How to deal with data from local labs

How to deal with data from local labs © 2010 PAREXEL International | Confidential

Laboratory Data Transfer with SDTM to the LB domain • Overview of topics: • Challenge getting lab results into LB • SDTM Requirements • Data provided by Central Laboratories • Data provided by Local Laboratories • Standardized units • Conversion factors • Non-Convertible Units 2

SDTM Requirement • The task: beside the original result and original unit coming from the DB add • LBTESTCDs when the lab test is not part of the CT • Original ranges • You need ONE standard unit per lab parameter • Standardized section, including • • • Converted result (if the standardized unit is different to the original unit) Standardized unit Converted ranges (if the standardized unit is different to the original unit) • Reference range identicators • Test methods and other supplemental information 3
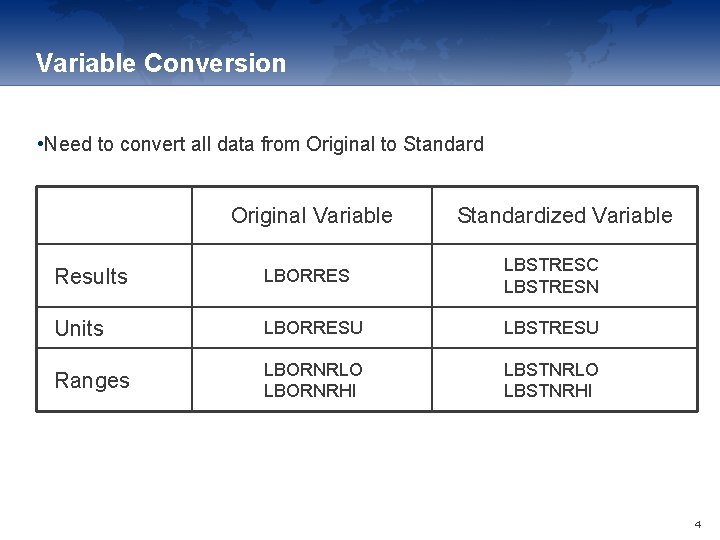
Variable Conversion • Need to convert all data from Original to Standard Original Variable Standardized Variable Results LBORRES LBSTRESC LBSTRESN Units LBORRESU LBSTRESU Ranges LBORNRLO LBORNRHI LBSTNRLO LBSTNRHI 4

Data provided by Central Laboratories • Central Laboratories are normally uncomplicated • Standardized exports from their database • Normally already only a single unit per test • Ranges attached to values consistent with the timepoint of the test • Reference range identicators in place • Additional information often given 5

Data provided by Local Laboratories • Local Laboratories are a challenge: • Result and unit entered in CRF, Ranges often collected separately • Every site could have their own unit per lab parameter • Conversion of „local“ unit to selected „standard“ unit needed • Define conversion factors per original unit which is different • Variability of syntax of local units • This includes similar units such as g/L == mg/m. L or semantic differences such as x 10^6/L == 10 E 6/L == 10^6/L • Use of „other unit“ fields • Both original and standard unit must be mapped to Controlled Terminology 6
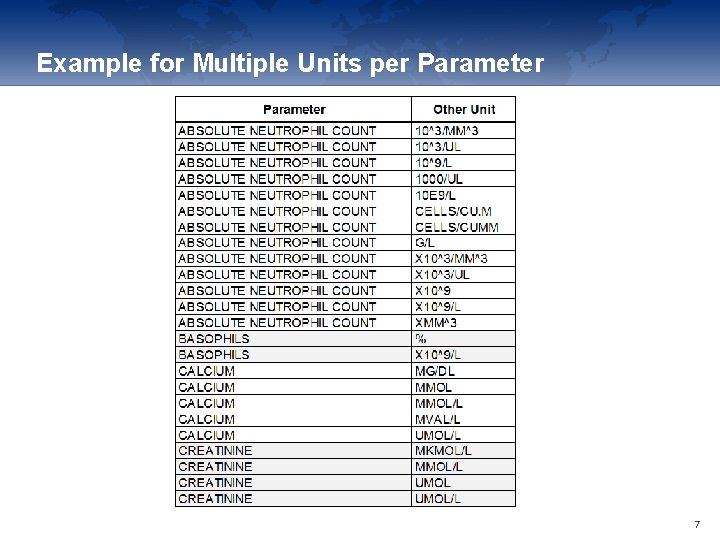
Example for Multiple Units per Parameter 7

