How much work is done by the gas
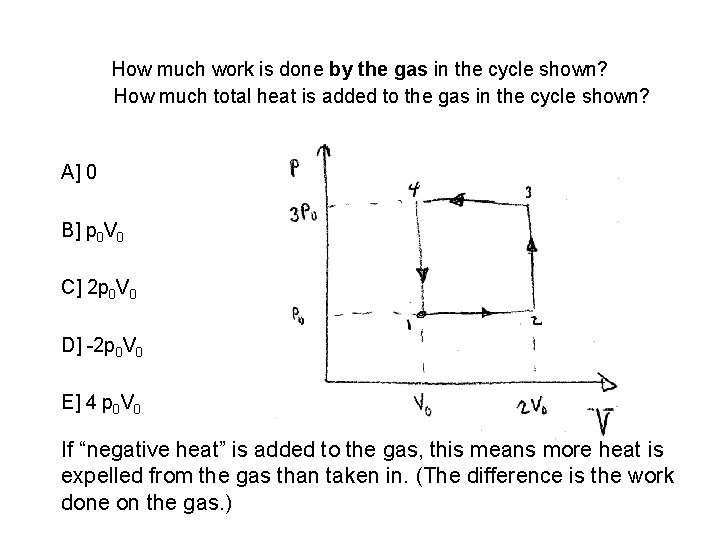
How much work is done by the gas in the cycle shown? How much total heat is added to the gas in the cycle shown? A] 0 B] p 0 V 0 C] 2 p 0 V 0 D] -2 p 0 V 0 E] 4 p 0 V 0 If “negative heat” is added to the gas, this means more heat is expelled from the gas than taken in. (The difference is the work done on the gas. )
![In one (ccw) Carnot cycle shown, the work done by the gas is: A] In one (ccw) Carnot cycle shown, the work done by the gas is: A]](http://slidetodoc.com/presentation_image_h/182560beb76b43a7016d9c892dc70909/image-2.jpg)
In one (ccw) Carnot cycle shown, the work done by the gas is: A] + B] C] 0
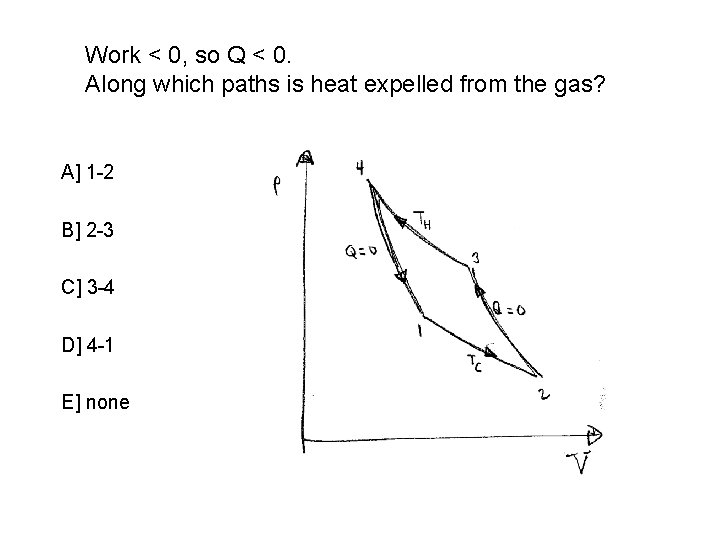
Work < 0, so Q < 0. Along which paths is heat expelled from the gas? A] 1 -2 B] 2 -3 C] 3 -4 D] 4 -1 E] none
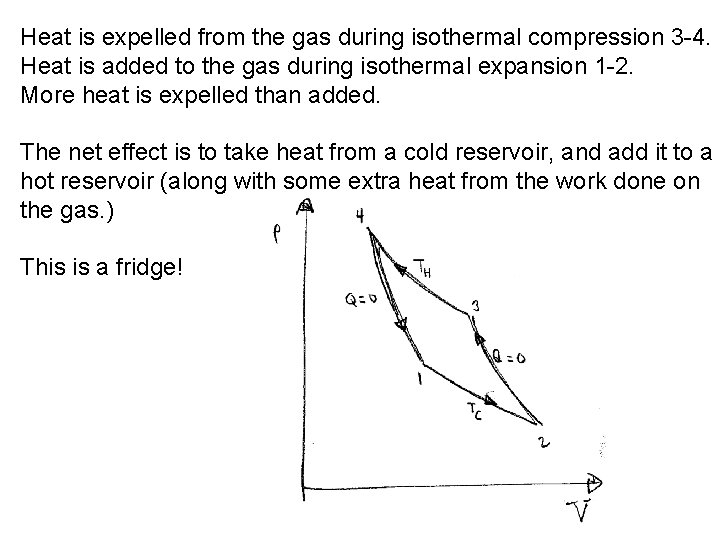
Heat is expelled from the gas during isothermal compression 3 -4. Heat is added to the gas during isothermal expansion 1 -2. More heat is expelled than added. The net effect is to take heat from a cold reservoir, and add it to a hot reservoir (along with some extra heat from the work done on the gas. ) This is a fridge!

We can never make an engine that is more efficient than a Carnot engine. Proof by contradiction: Hook up the magic engine to a Carnot engine running in reverse! (Carnot engines are reversible… they are then fridges. ) This arrangement will move heat from a cold reservoir to a hot one without external work! As that is impossible, no such engine is possible.
