How Does One Implement a Protocol for Antiplatelet











- Slides: 37

How Does One Implement a Protocol for Antiplatelet Therapy in ACS and During PCI? Jeffrey S Berger, MD, MS Associate Professor of Medicine and Surgery Director of Cardiovascular Thrombosis

Disclosures Astra Zeneca – Research Support and Serve on the executive committee for the EUCLID trial comparing ticagrelor and clopidogrel in PAD Takeda/Merck/Jn. J – Advisory Board Grants - American Heart Association Doris Duke Charitable Foundation National Heart Lung and Blood Institute

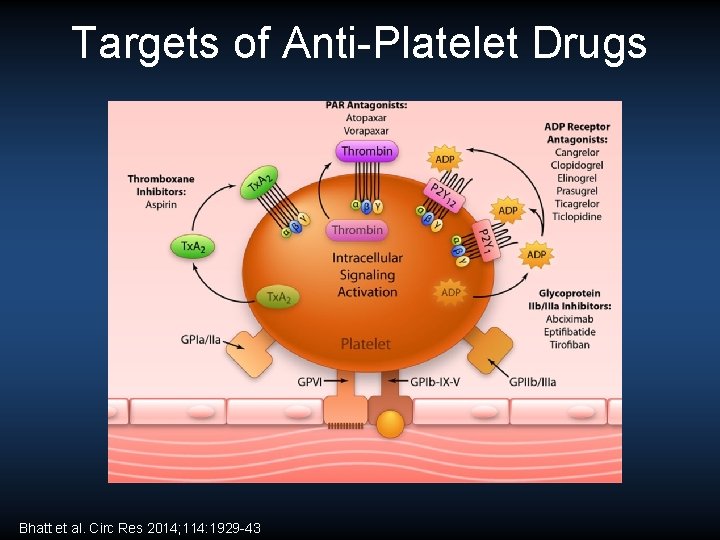
Targets of Anti-Platelet Drugs Bhatt et al. Circ Res 2014; 114: 1929 -43

Issues to Consider • • Drug Dose Combination Timing – Initiation – Discontinuation • Interactions (drug-drug; drug-environment) • Genetics and Platelet Phenotype • One must also consider: availability and cost

Current Guideline Based Protocols for Antiplatelet Therapy • ACS with Planned Intervention • Medically Managed ACS – Aspirin PLUS • Clopidogrel, or • Prasugrel, or • Ticagrelor • Elective PCI – Aspirin PLUS • Clopidogrel Need to Consider: GP IIb/IIIa Inhibitors, Cangrelor, Vorapaxar (2 weeks post MI) and Cilostazol are sometimes used

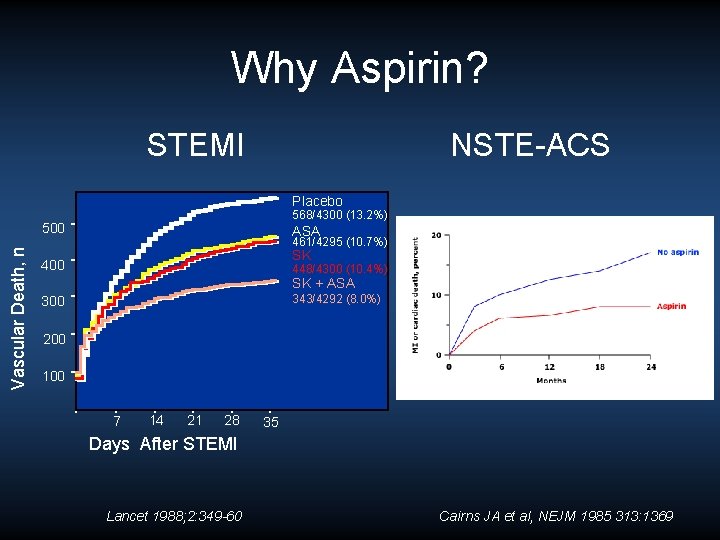
Why Aspirin? STEMI NSTE-ACS Placebo 568/4300 (13. 2%) Vascular Death, n 500 ASA 461/4295 (10. 7%) SK 400 448/4300 (10. 4%) SK + ASA 343/4292 (8. 0%) 300 200 100 7 14 21 28 35 Days After STEMI Lancet 1988; 2: 349 -60 Cairns JA et al, NEJM 1985 313: 1369

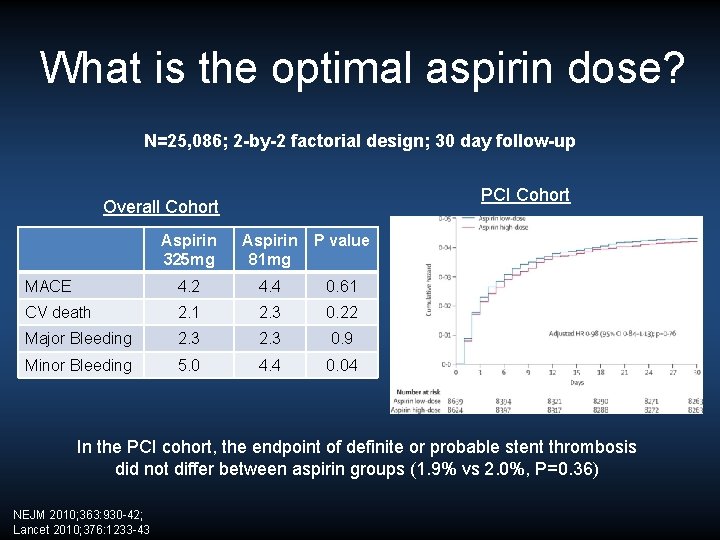
What is the optimal aspirin dose? N=25, 086; 2 -by-2 factorial design; 30 day follow-up PCI Cohort Overall Cohort Aspirin 325 mg Aspirin P value 81 mg MACE 4. 2 4. 4 0. 61 CV death 2. 1 2. 3 0. 22 Major Bleeding 2. 3 0. 9 Minor Bleeding 5. 0 4. 4 0. 04 In the PCI cohort, the endpoint of definite or probable stent thrombosis did not differ between aspirin groups (1. 9% vs 2. 0%, P=0. 36) NEJM 2010; 363: 930 -42; Lancet 2010; 376: 1233 -43

