D 1 > Placebo 14

Dose titration: statistics • Statistical comparisons between different treatment groups and the negative control group • it is acknowledged that dose determination lack of power 15

The parallel design • Advantages – easy to execute – total study lasts over one period – approved by Authorities • Disadvantages – "local information" (response at a given dose does not provide any information about another dose) – no information about the distribution of the individual patient's dose response.

Dose titration & PK/PD • Dose titration can be used to establish the critical value of the PK/PD index 17
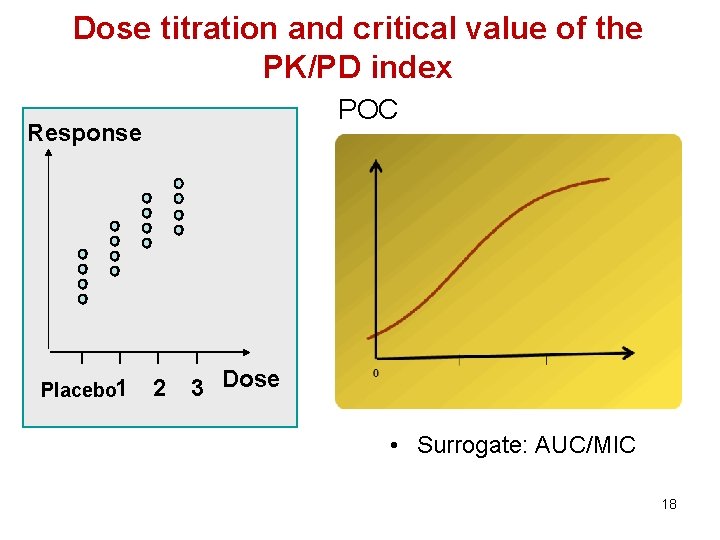
Dose titration and critical value of the PK/PD index POC Response Placebo 1 2 3 Dose • Surrogate: AUC/MIC 18

Probability of cure (POC) • Logistic regression can be used to link measures of drug exposure (AUC/MIC) to the probability of a clinical success Dependent variable Placebo effect sensitivity Independent variable 2 parameters: a (placebo effect) & b (slope of the exposure-effect curve) 19

Dose determination using a PK/PD approach 2

It has been developed surrogates indices (predictors) of antibiotic efficacy taking into account MIC (PD) and exposure antibiotic metrics (PK) Practically, 3 indices cover all situations: • AUC/MIC • Time>MIC • Cmax/MIC 21

EMA and the PK/PD approach • PK/PD relationship may be used to support dose regimen selection • In circumstances in which it is not feasible to generate extensive clinical efficacy data (e. g. in rare types of infections or against rare types of pathogens, including multidrug resistant pathogens that are rarely encountered) PK/PD analyses may also provide important supportive information on the potential efficacy of the test antibacterial agent. 22

EMA and the PK/PD approach • It is acknowledged that the PK/PD analyses will be based on PK data obtained from healthy or experimentally infected animals. Nevertheless, the sponsor is encouraged to collect PK data from naturally diseased animals using population kinetic models. • Knowledge of kinetic variability considerably increases the value of the PK/PD analysis 23