Data provided by Local Laboratories (2) • Need to collect the ranges per investigator per lab parameter • Need to collect each range per gender and age of the subject • Ranges have to be consistent with the unit of the value • Ranges sometimes are valid only for a period - have to be updated • Ranges may be from their lab, but they may have other references. • Inconstitencies between result units and range units possible • Data validation necessary for results out of range • Investigator assessments must be included 8

Data provided by Local Laboratories (3) • Other interesting things that could happen… • The site changes the unit and/or ranges from visit to visit per subject • Typos of units which has been entered per hand • Do you know „LAKHS/CMM“ ? • • Handle exotic local units – and find conversion factors Identify the meaning for some abbrivated units • You have to spend lots of resources to get the local lab part into a good shape • Paper CRFs are much harder to handle than e. DC e. CRFs 9

Standardized units • You need ONE standard unit per lab parameter • Who determines this standard unit? • There is no library to check for the correct unit per parameter • Only for some lab parameter a „SI unit“ is defined • Often a misunderstanding between „SI unit“ and „standardized unit“ • Different terms and syntax for the same unit 10

Standardized units (2) • Sponsor defined the standard unit for „his“ submission • Standard Unit must be defined -> sponsor specific • often SI where available, but might be protocol or CRF driven as well • Not all sponsors have such standardized unit per lab parameter => some times „standardized unit” per study • Another problem: use of the same LBTEST for different LBCATs • E. g. Glucose (GLUC) for Chemestry and Urinalysis • Should the same LBTESTCD/LBTEST for the different categories? 11

Conversion factors • Same problems and questions • Who determines this conversion factors? • There is no library to check for the correct conversion factors • Need to validate each conversion factor • Has to be hardcoded in the transfer program • New data could mean: update of transfer program - because of new units • Has to be used for values and ranges 12

What would make our life easier… • General consideration: each sponsor should have a lab model defined, e. g. a library, that defines… • LBTESTCDs for non-CT lab parameter • Laboratory categories for each lab test (need a concept of categorizing tests) • Standardized units per lab parameter • Conversion factors • Question: is there a need to create such library within CDISC? 13

Special Issue: Non-Convertible Units • Check for mixture of absolute and relative results for one parameter • e. g. Basophils: given in “X 10*9/L” and “%” • There are two different SDTM CT entries for Basophils: • absolute • relative Basophils BASO Basophils/Leukocytes BASOLE • Depending on the original unit use the correct LBTESTCD/LBTEST • Never convert such values since these are two separate parameters 14
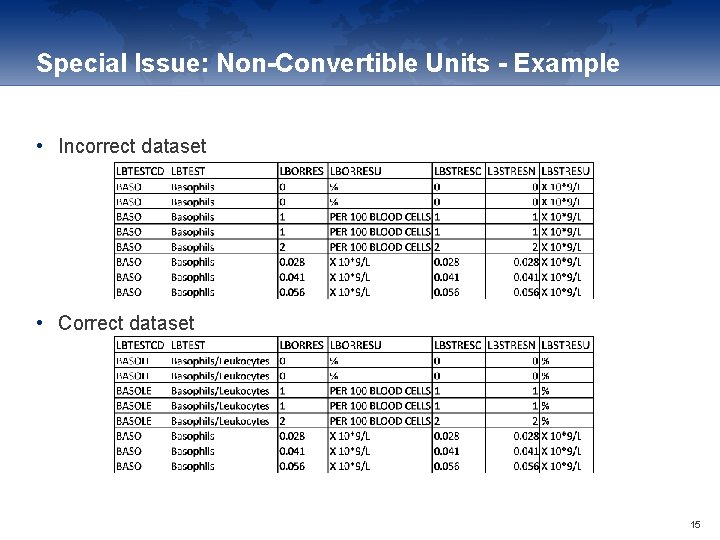
Special Issue: Non-Convertible Units - Example • Incorrect dataset • Correct dataset 15

It’s not only LB… • This standardized requirement may occurs for all Finding Domains • Everywhere using different units for one parameter check for issues • e. g. VS domain: • Temperature collected in °C and °F • Weight collected in kg and lbs • Height collected in cm and inch 16

Thank you 17
- Slides: 17