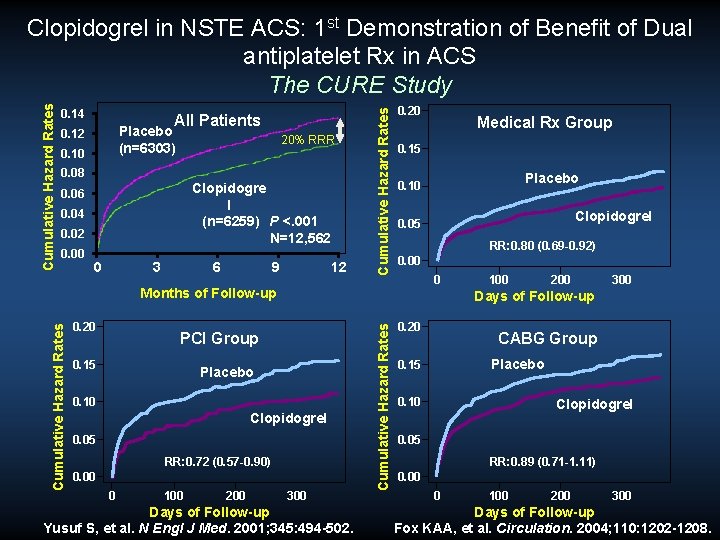
0. 14 All Patients Placebo (n=6303) 0. 12 0. 10 20% RRR 0. 08 Clopidogre l (n=6259) P <. 001 N=12, 562 0. 06 0. 04 0. 02 0. 00 0 3 6 9 12 Cumulative Hazard Rates Clopidogrel in NSTE ACS: 1 st Demonstration of Benefit of Dual antiplatelet Rx in ACS The CURE Study 0. 20 0. 15 0. 10 Clopidogrel 0. 05 RR: 0. 72 (0. 57 -0. 90) 0. 00 100 200 300 Days of Follow-up Yusuf S, et al. N Engl J Med. 2001; 345: 494 -502. Cumulative Hazard Rates Placebo 0 Clopidogrel 0. 05 RR: 0. 80 (0. 69 -0. 92) 0. 00 0 100 200 300 Days of Follow-up PCI Group 0. 15 Placebo 0. 10 Months of Follow-up 0. 20 Medical Rx Group 0. 20 CABG Group Placebo 0. 15 0. 10 Clopidogrel 0. 05 RR: 0. 89 (0. 71 -1. 11) 0. 00 0 100 200 300 Days of Follow-up Fox KAA, et al. Circulation. 2004; 110: 1202 -1208.

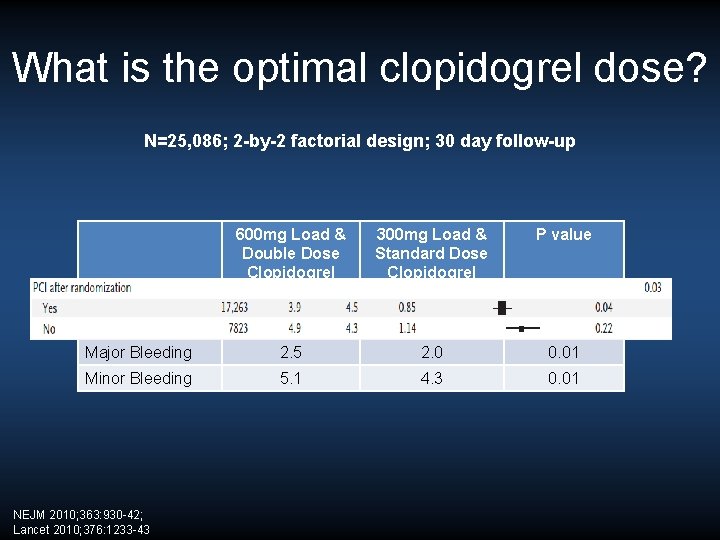
What is the optimal clopidogrel dose? N=25, 086; 2 -by-2 factorial design; 30 day follow-up 600 mg Load & Double Dose Clopidogrel 300 mg Load & Standard Dose Clopidogrel P value MACE 4. 2 4. 4 0. 3 CV death 2. 1 2. 2 0. 57 Major Bleeding 2. 5 2. 0 0. 01 Minor Bleeding 5. 1 4. 3 0. 01 NEJM 2010; 363: 930 -42; Lancet 2010; 376: 1233 -43

Limitations of Clopidogrel • Heterogenous antiplatelet response • Genetic polymorphisms associated with poor response • Drug-drug interaction • Environmental (smoking) interaction Bonello L, JACC 2010; 56: 919 -33 Ho PM, JAMA 2009; 301: 937 -44 Berger JS, Circulation 2009; 120: 2337 -44

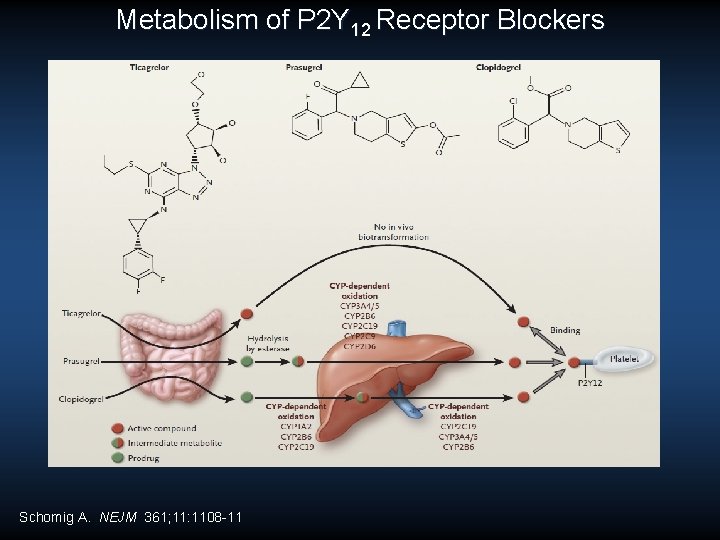
Metabolism of P 2 Y 12 Receptor Blockers Schomig A. NEJM 361; 11: 1108 -11