Demonstration of applicability of PK/PD concepts to determine a dosage regimen for tulathromycin in the calf 24
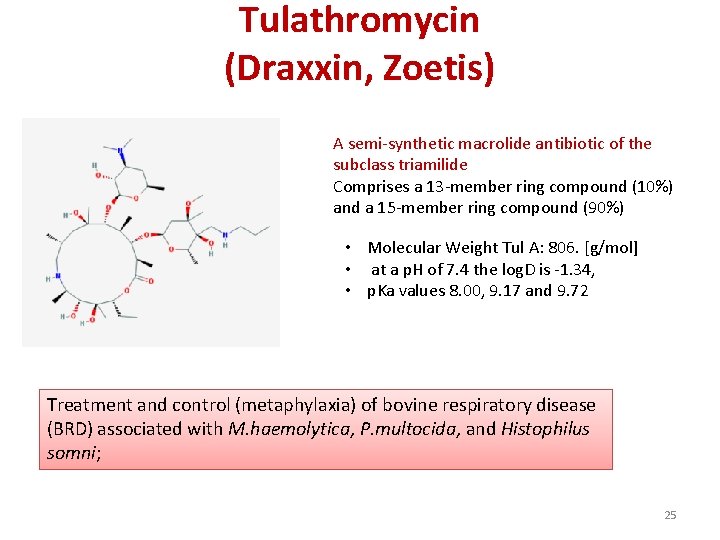
Tulathromycin (Draxxin, Zoetis) A semi-synthetic macrolide antibiotic of the subclass triamilide Comprises a 13 -member ring compound (10%) and a 15 -member ring compound (90%) • Molecular Weight Tul A: 806. [g/mol] • at a p. H of 7. 4 the log. D is -1. 34, • p. Ka values 8. 00, 9. 17 and 9. 72 Treatment and control (metaphylaxia) of bovine respiratory disease (BRD) associated with M. haemolytica, P. multocida, and Histophilus somni; 25
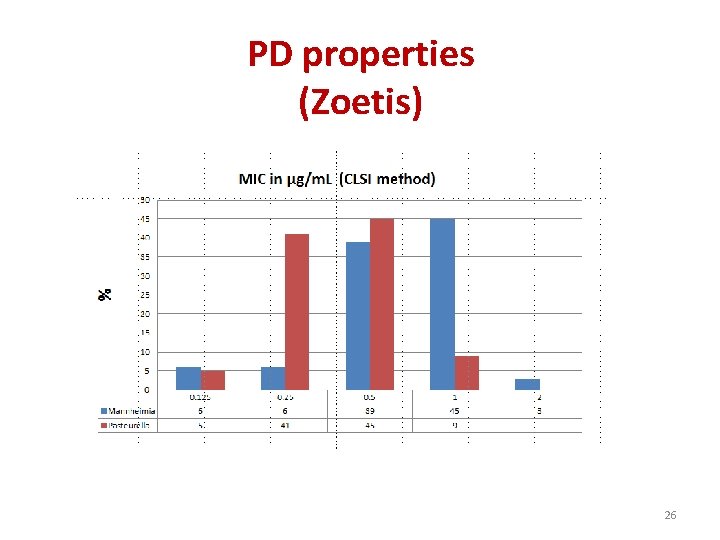
PD properties (Zoetis) 26
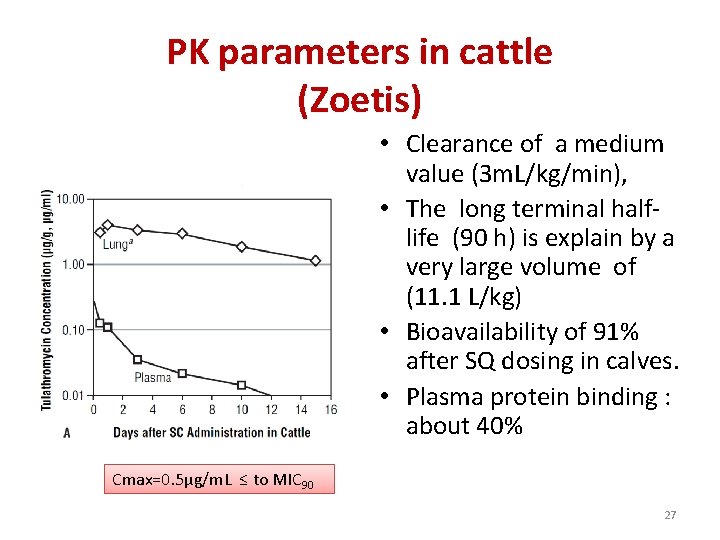
PK parameters in cattle (Zoetis) • Clearance of a medium value (3 m. L/kg/min), • The long terminal halflife (90 h) is explain by a very large volume of (11. 1 L/kg) • Bioavailability of 91% after SQ dosing in calves. • Plasma protein binding : about 40% Cmax=0. 5µg/m. L ≤ to MIC 90 27

The dose was determined in clinics • 1. 25, 2. 5 and 5 mg/kg were tested – 1. 25 mg/kg: success of 76. 9% – 2. 5 mg/kg: success of 86. 8% – 5. 0 mg/kg : no additional benefit 28
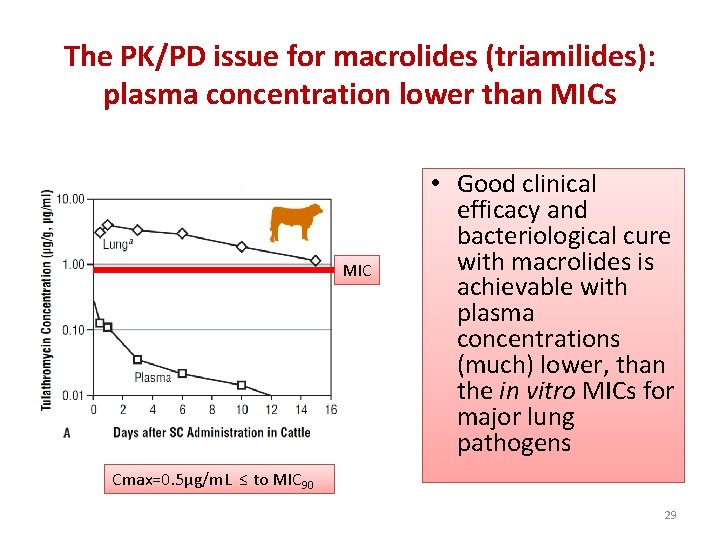
The PK/PD issue for macrolides (triamilides): plasma concentration lower than MICs MIC • Good clinical efficacy and bacteriological cure with macrolides is achievable with plasma concentrations (much) lower, than the in vitro MICs for major lung pathogens Cmax=0. 5µg/m. L ≤ to MIC 90 29