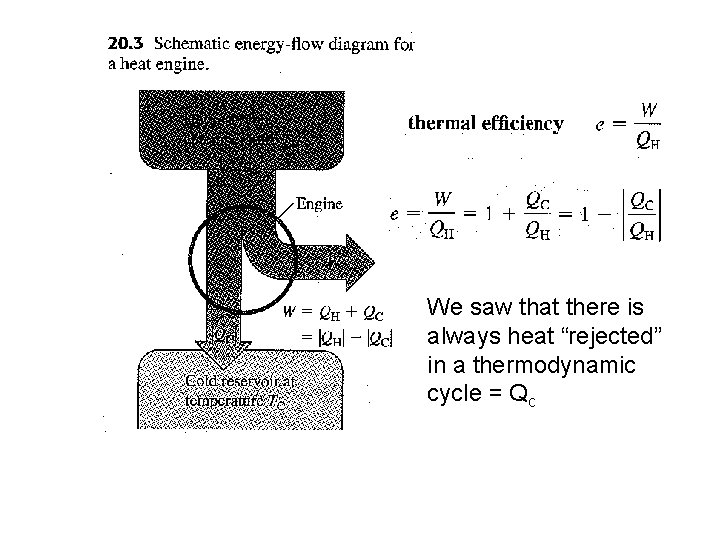
We saw that there is always heat “rejected” in a thermodynamic cycle = Qc
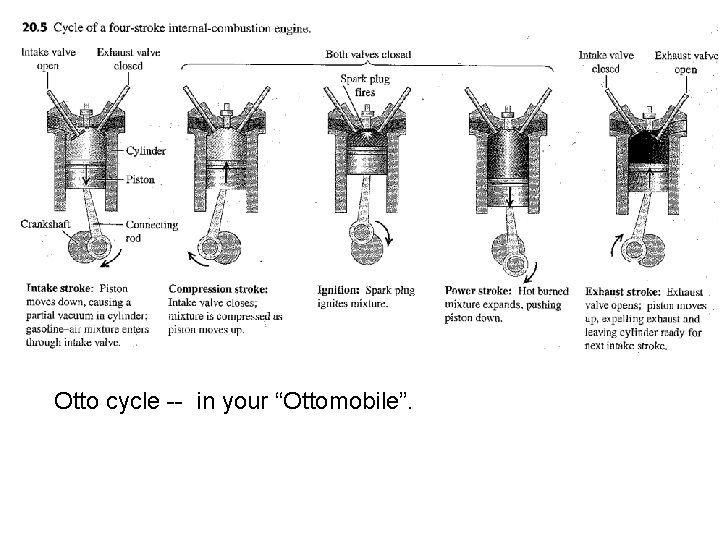
Otto cycle -- in your “Ottomobile”.
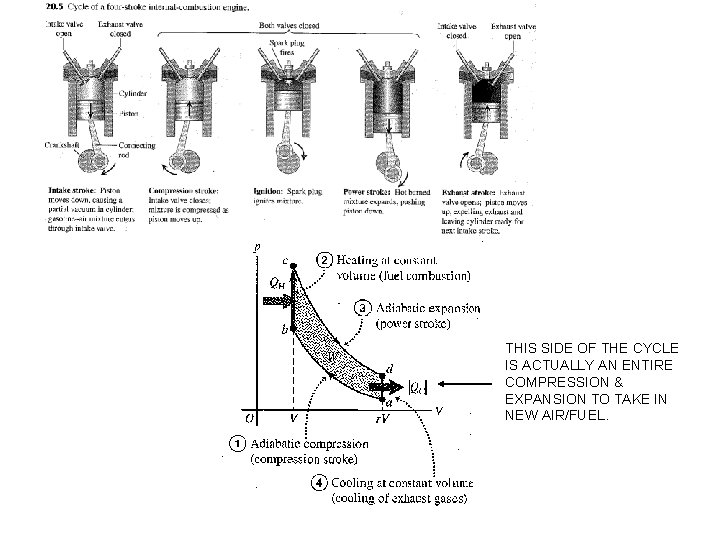
THIS SIDE OF THE CYCLE IS ACTUALLY AN ENTIRE COMPRESSION & EXPANSION TO TAKE IN NEW AIR/FUEL.
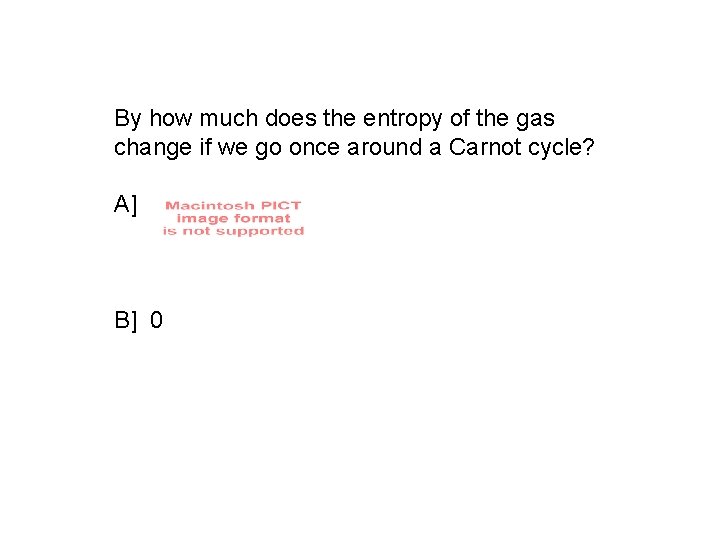
By how much does the entropy of the gas change if we go once around a Carnot cycle? A] B] 0

By how much does the entropy of the hot reservoir change if we go once around a Carnot cycle? A] B] C] 0 QH is the heat added to the gas from the hot reservoir

By how much does the total entropy of both reservoirs change if we go once around a Carnot cycle? A] B] C] 0 QH is the heat added to the gas from the hot reservoir

If there is such a thing as entropy of a gas, then it must be a state variable, I. e. it depends only on p, V, T, composition… it’s not a process variable. Another way to see this is to consider that any Carnot cycle leaves entropy unchanged, and any path in p. V can be considered as the sum of Carnot cycles!