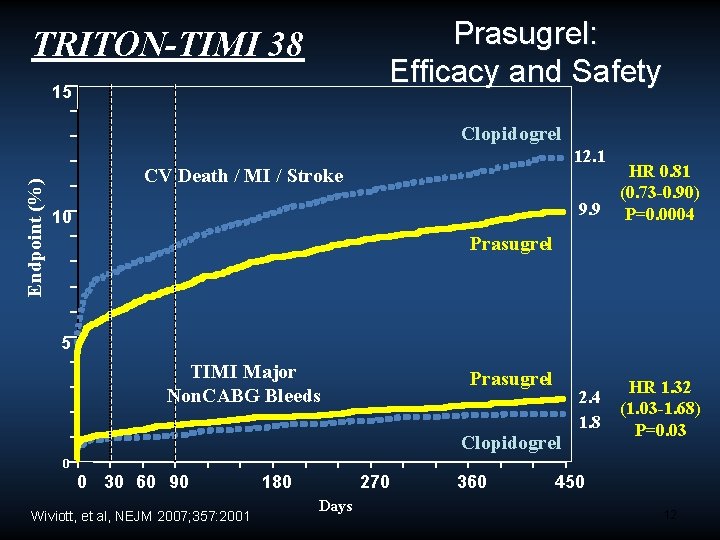
Prasugrel: Efficacy and Safety TRITON-TIMI 38 15 Endpoint (%) Clopidogrel 12. 1 CV Death / MI / Stroke 10 9. 9 HR 0. 81 (0. 73 -0. 90) P=0. 0004 2. 4 1. 8 HR 1. 32 (1. 03 -1. 68) P=0. 03 Prasugrel 5 TIMI Major Non. CABG Bleeds Prasugrel Clopidogrel 0 0 30 60 90 Wiviott, et al, NEJM 2007; 357: 2001 180 270 Days 360 450 12

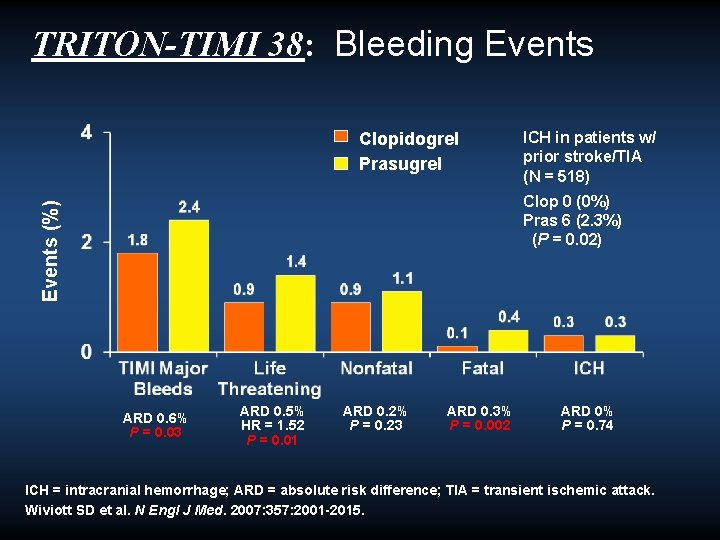
TRITON-TIMI 38: Bleeding Events Clopidogrel Prasugrel ICH in patients w/ prior stroke/TIA (N = 518) Events (%) Clop 0 (0%) Pras 6 (2. 3%) (P = 0. 02) ARD 0. 6% P = 0. 03 ARD 0. 5% HR = 1. 52 P = 0. 01 ARD 0. 2% P = 0. 23 ARD 0. 3% P = 0. 002 ARD 0% P = 0. 74 ICH = intracranial hemorrhage; ARD = absolute risk difference; TIA = transient ischemic attack. Wiviott SD et al. N Engl J Med. 2007: 357: 2001 -2015.

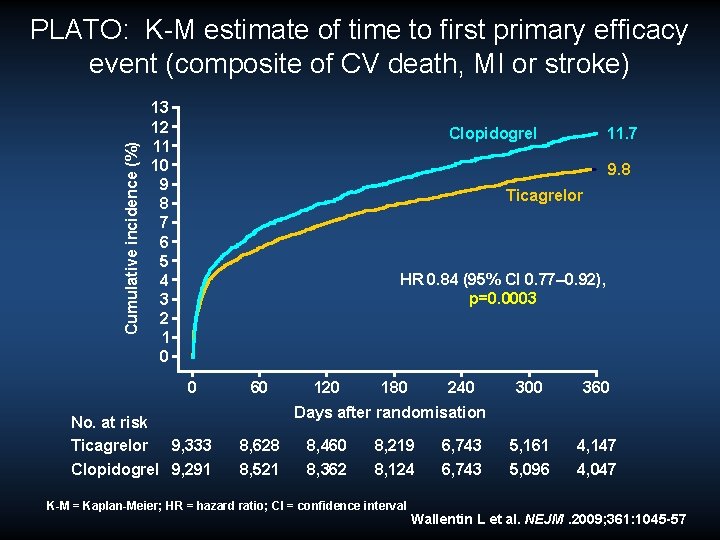
Cumulative incidence (%) PLATO: K-M estimate of time to first primary efficacy event (composite of CV death, MI or stroke) 13 12 11 10 9 8 7 6 5 4 3 2 1 0 11. 7 Clopidogrel 9. 8 Ticagrelor HR 0. 84 (95% CI 0. 77– 0. 92), p=0. 0003 0 No. at risk Ticagrelor 9, 333 Clopidogrel 9, 291 60 120 180 240 300 360 5, 161 5, 096 4, 147 4, 047 Days after randomisation 8, 628 8, 521 8, 460 8, 362 8, 219 8, 124 K-M = Kaplan-Meier; HR = hazard ratio; CI = confidence interval 6, 743 Wallentin L et al. NEJM. 2009; 361: 1045 -57