The issue for macrolides (triamilides): • PK/PD concepts to macrolides has been challenged rather than the validity of the in vitro MIC data obtained in matrices optimised for bacterial growth, as in Mueller Hinton Broth (MHB). – This has led some authors and Authorities to claim that there is no plasma concentrationeffect relationship for macrolides. 30

Aims of the study • To generate the appropriate PK and PD data for tulathromycin for M. haemolytica and P. multocida in calves to show that it is possible to establish therapeutically relevant in vivo PK/PD relationships for a macrolide as for any AMD 31

In vitro investigations 32
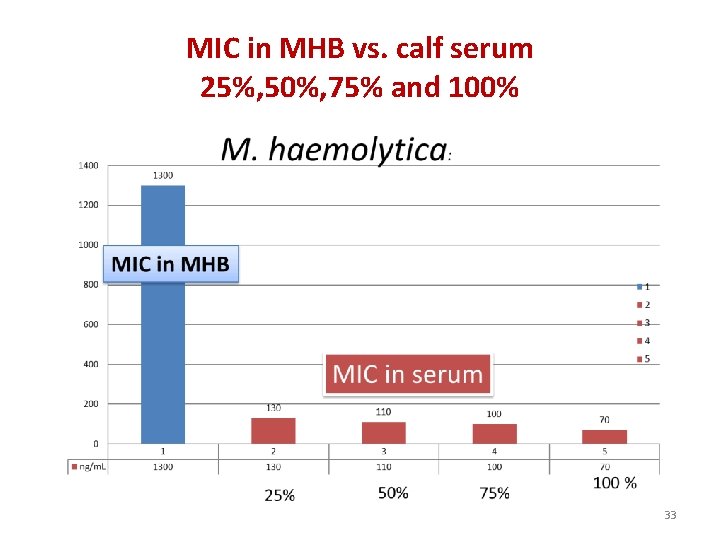
MIC in MHB vs. calf serum 25%, 50%, 75% and 100% 33

Determination of a scaling factor between MHB and serum • Minimum Inhibitory Concentrations (MIC) were approximately 50 times lower in calf serum than in Mueller Hinton Broth. • A scaling factor of 50 was used to transform epidemiological data (MIC) into relevant MIC concentration for a dosage regimen determination 34

The serum effect For azithromycin (closely related to tulathromycin) the presence of 40% serum during the MIC test decreased MICs by 26 -fold for serum-resistant Escherichia coli and 15 -fold for Staphylococcus aureus. 35

What PK/PD index to select? • Time>MIC or AUC/MIC? • Semi-mechanistic model predict that when the half-life is short, the best predictor is always T>MIC and when the half-life is long, the best predictor is always AUC/MIC whatever the antibiotic. • We used AUC/MIC 36

What is the size of the PK/PD index (AUC/MIC) for tulathromycin 37

Time-kill studies 38
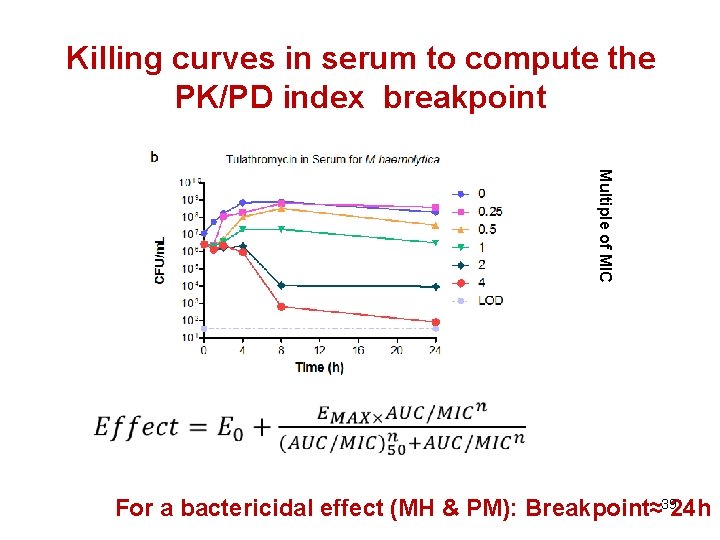
Killing curves in serum to compute the PK/PD index breakpoint Multiple of MIC For a bactericidal effect (MH & PM): Breakpoint≈3924 h