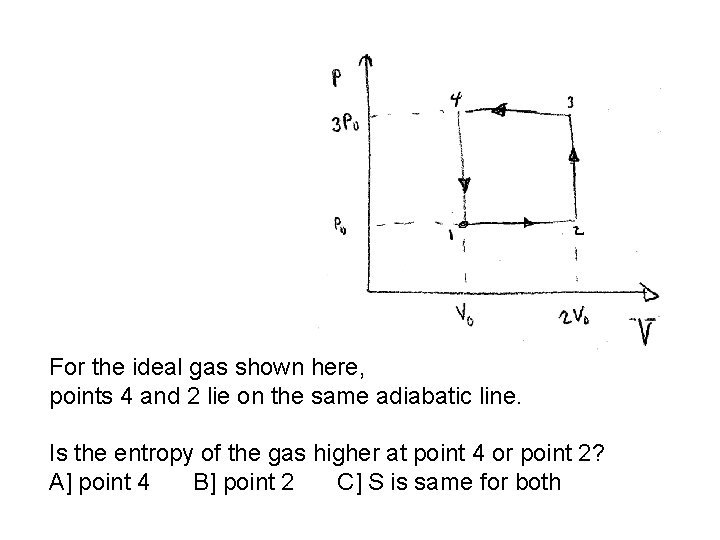
For the ideal gas shown here, points 4 and 2 lie on the same adiabatic line. Is the entropy of the gas higher at point 4 or point 2? A] point 4 B] point 2 C] S is same for both
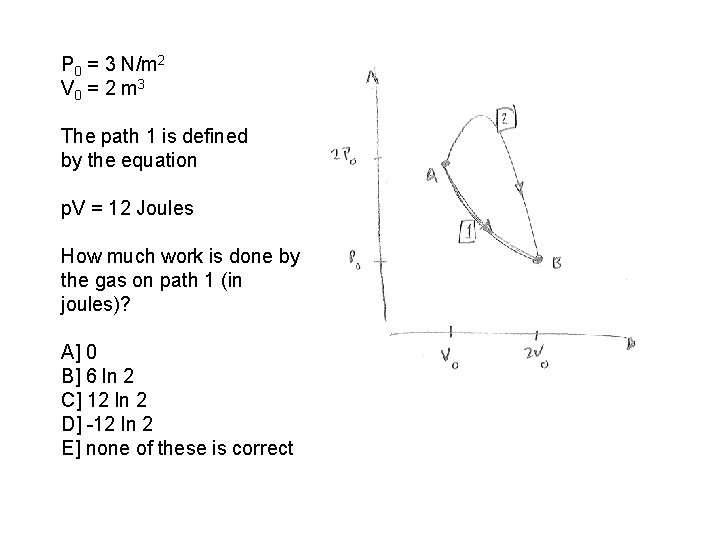
P 0 = 3 N/m 2 V 0 = 2 m 3 The path 1 is defined by the equation p. V = 12 Joules How much work is done by the gas on path 1 (in joules)? A] 0 B] 6 ln 2 C] 12 ln 2 D] -12 ln 2 E] none of these is correct
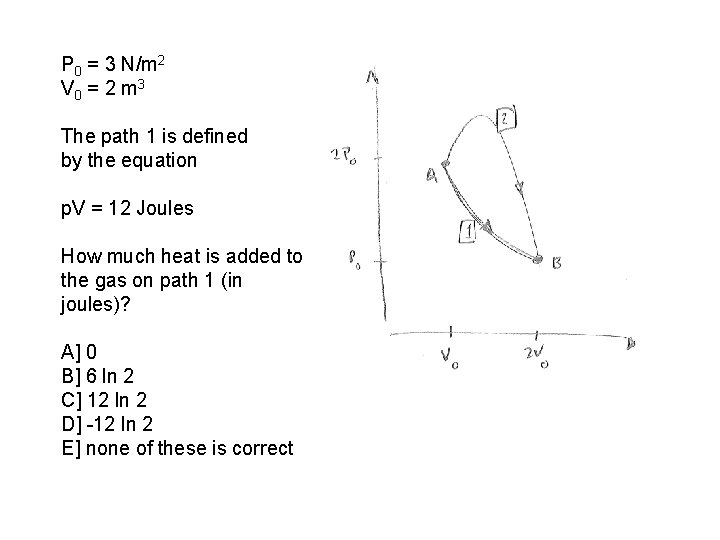
P 0 = 3 N/m 2 V 0 = 2 m 3 The path 1 is defined by the equation p. V = 12 Joules How much heat is added to the gas on path 1 (in joules)? A] 0 B] 6 ln 2 C] 12 ln 2 D] -12 ln 2 E] none of these is correct
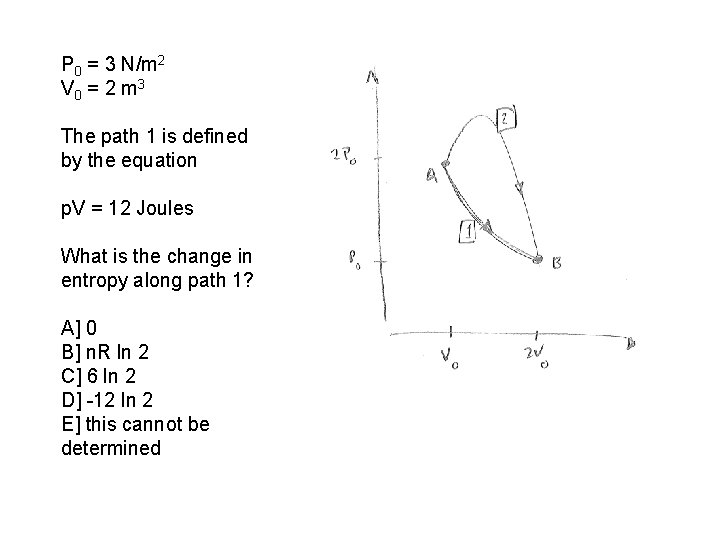
P 0 = 3 N/m 2 V 0 = 2 m 3 The path 1 is defined by the equation p. V = 12 Joules What is the change in entropy along path 1? A] 0 B] n. R ln 2 C] 6 ln 2 D] -12 ln 2 E] this cannot be determined
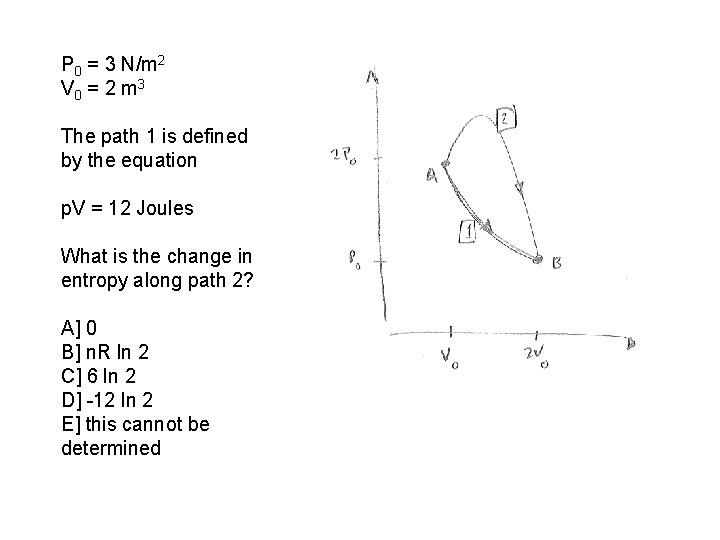