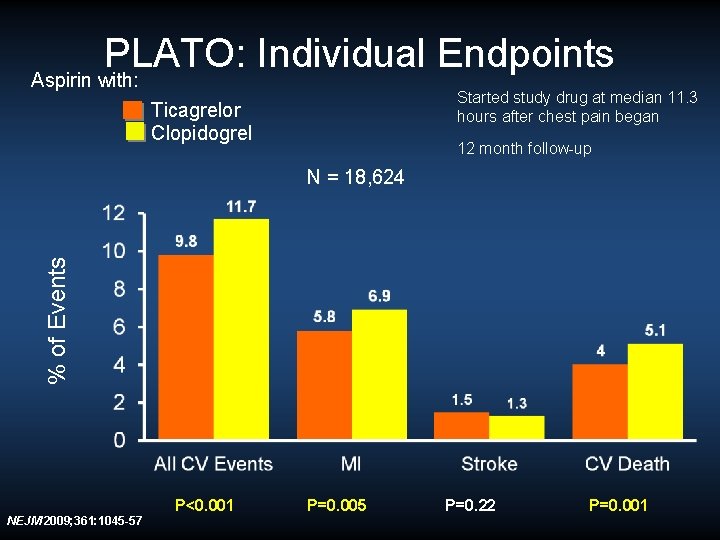
PLATO: Individual Endpoints Aspirin with: Started study drug at median 11. 3 hours after chest pain began Ticagrelor Clopidogrel 12 month follow-up % of Events N = 18, 624 NEJM 2009; 361: 1045 -57 P<0. 001 P=0. 005 P=0. 22 P=0. 001

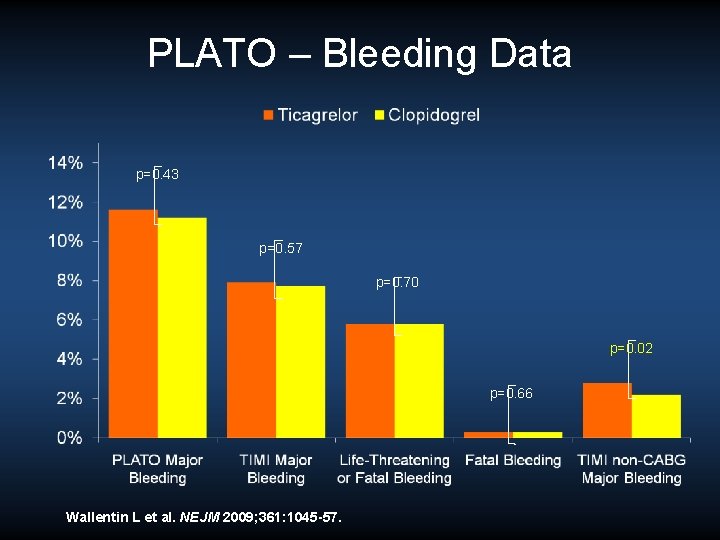
PLATO – Bleeding Data p=0. 43 p=0. 57 p=0. 70 p=0. 02 p=0. 66 Wallentin L et al. NEJM 2009; 361: 1045 -57.

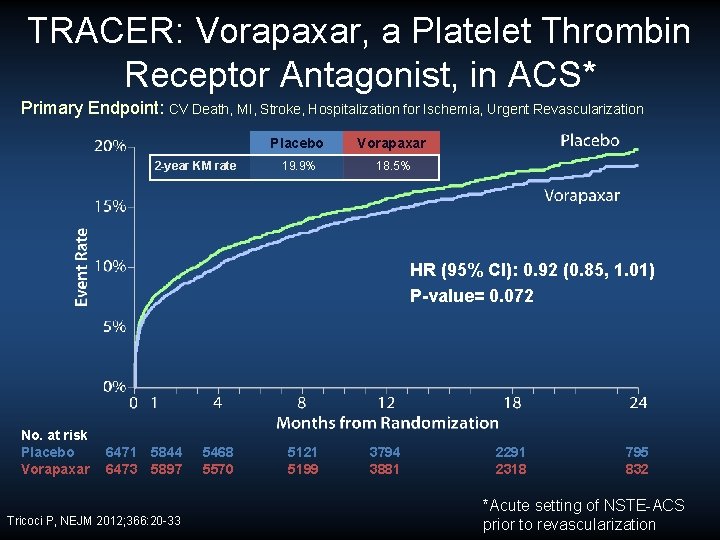
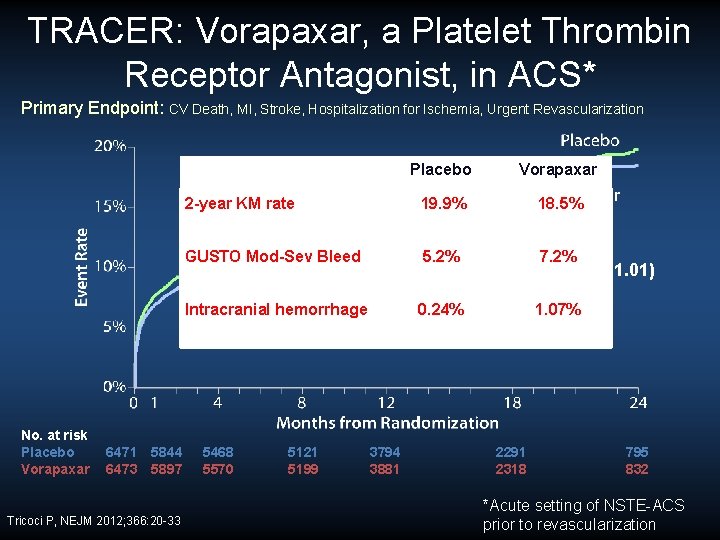
TRACER: Vorapaxar, a Platelet Thrombin Receptor Antagonist, in ACS* Primary Endpoint: CV Death, MI, Stroke, Hospitalization for Ischemia, Urgent Revascularization 2 -year KM rate Placebo Vorapaxar 19. 9% 18. 5% HR (95% CI): 0. 92 (0. 85, 1. 01) P-value= 0. 072 No. at risk Placebo Vorapaxar 6471 5844 6473 5897 Tricoci P, NEJM 2012; 366: 20 -33 5468 5570 5121 5199 3794 3881 2291 2318 795 832 *Acute setting of NSTE-ACS prior to revascularization

TRACER: Vorapaxar, a Platelet Thrombin Receptor Antagonist, in ACS* Primary Endpoint: CV Death, MI, Stroke, Hospitalization for Ischemia, Urgent Revascularization 2 -year KM rate GUSTO Mod-Sev Bleed 6471 5844 6473 5897 Tricoci P, NEJM 2012; 366: 20 -33 5468 5570 5121 5199 Vorapaxar 19. 9% 18. 5% 5. 2% 7. 2% 0. 24% 1. 07% HR (95% CI): 0. 92 (0. 85, 1. 01) P-value= 0. 072 Intracranial hemorrhage No. at risk Placebo Vorapaxar Placebo 3794 3881 2291 2318 795 832 *Acute setting of NSTE-ACS prior to revascularization