Data analysis • 40
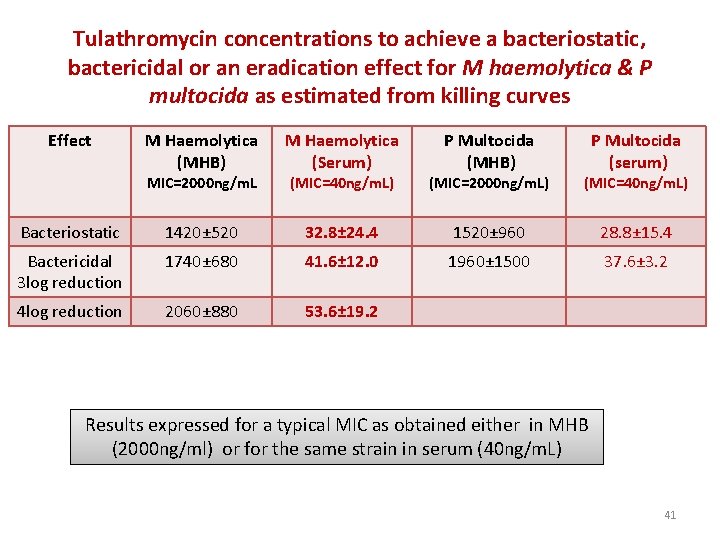
Tulathromycin concentrations to achieve a bacteriostatic, bactericidal or an eradication effect for M haemolytica & P multocida as estimated from killing curves Effect M Haemolytica (MHB) M Haemolytica (Serum) (MIC=40 ng/m. L) (MIC=2000 ng/m. L) (MIC=40 ng/m. L) Bacteriostatic 1420± 520 32. 8± 24. 4 1520± 960 28. 8± 15. 4 Bactericidal 3 log reduction 1740± 680 41. 6± 12. 0 1960± 1500 37. 6± 3. 2 4 log reduction 2060± 880 53. 6± 19. 2 MIC=2000 ng/m. L P Multocida (MHB) P Multocida (serum) Results expressed for a typical MIC as obtained either in MHB (2000 ng/ml) or for the same strain in serum (40 ng/m. L) 41

In vivo investigation 42

PK investigations 43
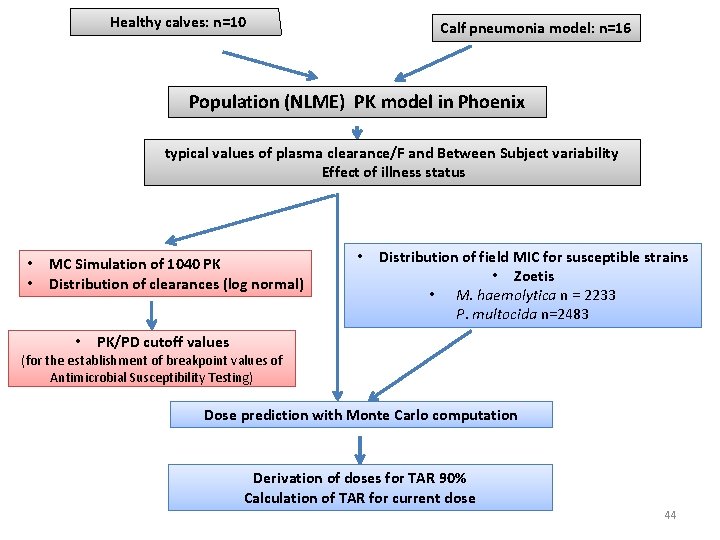
Healthy calves: n=10 Calf pneumonia model: n=16 Population (NLME) PK model in Phoenix typical values of plasma clearance/F and Between Subject variability Effect of illness status • • MC Simulation of 1040 PK Distribution of clearances (log normal) • • Distribution of field MIC for susceptible strains • Zoetis • M. haemolytica n = 2233 P. multocida n=2483 PK/PD cutoff values (for the establishment of breakpoint values of Antimicrobial Susceptibility Testing) Dose prediction with Monte Carlo computation Derivation of doses for TAR 90% Calculation of TAR for current dose 44
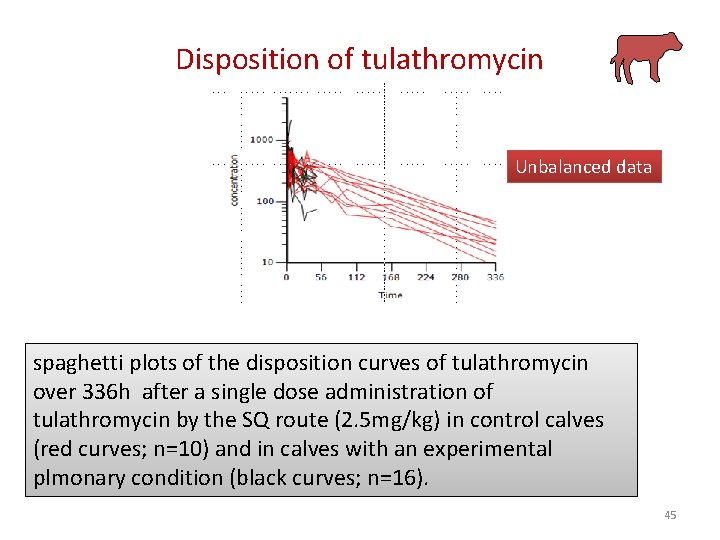
Disposition of tulathromycin Unbalanced data spaghetti plots of the disposition curves of tulathromycin over 336 h after a single dose administration of tulathromycin by the SQ route (2. 5 mg/kg) in control calves (red curves; n=10) and in calves with an experimental plmonary condition (black curves; n=16). 45
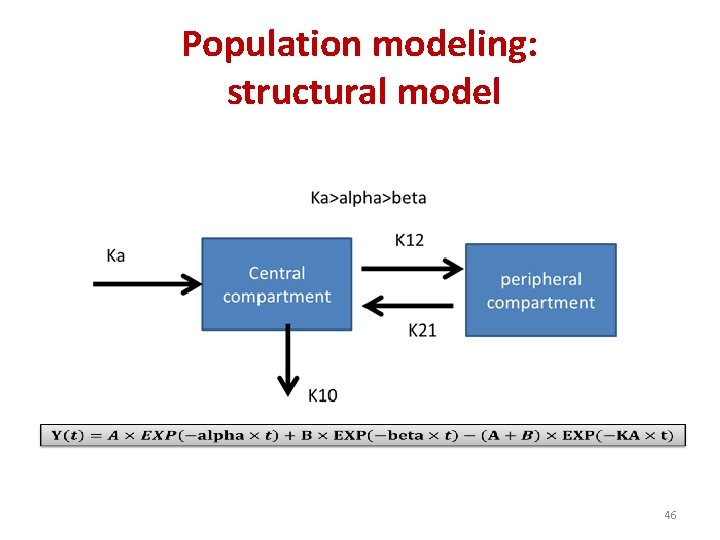
Population modeling: structural model 46