P 0 = 3 N/m 2 V 0 = 2 m 3 The path 1 is defined by the equation p. V = 12 Joules What is the change in entropy along path 2? A] 0 B] n. R ln 2 C] 6 ln 2 D] -12 ln 2 E] this cannot be determined
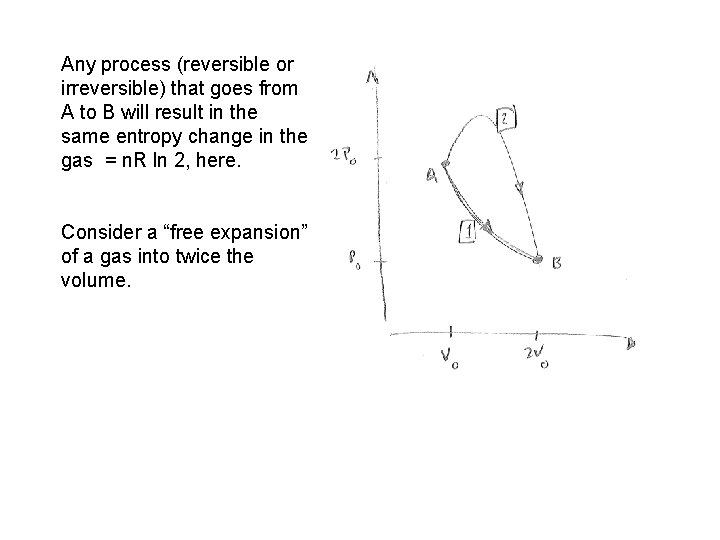
Any process (reversible or irreversible) that goes from A to B will result in the same entropy change in the gas = n. R ln 2, here. Consider a “free expansion” of a gas into twice the volume.
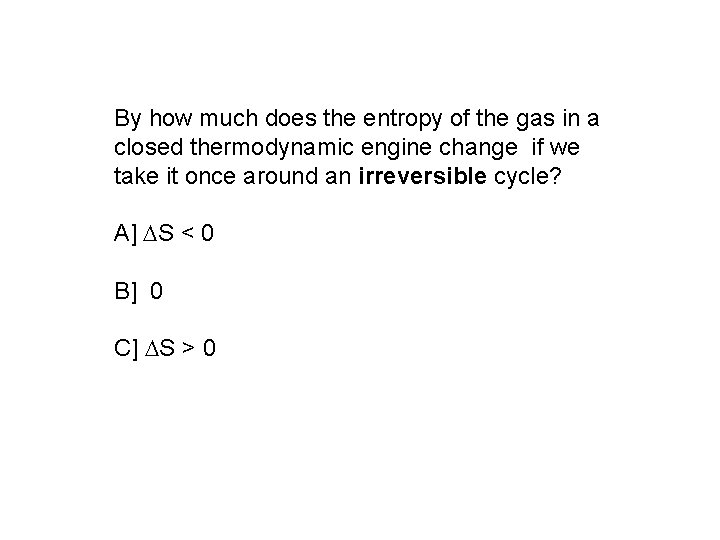
By how much does the entropy of the gas in a closed thermodynamic engine change if we take it once around an irreversible cycle? A] S < 0 B] 0 C] S > 0

By how much does the entropy of the surrounding reservoirs change if we take a closed thermodynamic engine once around an irreversible cycle? A] S < 0 B] 0 C] S > 0

By how much does the entropy of the surrounding reservoirs change if we take a closed thermodynamic engine once around an irreversible cycle? C] S > 0 Consider a hot reservoir T > TH. This reservoir loses entropy (just as with the reversible cycle), but loses a little less than when T = TH. The cold reservoir with T < Tc gains entropy, but gains a little more than when T=Tc.