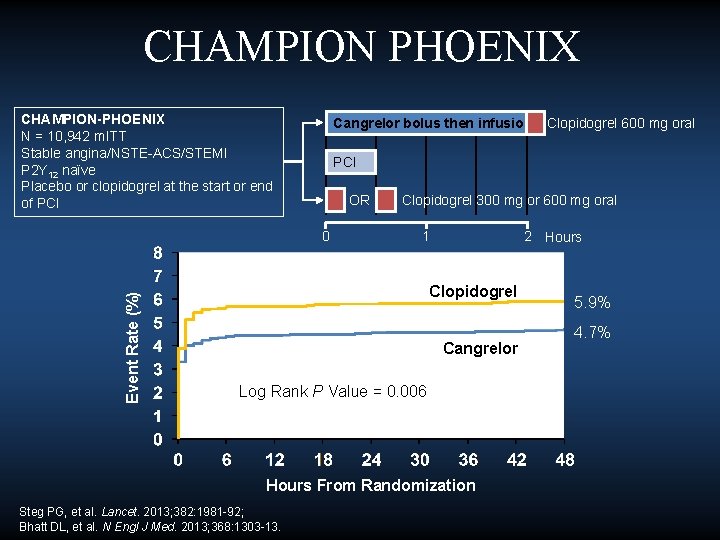
Intravenous Antiplatelet Agents GP IIb/IIIa Inhibition ADP Receptor Inhibition (abciximab, eptifibatide, tirofiban) (Cangrelor) • Improves ischemic outcomes in patients with ACS undergoing PCI • in most studies patients were treated with ticlopidine or clopidogrel - not with a potent P 2 Y 12 -receptor inhibitor • Use is limited because of bleeding complications • The role of cangrelor as an adjunctive antiplatelet therapy in patients requiring PCI has been assessed in three largescale, phase III RCTs. – CHAMPION-PCI – CHAMPION-PLATFORM – CHAMPION PHOENIX

CHAMPION PHOENIX CHAMPION-PHOENIX N = 10, 942 m. ITT Stable angina/NSTE-ACS/STEMI P 2 Y 12 naïve Placebo or clopidogrel at the start or end of PCI Cangrelor bolus then infusion PCI OR Event Rate (%) 0 Clopidogrel 300 mg or 600 mg oral 1 2 Hours Clopidogrel Cangrelor Log Rank P Value = 0. 006 Hours From Randomization Steg PG, et al. Lancet. 2013; 382: 1981 -92; Bhatt DL, et al. N Engl J Med. 2013; 368: 1303 -13. Clopidogrel 600 mg oral 5. 9% 4. 7%

What About Medical Therapy in ACS?

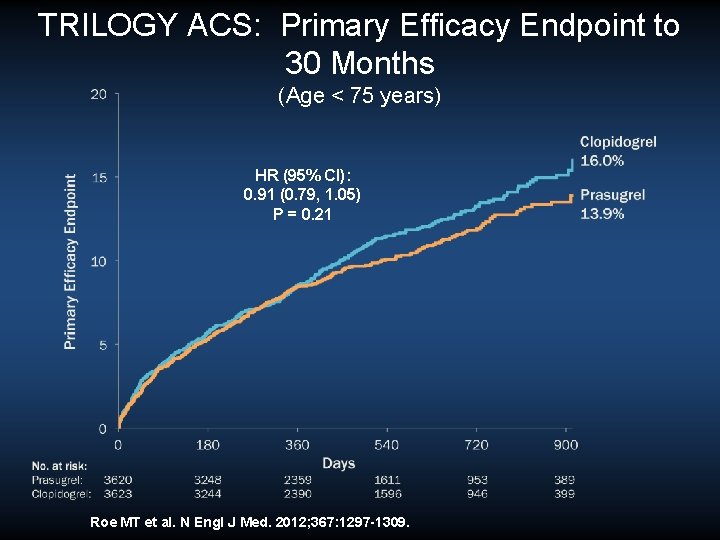
TRILOGY ACS: Primary Efficacy Endpoint to 30 Months (Age < 75 years) HR (95% CI): 0. 91 (0. 79, 1. 05) P = 0. 21 Roe MT et al. N Engl J Med. 2012; 367: 1297 -1309.

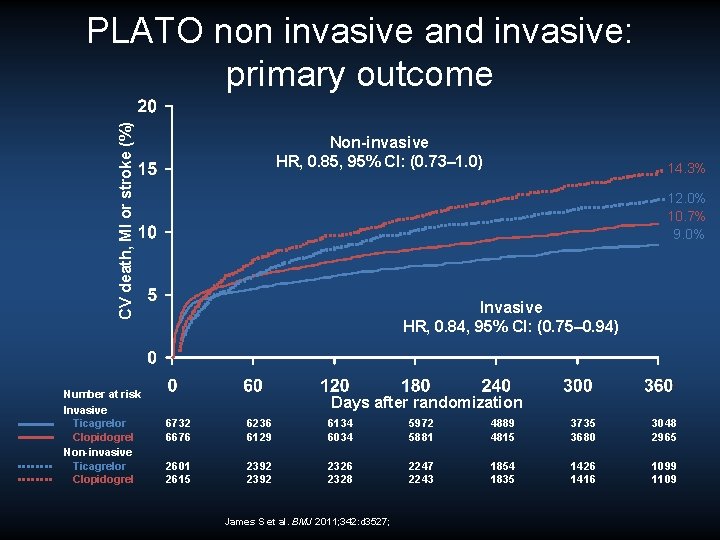
CV death, MI or stroke (%) PLATO non invasive and invasive: primary outcome Number at risk Invasive Ticagrelor Clopidogrel Non-invasive HR, 0. 85, 95% CI: (0. 73– 1. 0) 14. 3% 12. 0% 10. 7% 9. 0% Invasive HR, 0. 84, 95% CI: (0. 75– 0. 94) Days after randomization 6732 6676 6236 6129 6134 6034 5972 5881 4889 4815 3735 3680 3048 2965 2601 2615 2392 2326 2328 2247 2243 1854 1835 1426 1416 1099 1109 James S et al. BMJ 2011; 342: d 3527;