The Between subject variability (BSV) was modeled using an exponential model 47

Model of the residual variance • 48

Model with covariates • The only considered covariate was the health status, a categorical covariate with two levels (0=pneumonia and 1=control condition). • Actually not significant 49
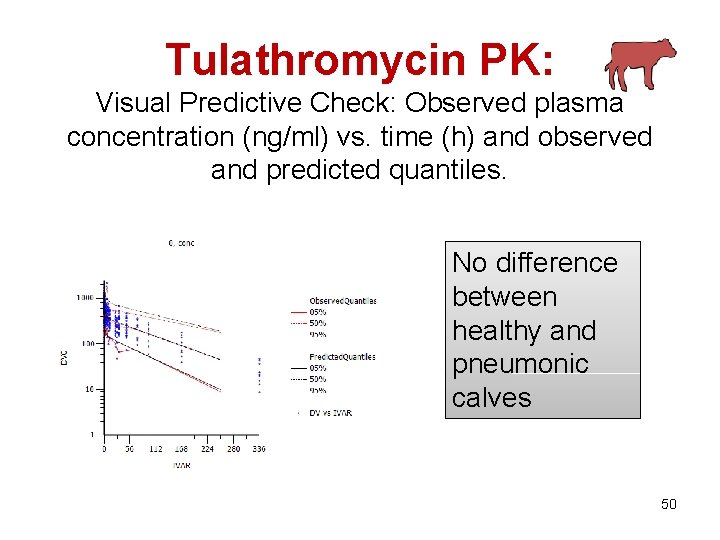
Tulathromycin PK: Visual Predictive Check: Observed plasma concentration (ng/ml) vs. time (h) and observed and predicted quantiles. No difference between healthy and pneumonic calves 50

Computation of PK/PD cutoffs for Antimicrobial Susceptibility Testing (AST) • (see presentation on the establishment of a clinical cutoff for AST) 53

