How do we Measure Matter https www brainpop

How do we Measure Matter? https: //www. brainpop. com/science/matterandchemistry/ measuringmatter/

Weight is a measure of the force of gravity on a object. Ø In everyday life, weight is a useful measurement of how much matter an object contains. Ø On Earth, all objects are attracted downward by Earth’s gravity.

Mass is the amount of matter in an object; size. Ø Mass is not the same as weight, even though we often calculate an object's mass by measuring its weight with a spring scale. Ø Unit of measure: gram Ø An object on the Moon would weigh less than it would on Earth because of the lower gravity, but it would still have the same mass. weigh less than it Earth because of gravity, but it the same mass the


Density is how compact the mass in an object is Ø Unit of measurement: g/cm 3 Ø Density of an object does not change! Ø Density of water is 1. 0 g/cm 3. An object will float on water if its density is less than 1. 0 g/cm 3.
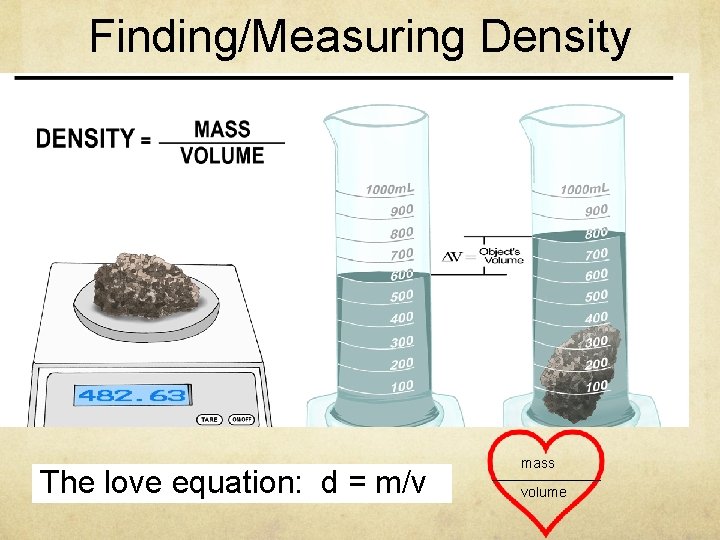
Finding/Measuring Density The love equation: d = m/v mass _______ volume

Formulas for finding Mass, Volume, and Density M D V Mass = Density x Volume = Mass/Density = Mass/Volume

Let’s measure! Problem 1 The mass of a silver bracelet is 2. 5 grams. It occupies a volume of 48 cm 3. What is its density? D = M/V D = 2. 5/48 D= 0. 05208 cm 3

Problem 2 The density of silver (Ag) is 10. 5 g/cm 3. Find the mass of Ag that occupies 965 cm 3 of space. M = D X V M = 10. 5 x 965 M = 10132. 5 g To check: 10. 5 = 10132. 5/965

Problem 3 A 2. 75 kg sample of a substance occupies a volume of 250. 0 m. L. What is its density? D = M/V D = 2. 75/250. 0 D = 0. 011 g/cm 3

Problem 4 A rectangular block of lead (Pb) measures 20. 0 mm X 30 mm X 45 mm. If the mass of the Pb is 10 g, find its density. V = 20 X 30 X 45 V = 27000 cm D = M/V D = 10/27000 D= 0. 00037 g/cm 3

Problem 5 A cube of gold (Au), which has all equal sides, has a side length of 1. 55 cm. If the sample is found to have a mass of 71. 9 g, find the density of the Au. V = 1. 55 x 1. 55 V = 3. 7238 cm D = M/V D = 71. 9/3. 7238 D = 19. 3802 g/cm 3

Problem 6 The density of 5 liquids is measured as follows: Liquid 1: 1. 0 g/ml Liquid 2: 1. 38 g/ml Liquid 3: 0. 77 g/ml Liquid 4: 2. 95 g/ml Liquid 5: 0. 056 g/ml Liquid 5 Liquid 3 Liquid 1 Liquid 2 Liquid 4 Draw a picture of all 5 liquids in a test tube how they would layer according to density.
- Slides: 13