Antiplatelet Rx after ACS I IIa IIb III After PCI, aspirin should be continued indefinitely. It is reasonable to use 81 mg of aspirin daily in preference to higher maintenance doses (IIa-B). In patients receiving a stent (BMS or DES) during PCI for ACS, P 2 Y 12 inhibitor therapy should be given for at least 12 months. Options include: - clopidogrel 75 mg daily, - prasugrel 10 mg daily, or - ticagrelor 90 mg twice daily. For UA/NSTEMI patients in whom an initial conservative strategy is selected, clopidogrel or ticagrelor … should be … administered for up to 12 months. 2011 ACCF/AHA PCI Guidelines & 2012 ACCF/AHA UA/NSTEMI Focused Update

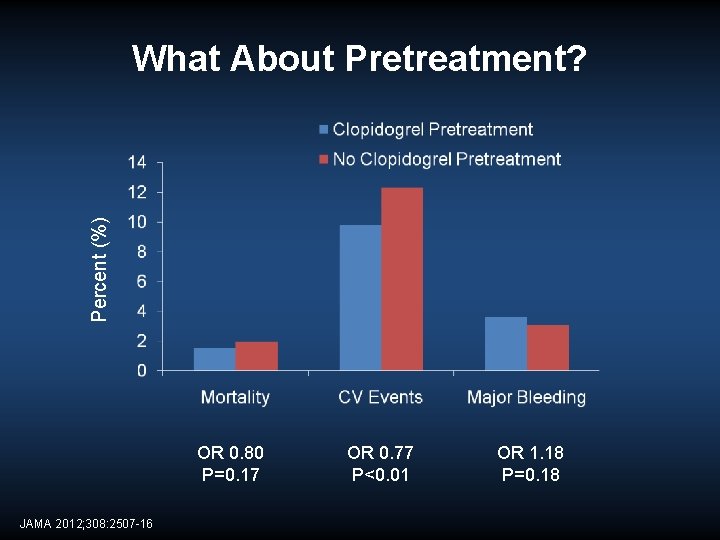
Percent (%) What About Pretreatment? OR 0. 80 P=0. 17 JAMA 2012; 308: 2507 -16 OR 0. 77 P<0. 01 OR 1. 18 P=0. 18

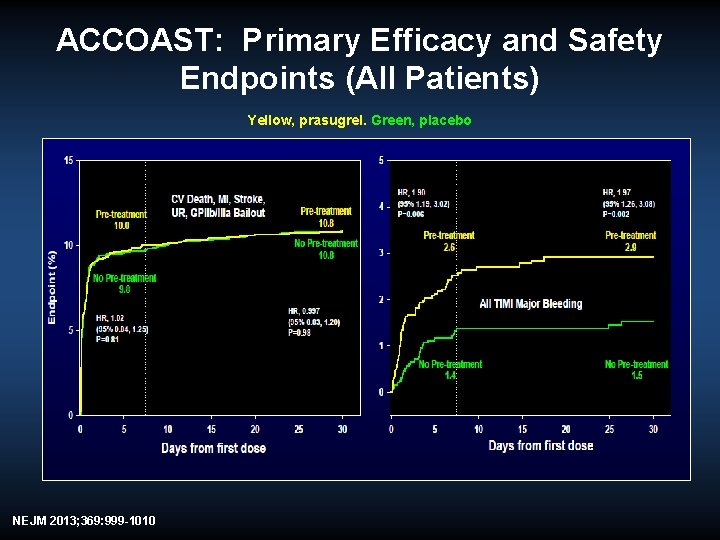
ACCOAST: Primary Efficacy and Safety Endpoints (All Patients) Yellow, prasugrel. Green, placebo NEJM 2013; 369: 999 -1010

Do We Need Aspirin in the Current Era?

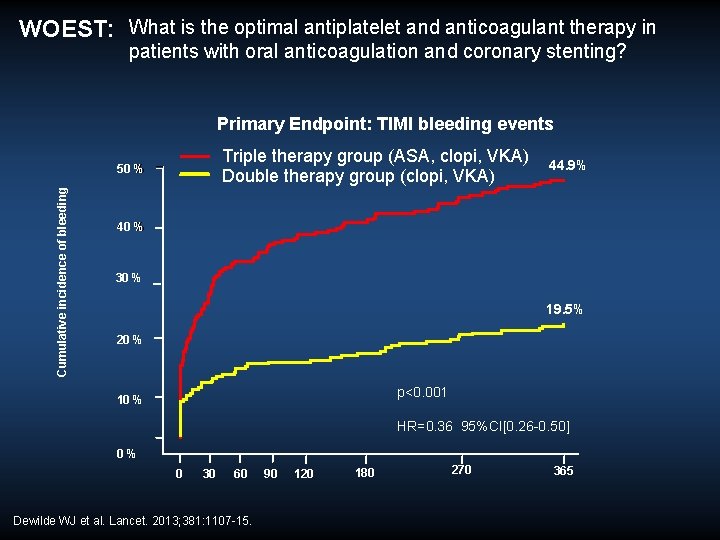
WOEST: What is the optimal antiplatelet and anticoagulant therapy in patients with oral anticoagulation and coronary stenting? Primary Endpoint: TIMI bleeding events Triple therapy group (ASA, clopi, VKA) Double therapy group (clopi, VKA) Cumulative incidence of bleeding 50 % 44. 9% 40 % 30 % 19. 5% 20 % p<0. 001 10 % HR=0. 36 95%CI[0. 26 -0. 50] 0 % 0 30 60 Dewilde WJ et al. Lancet. 2013; 381: 1107 -15. 90 120 180 270 365