Cutoff PK/PD (Copkpd) : • We have to compute the percentage of calves able to achieve some target values for the PK/PD index (here AUC/MIC) and that, for the different possible MICs encountered in calves. – In order to compute these critical values the pop model was used (without covariate) to generate a large vector of AUC using a Monte Carlo tool. 54
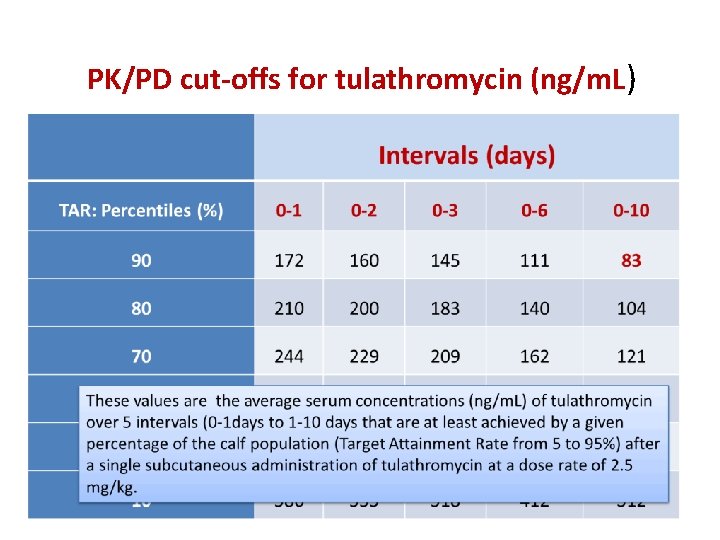
PK/PD cut-offs for tulathromycin (ng/m. L) 55
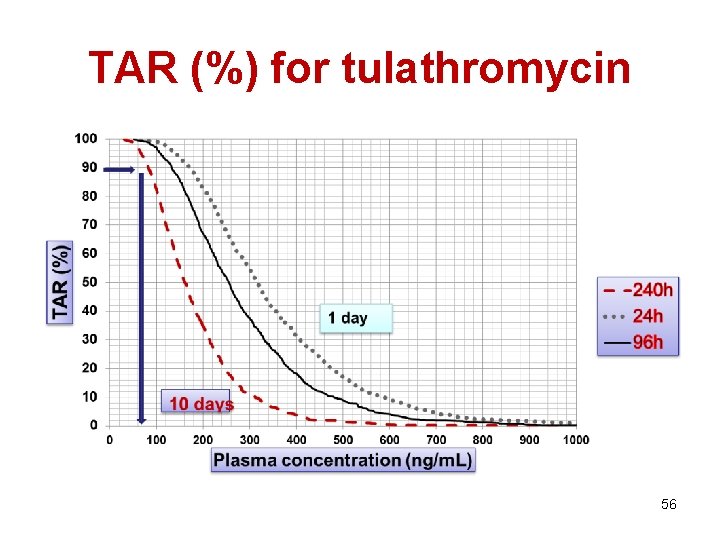
TAR (%) for tulathromycin 56

Population Dosage prediction • The adequacy of the current dosage of tulathromycin (2. 5 mg/kg) was explored by computing population doses covering different TAR i. e. different percentages of the population to assess the ability of a PK/PD model to validate or not the current dose for tulathromycin. 57
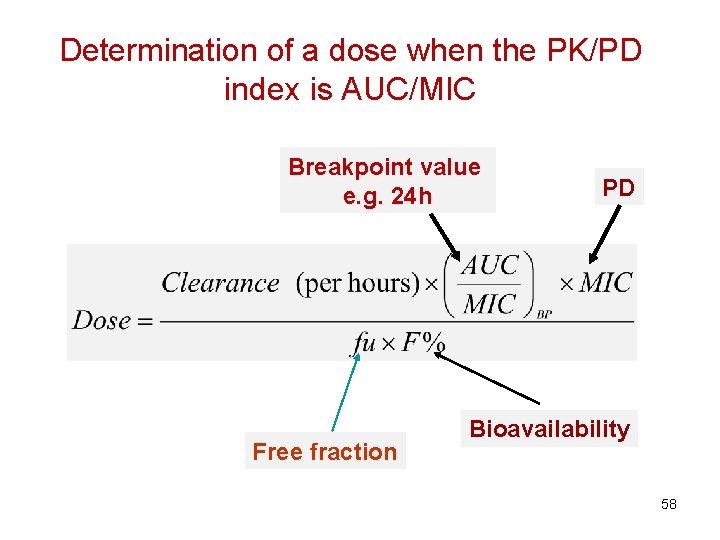
Determination of a dose when the PK/PD index is AUC/MIC Breakpoint value e. g. 24 h Free fraction PD Bioavailability 58
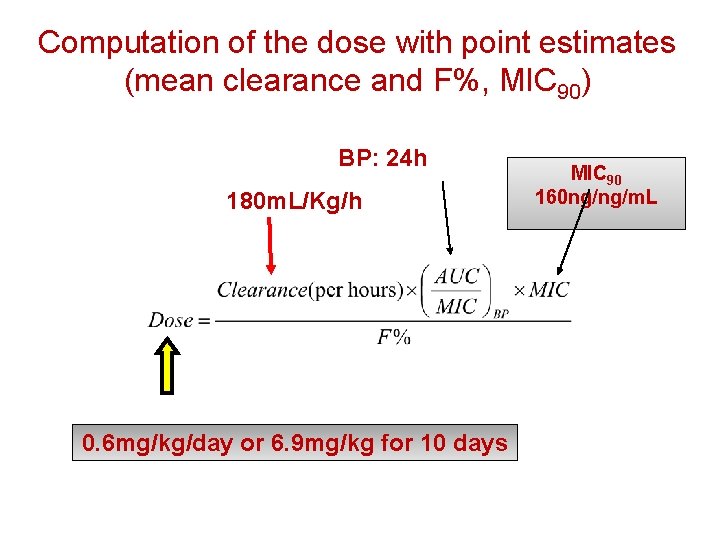
Computation of the dose with point estimates (mean clearance and F%, MIC 90) BP: 24 h 180 m. L/Kg/h 0. 6 mg/kg/day or 6. 9 mg/kg for 10 days MIC 90 160 ng/ng/m. L