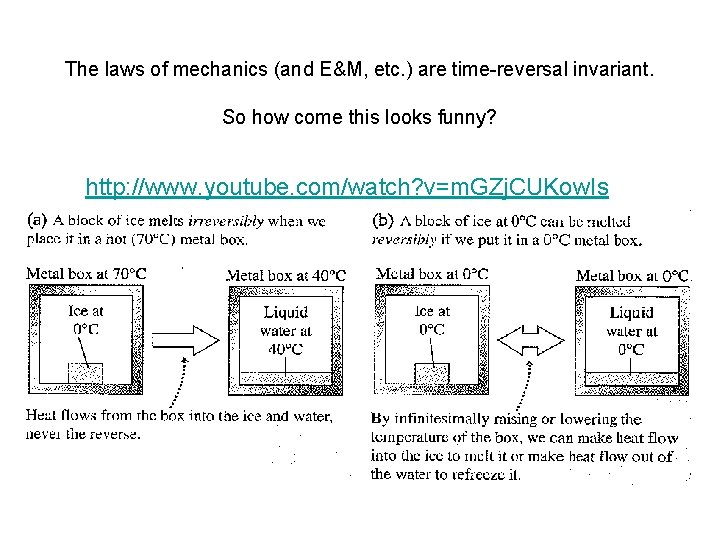
The laws of mechanics (and E&M, etc. ) are time-reversal invariant. So how come this looks funny? http: //www. youtube. com/watch? v=m. GZj. CUKow. Is

Heat flows from a hot object to a cold object in contact with it because: A] the hot object has more total internal energy and heat flows until both objects have the same internal energy B] the hot object has more total energy per moleculeand heat flows until both objects have the same energy per molecule C] the hot object has more translational kinetic energy per molecule - and heat flows until both objects have the same translational kinetic energy per molecule.
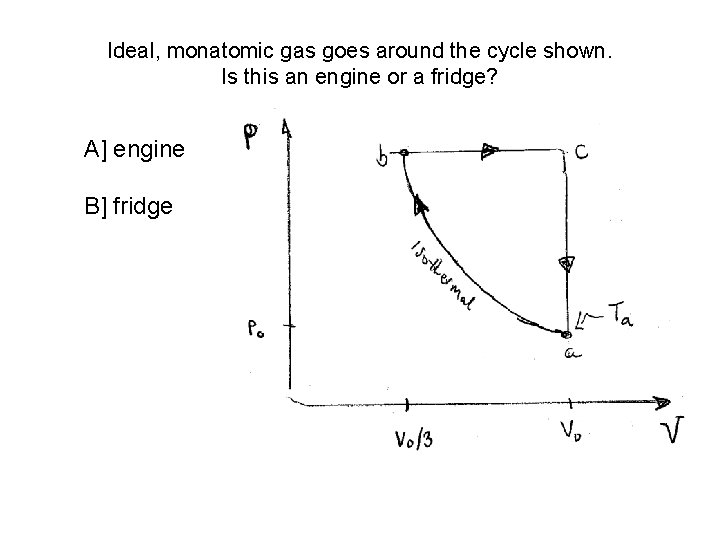
Ideal, monatomic gas goes around the cycle shown. Is this an engine or a fridge? A] engine B] fridge
![What is the temperature at c? A] Ta B] 2 Ta C] 3 Ta What is the temperature at c? A] Ta B] 2 Ta C] 3 Ta](http://slidetodoc.com/presentation_image_h/182560beb76b43a7016d9c892dc70909/image-25.jpg)
What is the temperature at c? A] Ta B] 2 Ta C] 3 Ta D] Ta/3 E] cannot determine
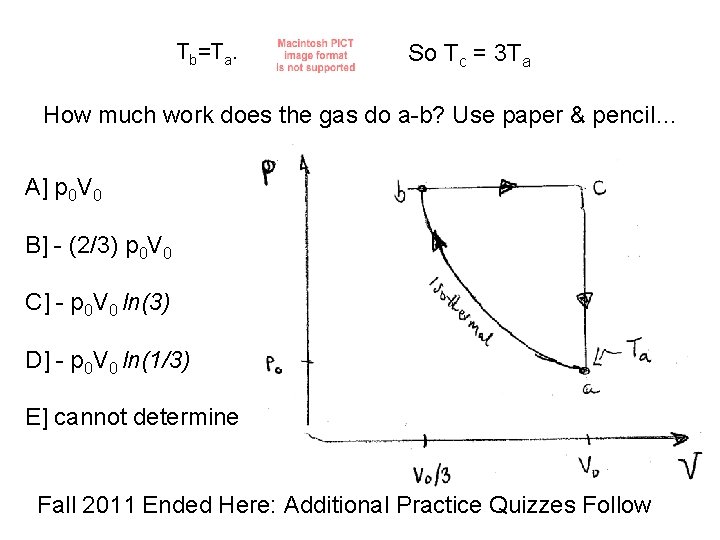