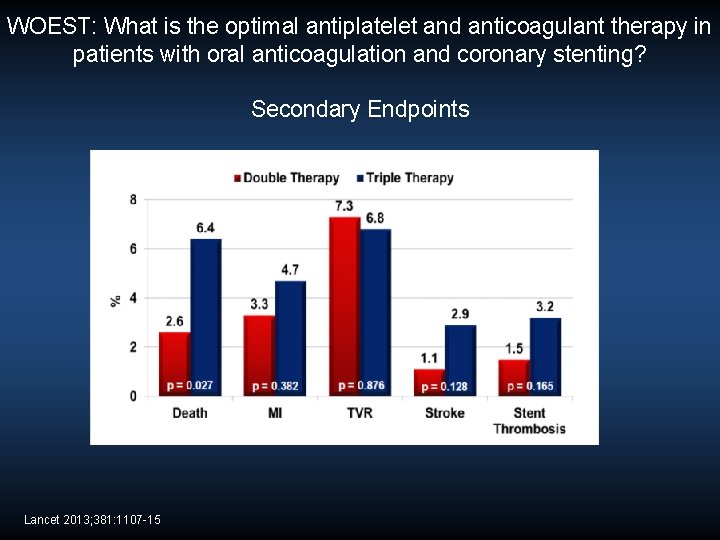
WOEST: What is the optimal antiplatelet and anticoagulant therapy in patients with oral anticoagulation and coronary stenting? Secondary Endpoints Lancet 2013; 381: 1107 -15

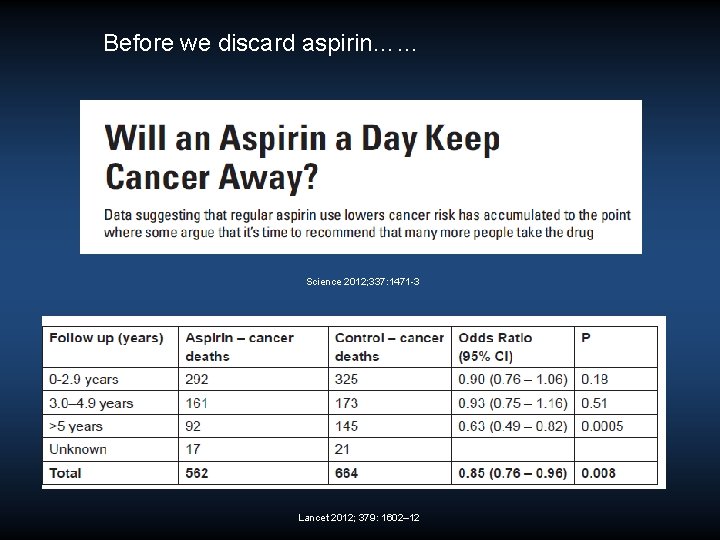
Before we discard aspirin…… Science 2012; 337: 1471 -3 Lancet 2012; 379: 1602– 12

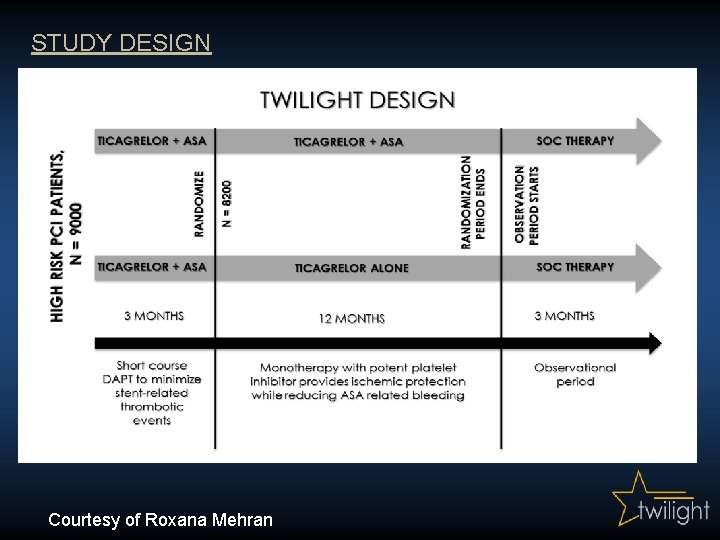
STUDY DESIGN Courtesy of Roxana Mehran

Emerging Protocols for Antiplatelet Therapy in ACS and During PCI

Established and Emerging Antiplatelet Therapies Serotonin Receptor Inhibitors GP VI Antagonists Thromboxane Antagonist GP IIb/IIIa Antagonist PAR-1 Antagonists P 2 Y 12 Inhibitors Irreversible/Reversible Intravenous/Oral Others

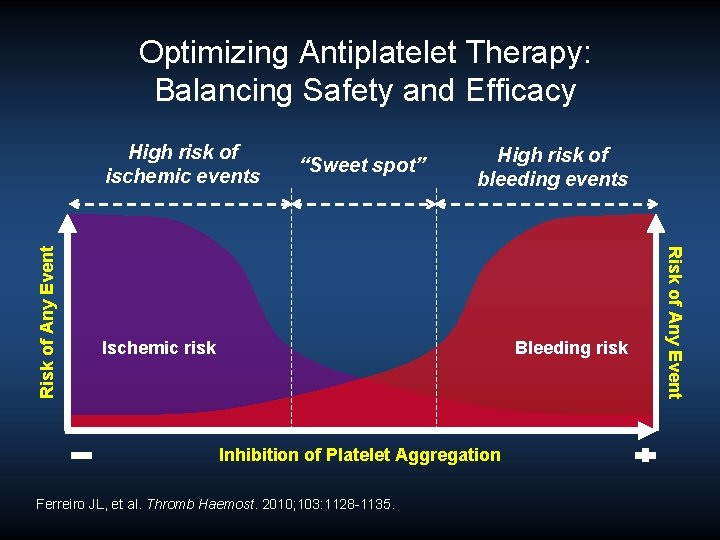
Optimizing Antiplatelet Therapy: Balancing Safety and Efficacy “Sweet spot” High risk of bleeding events Ischemic risk Bleeding risk Inhibition of Platelet Aggregation Ferreiro JL, et al. Thromb Haemost. 2010; 103: 1128 -1135. Risk of Any Event High risk of ischemic events