Dose distribution using Monte Carlo Computation • using actual MIC distributions of M. haemolytica and P. multocida, we have generated by Monte Carlo computation (MCC) the population distribution of the tulathromycin doses to determine the corresponding TAR for the currently marketed dose of 2. 5 mg/kg. 61
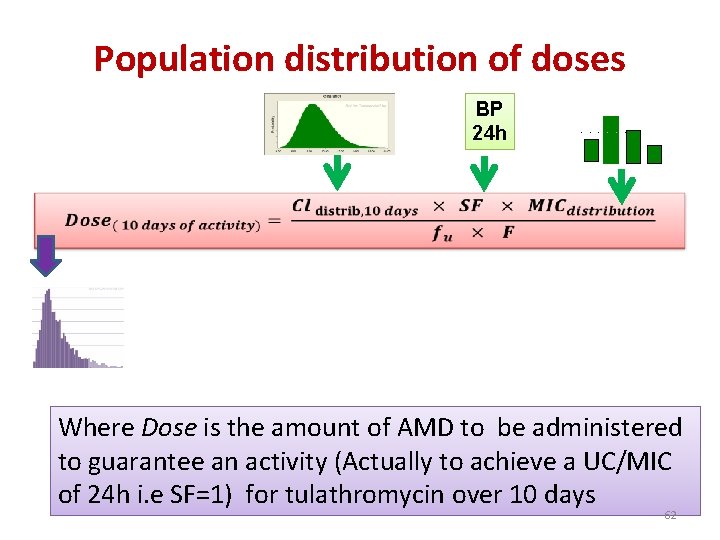
Population distribution of doses BP 24 h Where Dose is the amount of AMD to be administered to guarantee an activity (Actually to achieve a UC/MIC of 24 h i. e SF=1) for tulathromycin over 10 days 62
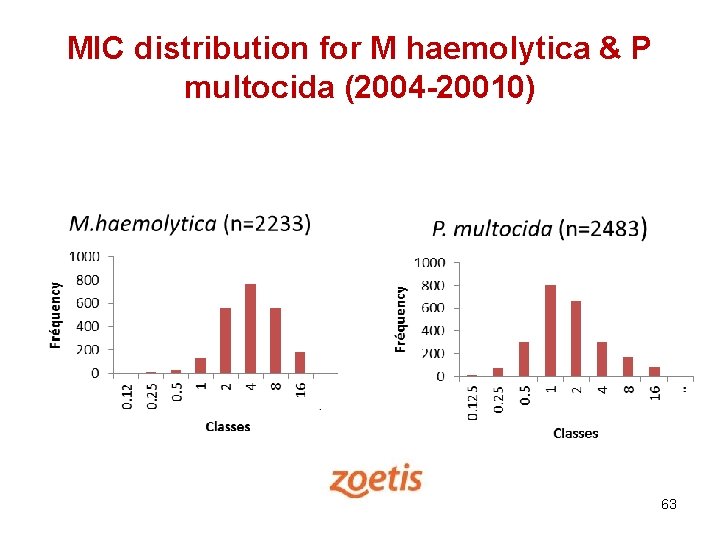
MIC distribution for M haemolytica & P multocida (2004 -20010) 63