Tb=Ta. So Tc = 3 Ta How much work does the gas do a-b? Use paper & pencil… A] p 0 V 0 B] - (2/3) p 0 V 0 C] - p 0 V 0 ln(3) D] - p 0 V 0 ln(1/3) E] cannot determine Fall 2011 Ended Here: Additional Practice Quizzes Follow
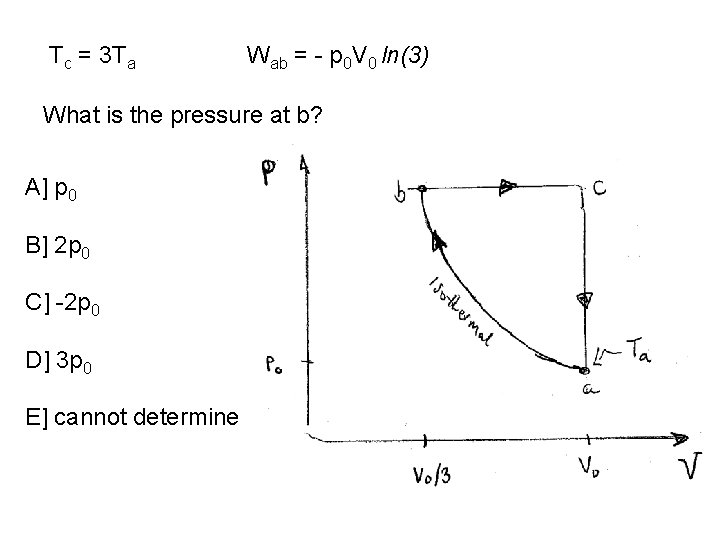
Tc = 3 Ta Wab = - p 0 V 0 ln(3) What is the pressure at b? A] p 0 B] 2 p 0 C] -2 p 0 D] 3 p 0 E] cannot determine
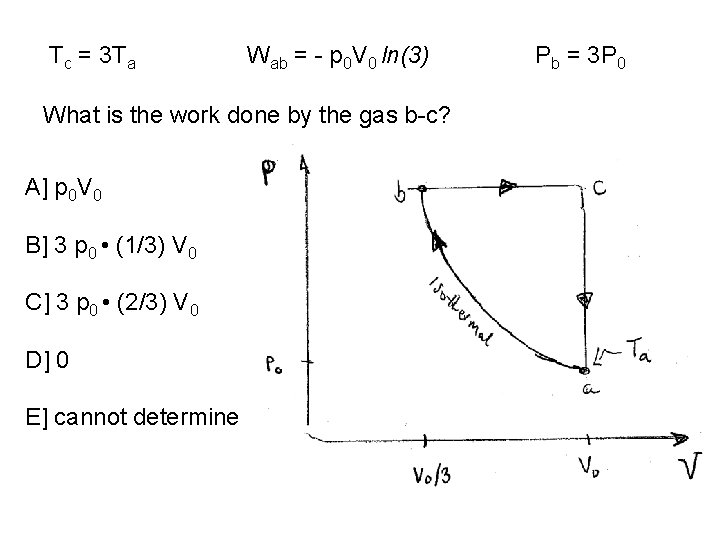
Tc = 3 Ta Wab = - p 0 V 0 ln(3) What is the work done by the gas b-c? A] p 0 V 0 B] 3 p 0 • (1/3) V 0 C] 3 p 0 • (2/3) V 0 D] 0 E] cannot determine Pb = 3 P 0
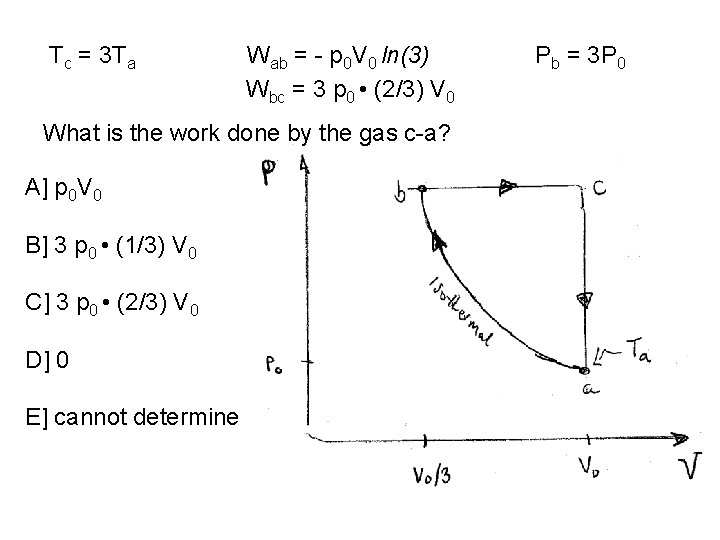
Tc = 3 Ta Wab = - p 0 V 0 ln(3) Wbc = 3 p 0 • (2/3) V 0 What is the work done by the gas c-a? A] p 0 V 0 B] 3 p 0 • (1/3) V 0 C] 3 p 0 • (2/3) V 0 D] 0 E] cannot determine Pb = 3 P 0
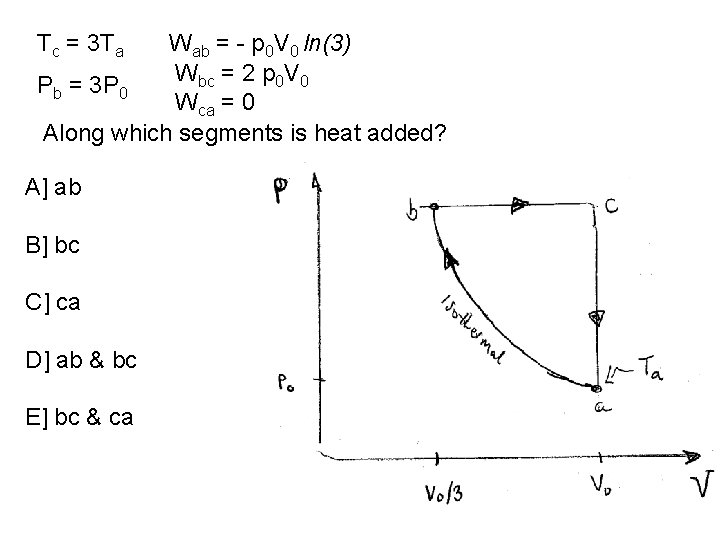
Tc = 3 Ta Wab = - p 0 V 0 ln(3) Wbc = 2 p 0 V 0 Pb = 3 P 0 Wca = 0 Along which segments is heat added? A] ab B] bc C] ca D] ab & bc E] bc & ca
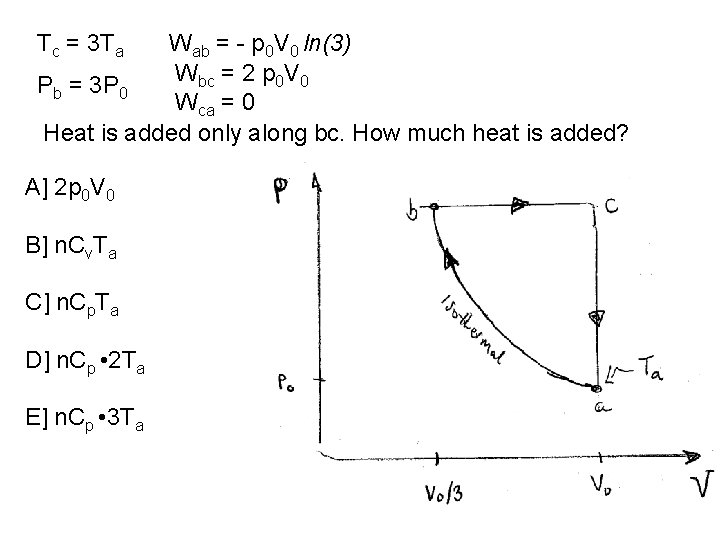
Tc = 3 Ta Wab = - p 0 V 0 ln(3) Wbc = 2 p 0 V 0 Pb = 3 P 0 Wca = 0 Heat is added only along bc. How much heat is added? A] 2 p 0 V 0 B] n. Cv. Ta C] n. Cp. Ta D] n. Cp • 2 Ta E] n. Cp • 3 Ta