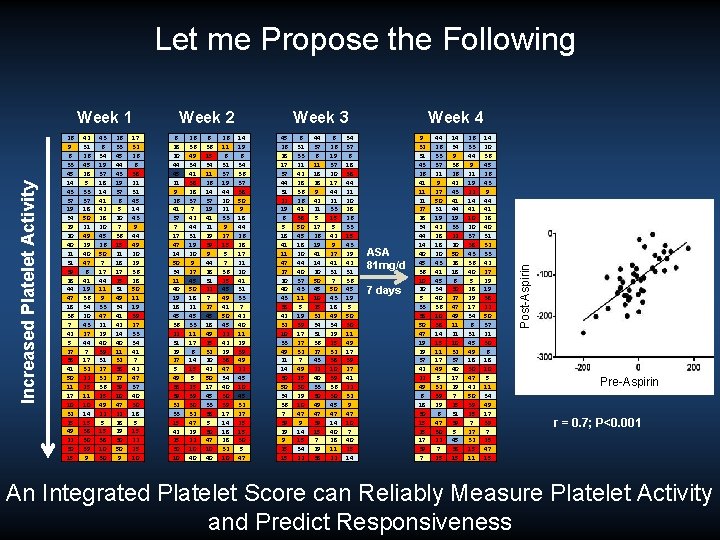
Let me Propose the Following 26 9 6 33 45 14 43 37 19 34 29 20 40 21 31 39 28 44 47 18 36 7 42 5 27 38 41 50 11 17 10 32 25 49 22 30 15 42 31 26 45 28 5 33 37 18 50 21 49 29 40 47 6 41 19 36 34 20 43 27 44 7 17 32 22 25 11 10 14 15 38 30 39 9 43 6 34 19 37 18 14 41 42 28 20 45 26 50 7 17 44 11 9 33 47 21 29 40 39 31 27 32 36 25 49 22 5 15 38 10 30 26 33 45 44 43 19 37 6 5 20 7 36 15 21 18 17 25 31 49 34 41 42 14 40 11 32 38 27 39 10 47 22 28 29 30 50 9 17 32 26 6 38 21 31 45 14 43 9 44 49 20 29 36 28 50 11 19 39 27 33 34 41 7 42 47 37 40 30 18 5 15 22 25 10 Week 2 6 28 20 44 45 21 9 26 41 37 7 17 47 14 50 34 11 40 19 18 45 36 22 31 29 27 5 49 38 39 32 33 15 42 25 30 10 26 36 49 34 41 38 28 37 7 42 44 31 19 20 9 27 45 50 18 21 45 33 11 17 6 14 15 5 25 39 30 32 47 29 22 10 40 6 36 15 34 11 26 14 37 19 41 21 29 39 9 44 28 31 22 7 27 45 18 49 25 32 20 42 50 17 45 33 38 5 30 47 10 40 26 11 6 31 37 19 44 20 21 33 9 27 15 5 7 36 25 45 49 41 50 45 22 42 29 38 47 34 40 30 39 17 14 18 28 32 10 Week 3 14 19 6 34 36 37 38 50 9 18 44 26 28 17 21 20 41 31 33 7 42 40 11 29 39 49 22 45 10 45 32 27 25 15 30 5 47 45 26 28 17 37 44 31 22 19 6 5 18 41 11 47 27 20 40 43 38 42 32 10 33 49 21 14 30 50 34 36 7 39 29 9 25 15 6 31 33 21 42 28 36 26 41 38 50 45 18 20 44 40 37 43 11 5 19 39 17 27 32 7 49 25 30 29 10 47 9 14 15 34 22 44 37 6 11 18 28 9 42 21 5 17 26 19 41 14 20 50 45 10 25 32 34 31 36 27 43 22 40 33 30 49 47 39 15 7 29 38 6 26 19 37 20 17 44 21 33 15 5 42 9 27 41 31 7 50 43 18 49 34 29 25 32 38 10 39 36 30 45 47 14 40 28 11 22 34 37 6 18 38 44 21 20 28 26 33 15 43 29 42 31 36 45 19 5 50 30 11 49 17 39 27 41 22 32 9 47 10 7 40 25 14 Week 4 ASA 81 mg/d 7 days 9 32 31 43 26 41 11 21 27 28 34 44 14 40 45 36 10 20 5 33 38 50 47 19 29 37 42 22 49 6 18 30 15 25 17 39 7 44 26 33 37 21 9 27 50 31 19 42 28 18 20 43 41 45 34 40 36 10 38 14 15 11 17 49 5 32 39 29 6 47 30 22 7 25 14 34 9 36 26 42 43 41 44 19 33 22 20 50 28 18 6 30 27 47 49 11 21 10 32 37 40 17 29 7 25 31 39 5 45 38 15 26 33 44 9 21 19 22 14 41 10 20 37 38 43 36 40 5 28 29 17 34 6 31 45 49 18 30 47 42 50 39 25 7 27 32 15 11 14 20 36 45 26 43 9 44 41 28 40 31 32 33 42 27 29 19 38 22 50 37 21 30 6 18 10 5 11 34 49 17 39 7 25 47 15 Post-Aspirin Increased Platelet Activity Week 1 Pre-Aspirin r = 0. 7; P<0. 001 An Integrated Platelet Score can Reliably Measure Platelet Activity and Predict Responsiveness

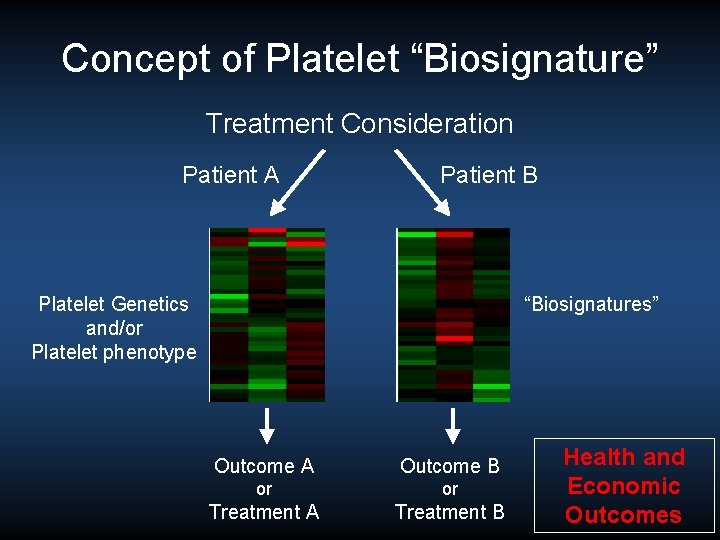
Concept of Platelet “Biosignature” Treatment Consideration Patient A Patient B Platelet Genetics and/or Platelet phenotype “Biosignatures” Outcome A Outcome B or or Treatment A Treatment B Health and Economic Outcomes

Thank You