• An add-in design to help Excel spreadsheet modelers perform Monte Carlo simulations • Others features – Search optimal solution (e. g. dose) by finding the best combination of decision variables for the best possible results
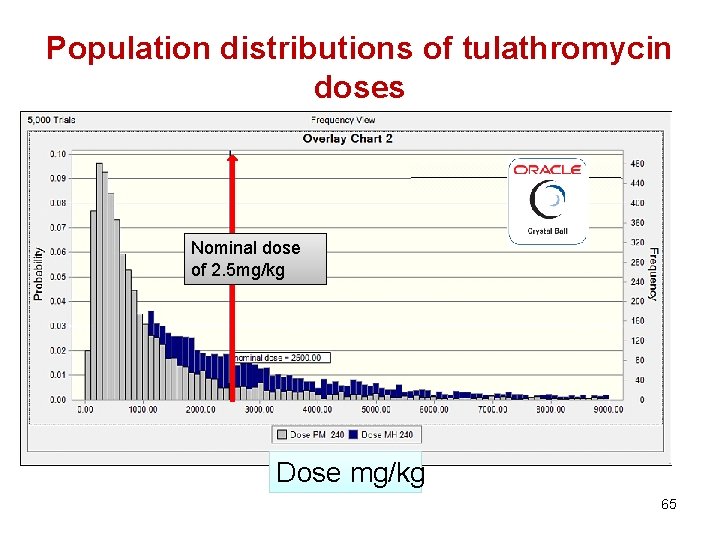
Population distributions of tulathromycin doses Nominal dose of 2. 5 mg/kg Dose mg/kg 65
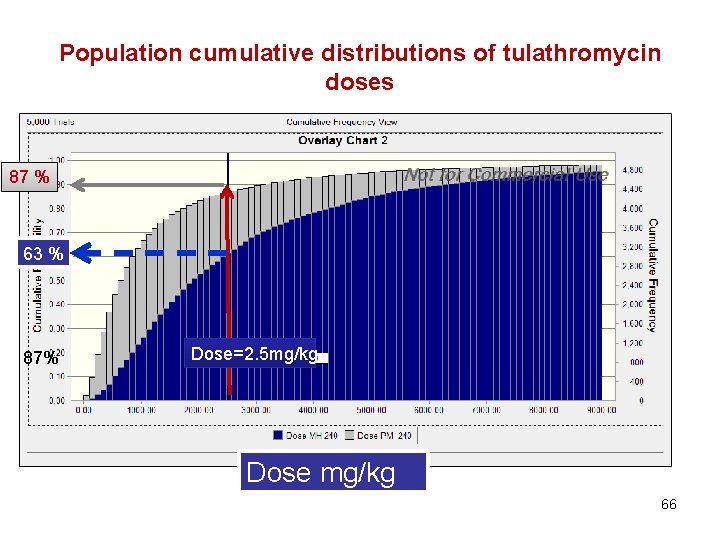
Population cumulative distributions of tulathromycin doses 87 % 63 % 87% Dose=2. 5 mg/kg Dose mg/kg 66

Dose computation • For a TAR of 90%, the computed dose for M. haemolytica was 5. 3 mg/kg, indicating that, with this dose, the average serum concentration of tulathromycin over the first 240 h following administration of this dose will be equal to the MIC of M. haemolytica in 90% of calves. • For the nominal dose of 2. 5 mg/kg, the corresponding TAR for M. haemolytica was 65. 9%. For a TAR of 90%, the computed dose for P. multocida was 2. 52 mg/kg and for the nominal dose of 2. 50 mg/kg, the TAR was 87. 2%. 67

Sensitivity analysis • Analyze the contribution of the different variables to the final result (predicted dose) • Allow to detect the most important drivers of the model 68

Sensitivity analysis 69

Sensitivity analysis • To identify the main sources of variability amongst the factors controlling the dose (plasma clearance, MICs or fu), – P. multocida • MIC : 76. 7% • clearance (23. 0%) • fu was negligible. – M. haemolytica, • MIC : 70. 3% • Clearance: 29. 2% 70

Conclusions 1. The dosage of tulathromycin can be documented using standard PK/PD concepts; 2. the recommended dose of tulathromycin (2. 5 mg/kg) is consistent with the computed population doses with computed TARs of 66% and 87% for a 2. 50 mg/kg dose for M. haemolytica and P. multocida, respectively; 3. PK/PD cut-offs are consistent with the current BPs of AST issued by the CLSI and EMA; 4. the main source of variability to take into account to determine a dose for a given animal is of PD origin (MIC) and not the actual tulathromycin exposure. 71

Dose confirmation 72

Dose-confirmation studies • At least one dose confirmation study must be presented if the dose finding is based on in vitro PD data only. 73

Dose-confirmation studies • The aim of dose-confirmation studies is to confirm the efficacy of the selected dosage regimen in individual animals (treatment claims) or groups of animals (including metaphylaxis claims) under controlled clinical conditions. – These studies can be performed using experimental models of infections but well controlled studies using naturally infected animals are preferred 74

Dose-confirmation studies • Dose confirmation studies may allow for the assessment of relapse among animals that were considered successfully treated at the time of primary efficacy assessment. • The objective is to distinguish between relapse and re-infection; 75

Determination of the duration of treatment 76

EMA and the duration of treatment • The recommended treatment duration could be justified on basis of the time course of disease progress. • Typically during clinical trials • For a single dose, OK/PD consideration can support the duration of treatment (as we have done for tulathromycin) 77

General conclusion 78

"The design of appropriate dosage regimens may be the single most important contribution of clinical pharmacology to the resistance problem" Schentag et al. Annals of Pharmacotherapy, 30: 1029 -1031 79
- Slides: 76