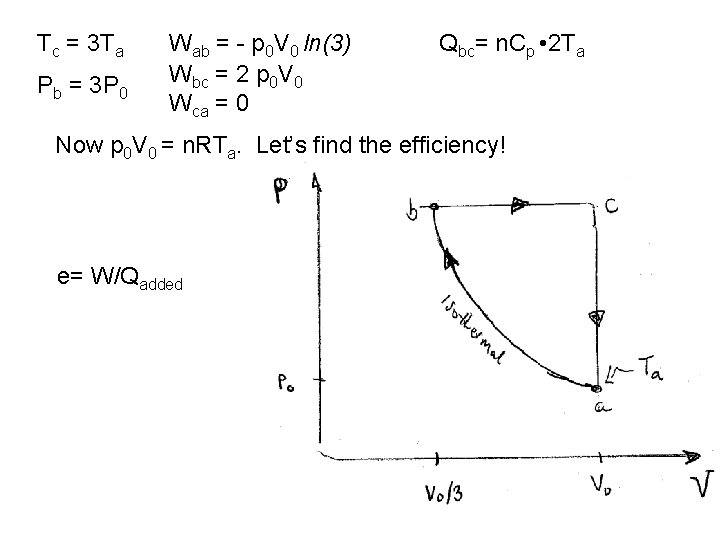
Tc = 3 Ta Pb = 3 P 0 Wab = - p 0 V 0 ln(3) Wbc = 2 p 0 V 0 Wca = 0 Qbc= n. Cp • 2 Ta Now p 0 V 0 = n. RTa. Let’s find the efficiency! e= W/Qadded
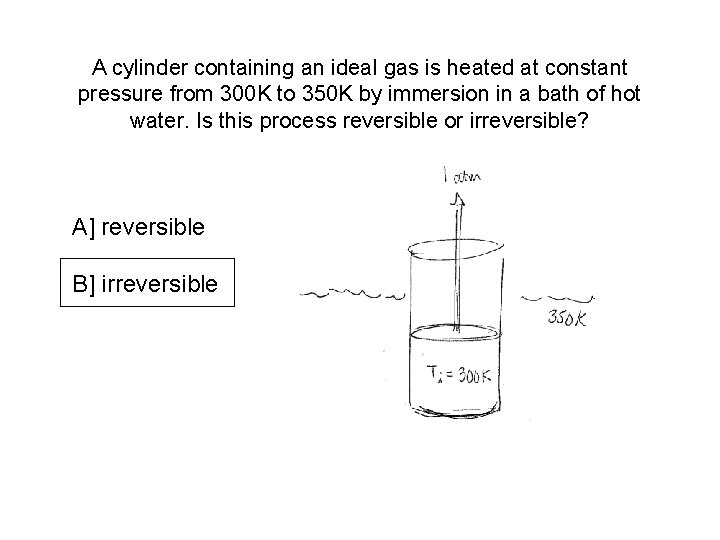
A cylinder containing an ideal gas is heated at constant pressure from 300 K to 350 K by immersion in a bath of hot water. Is this process reversible or irreversible? A] reversible B] irreversible

A hot piece of metal is placed in an insulating box filled with a polyatomic gas. When thermal equilibrium has been reached: A] the metal and the gas have equal total energy B] the average energy per atom in the metal is equal to the average energy per molecule in the gas C] the average kinetic energy per atom in the metal is equal to the average translational kinetic energy per molecule in the gas D] the average kinetic energy per atom in the metal is equal to the average kinetic energy per atom in the gas
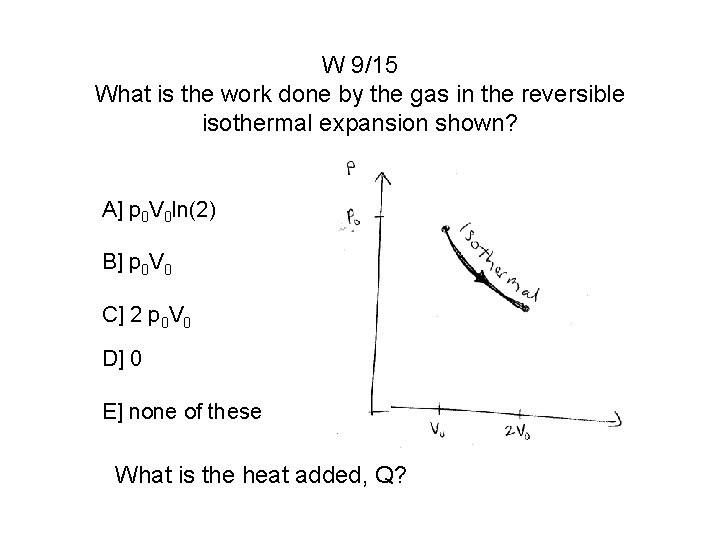
W 9/15 What is the work done by the gas in the reversible isothermal expansion shown? A] p 0 V 0 ln(2) B] p 0 V 0 C] 2 p 0 V 0 D] 0 E] none of these What is the heat added, Q?
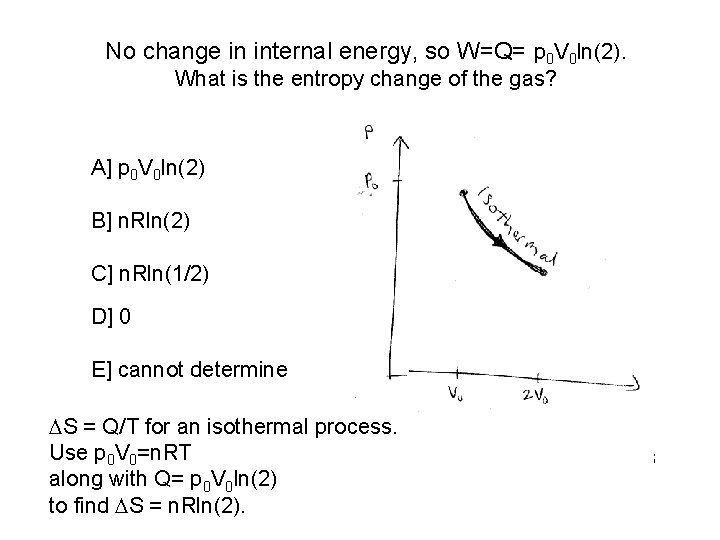
No change in internal energy, so W=Q= p 0 V 0 ln(2). What is the entropy change of the gas? A] p 0 V 0 ln(2) B] n. Rln(2) C] n. Rln(1/2) D] 0 E] cannot determine S = Q/T for an isothermal process. Use p 0 V 0 is=n. RT What the entropy change in the hot reservoir which is along withheat Q= pto ln(2) 0 V 0 adding the gas? to find S = n. Rln(2).
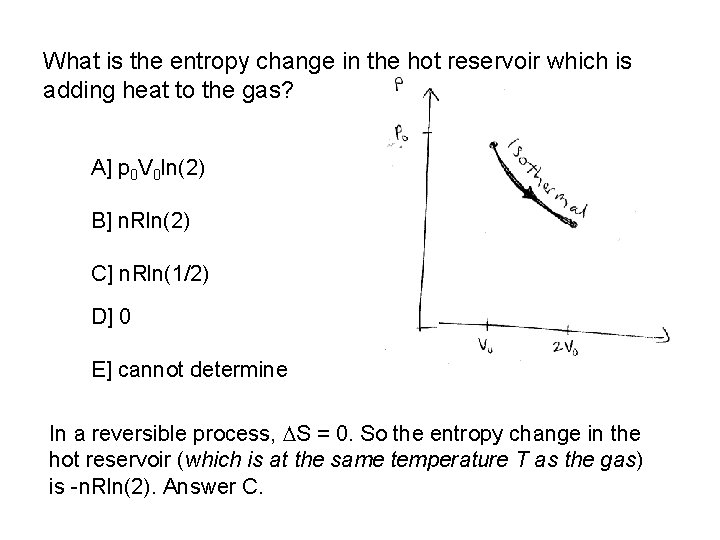
What is the entropy change in the hot reservoir which is adding heat to the gas? A] p 0 V 0 ln(2) B] n. Rln(2) C] n. Rln(1/2) D] 0 E] cannot determine In a reversible process, S = 0. So the entropy change in the hot reservoir (which is at the same temperature T as the gas) is -n. Rln(2). Answer C.
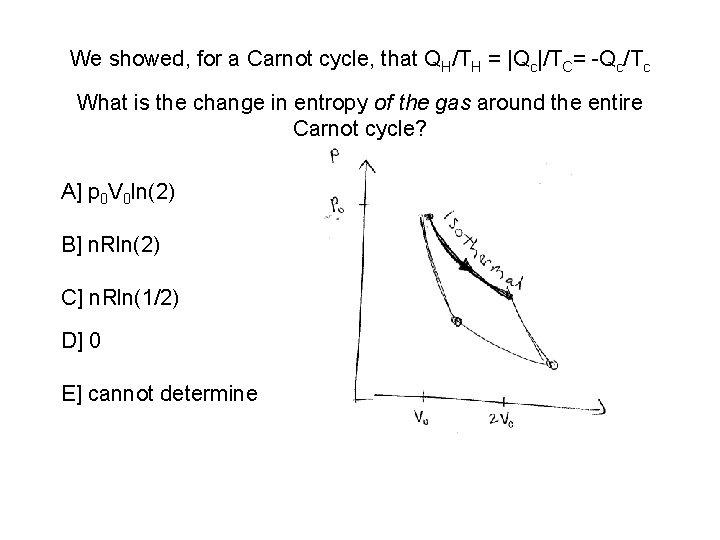
We showed, for a Carnot cycle, that QH/TH = |Qc|/TC= -Qc/Tc What is the change in entropy of the gas around the entire Carnot cycle? A] p 0 V 0 ln(2) B] n. Rln(2) C] n. Rln(1/2) D] 0 E] cannot determine

Any reversible process consists of “adjoining” Carnot cycles. S for adjoining segments cancels. So: Entropy, like Internal Energy, is a “state” variable, and depends only on the state of a system (p, V for a gas). -> You can calculate entropy changes for irreversible processes by taking a reversible path to the same endpoint. Example: free expansion to double the volume. Tf = Ti.
- Slides: 39